Alcaligenes faecalis NEUF2011
A Microbial Biorealm page on the genus Alcaligenes faecalis NEUF2011
Leifson flagella stain of Alcaligenes faecalis (digitally colorized). Photograph by Dr. William A. Clark (1).
Classification
Higher order taxa
- Domain: Bacteria
- Phylum: Proteobacteria
- Class: Beta Proteobacteria
- Order: Burkholderiales
- Family: Alcaligenaceae
- Order: Burkholderiales
- Class: Beta Proteobacteria
- Phylum: Proteobacteria
Species
- Genus: Alcaligenes
- Species: faecalis
Alcaligenes faecalis
Description and significance
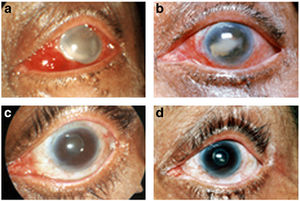
(a) The left eye shows edematous lids, congestion, and exudates in the anterior chamber on second postoperative day. (b) The eye on the eighth postoperative day after intravitreal injection showing reduction of exudates. (c) The eye with posterior capsule opacification at the end of second month. (d) The eye after Nd:Yag opening. Photograph by S Kaliaperumal et al. (6).
Alcaligenes faecalis occur in water and soil. The microbe has peritrichous flagellar arrangement which allows for motility (2). It is a gram-negative, rod-shaped organism observed at 0.5-1.0 μm x 0.5-2.6 μm in diameter. An aerobic microbe, A. faecalis is optimal at temperatures between 20-37 °C (11).
This microbe is most commonly seen in the clinical laboratory. Most infections caused by A. faecalis are opportunistic and acquired from moist items such as nebulizers, respirators, and lavage fluids. When an infection occurs, it is usually in the form of a urinary tract infection (2). However, A. faecalis is also known to be the pathogen that causes bacterial keratitis and postoperative endophthalmitis. Numerous strains have been isolated from clinical material such as blood, urine and feces (11).
A study on metabolic energy observed Alcaligenes faecalis surviving in cultures of 10 g/L of aqueous arsenic. The survival of the microbe in arsenic is important in bioremediation of environments contaminated with aqueous arsenic (12). In environments with high arsenic, the community must be wary of the likely presence of A. faecalis and its tendency to cause infections.
Genome structure

Presumed gene product lengths (in amino acids [aa]) and functions are indicated. Diagram by Silver and Phung (7).
Despite its relevance to the medical field, the complete genome of Alcaligenes faecalis, or any of the Alcaligenes species, has not been sequenced.
Only parts of the A. faecalis genome have been sequenced by researchers. Silver and Phung (2005) sequenced a 71-kb linear A. faecalis DNA region responsible for encoding arsenite oxidase as well as associated functions. More than 20 genes are located in this region, forming a "gene island" responsible for arsenic oxidation, resistance, and metabolism. These include a large molybdopterin-containing peptide subunit and a small [2Fe-2S] subunit, both responsible for arsenic oxidase.
Plasmid DNA encoding virulence and resistance has been identified in A. faecalis (9).
Cell structure and metabolism
As Alcaligenes faecalis is a gram-negative bacterium, it possesses an outer membrane (composed of lipopolysaccharides, phospholipids, and proteins), a thin peptidoglycan layer, and a periplasm. These three layers form the gram-negative cell envelope.
A. faecalis is unusual among gram-negative bacteria for it's ability to aerobically desaturate saturated fatty acids in order to produce monosaturated fatty acids (3). Higher-order organisms such as animals, protozoa, and various types of algae have this ability, while most bacteria and almost all gram-negative bacteria use an anaerobic pathway. A. faecalis has also demonstrated the ability to enzymatically metabolize arsenite (AsO2-, oxidation state +3) to the less harmful arsenate (AsO4-, oxidation state +5). This bacterium could be useful for neutralization of environments contaminated by arsenite.
A. faecalis is a heterotrophic denitrifier, meaning that it breaks down NO3, NO2-, and NH4+ using organic C as an carbon and energy source. The mechanism of NO2- conversion to NO by A. faecalis has been attributed to nitrite reductase, a copper-containing enzyme (13). In some ecosystems, much of the atmospheric NO and N2O produced by soil bacteria is a product of heterotrophic nitrification (10).
Ecology
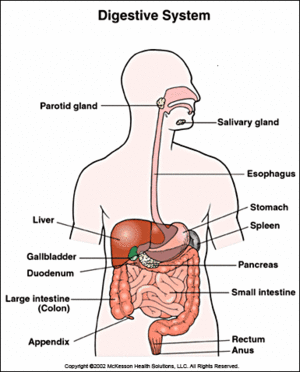
The human alimentary canal is inhabited by Alcaligenes faecalis. Diagram by UCLA Health System Transplantation Services (16).
Alcaligenes faecalis is an aerobic bacillus known to inhabit soil and water environments (5). Optimal temperature for the microbe ranges from 20 °C to 37 °C (11). A. faecalis also inhabits the alimentary canal of humans (5).
This microbe is known to contaminate medical devices in the clinical environment, which can lead to infection. In the human eye, A. faecalis is likely a cause of postoperative endophthalmitis, and it has been isolated from corneal ulcers (6). Kahveci et al. (2011) found that contamination of peritoneal dialysis catheters with the mircobe may be linked to patients developing peritonitis (5).
The microbe has been isolated from human bodily fluids in association with both open wounds and ear discharge. A. faecalis has also been found in faeces (6).
Recently, researchers have shown that a biosurfactant produced by Alcaligenes faecalis may be a promising agent to bioremediate oil spills (3).
Pathology
Alcaligenes faecalis exists in the alimentary canal, but systemic infections with the microbe do not occur in the majority of healthy humans. Infections caused by A. faecalis have been observed in humans with compromised immune systems and uncompromised immune systems. Many infections caused by the microbe are a result of contamination of medical devices. (5)
Kaliaperumal et al. (2006) have shown that A. faecalis may be a causative agent in postoperative endophthalmitis in the human eye (see figure 2). Infection with the microbe likely stems from exposure to other parts of the body or contaminated instruments. (6) Patients on endophthalmitis may experience swelling of the eyelid, redness, and permanent loss of vision (15).
Kahveci et al. (2011) found that catheters contaminated with A. faecalis may be associated with development of peritonitis in peritoneal dialysis patients. Symptoms of peritonitis include vomiting, passing little or no stool, sore and bloated abdomen, and fever. (14)
The microbe has been found to cause urinary tract infections in humans (2). In addition, A. faecalis has been found in corneal ulcers, feces, blood, and other bodily fluids (6).
In addition to infecting humans, A. faecalis is a pathogen of domesticated chickens, domesticated turkeys, and other birds. Simmons et al. (1980) were able to isolate A. faecalis from broiler chicks, and it was likely related to respiratory disease in the chicks (7).
Although A. faecalis has been isolated from human and non-human hosts, the pathogenic mechanism of the microbe is largely unknown. Many of the studies investigating the virulence of the microbe have been conducted on mammalian and avian species, and some of the proposed mechanisms may be carried out in human hosts. (6)
Current Research
- Crude biosurfactant from thermophilic Alcaligenes faecalis: Feasibility in petro-spill bioremediation(3)
Petroleum hydrocarbons are major environmental pollutants. Bioremediation at the site of contamination is considered an environmentally friendly means of petroleum hydrocarbon clean-up. Natural biodegradation in the environment is limited by the hydrophobic properties of hydrocarbons. Bharali et al. explored the use of A. faecalis to promote biodegradation of petroleum hydrocarbons (3).
A. faecalis produces biosurfactant compounds that increase the hydrophobicity of the cell surface during growth on hydrocarbons that enhances the contact with the hydrocarbons and as a result increases hydrocarbon degradation. The capacity to produce biosurfactant was studied by growing A. faecalis in salt media with a variety of hydrophobic substrates (diesel, kerosene, crude oil) as the carbon source. Under different substrate concentrations, surface tension and the rate of biosurfactant production was measured.
It was found that the biosurfactant produced by A. faecalis possessed high surface activity, decreasing surface tension adequately to allow for degradation by the microorganism. In addition to the surface activity of the biosurfactant, it was found to be stable over high temperatures, a range of pH and different salt concentrations, and also possessed antimicrobial properties against a variety of bacteria and fungi. The versatile nature of the biosurfactant produced by A. faecalis makes A. faecalis an excellent candidate for use in bioremediation of hydrocarbon pollutions such as oil spills.(3)
- Improvement in ammonium removal efficiency in wastewater treatment by mixed culture of Alcaligenes faecalis No. 4 and L1(4)
Ammonium removal is a required step in the treatment of wastewater. Typically this process is completed by the use of autotrophic nitrifiers and heterotrophic denitrifiers in two separate (aerobic and anaerobic) tanks. The growth of autotrophic nitrifiers is very slow requiring a long hydraulic retention time in treatment plants. Alcaligenes faecalis has been identified as a heterotrophic nitrifier and aerobic denitrifier and converts ammonium into denitrification products simultaneously. Unfortunately the ammonium removal rate of heterotrophic nitrifiers is lower than that of autotrophic nitrifiers, alternatively the growth of heterotrophic bacteria is faster than that of autotrophs.
Joo et al (2006) explored the efficiency of a strain of A. faecalis with high denitrification behavior isolated from their laboratory called A. faecalis no. 4, combined with a mutant strain of A. faecalis, L1, that has a higher ammonium removal rate and a weaker denitrification ability than no. 4 in wastewater treatment. The aim of the study was to confirm the improved efficiency of the mixed culture that takes advantage of the high ammonium removal capacity of L1 and the high denitrification capacity of no. 4.
Joo examined the change in efficiency by comparing the ammonium removal and denitrification by the combined L1 and no. 4 cultures with single cultures of each strain. Joo found that in the mixed culture the ammonium removal rate was twofold higher than that in a single culture of no. 4 and similar to the rate in the single culture of L1. The denitrification rate of the combined culture was exceptionally high compared to the single culture of L1. In addition to the improved ammonium removal, Joo found that the number of viable cells in the mixed culture decreased compared to the single cultures resulting in reduced sludge production, the accumulation of which is a major challenge in wastewater treatment by microorganisms. Joo et al concluded that the combined single tank use of A. faecalis strains L1 and no. 4 is an improved alternative to the current methods of ammonium removal in wastewater (4).
Interesting Fact
Peritonitis (inflammation of the thin tissue that lines the inner wall of the abdomen and covers most of the abdominal organs) is commonly found in peritoneal dialysis (PD) patients due to contamination of the dialysis catheter. The most common pathogens causing peritonitis in PD patients are gram positive (Staphylococcus epidermidis and Staphylococcus aureus). Recently there have been unusual cases of A. faecalis causing peritonitis in PD patients. A. faecalis is not only a Gram negative bacterium but also an environmental organism. Both of these characteristics are rarely found to cause such significant infections and has since been identified as a pathogen of consideration in the treatment of clinical peritonitis cases.(5)
References
2. Winn, W., Sommers, H., Koneman, E., Janda, W., Dowell, V., and Allen, S (1988). "Color Atlas and Textbook of Diagnostic Microbiology". 'J.B. Lippincott Company'. Edition 3: 184, 200-201.
3. Bharali, P., Das, S., Konwar, B.K., and Thakur, A.J. (2001) Crude biosurfactant from thermophilic Alcaligenes faecalis: Feasibility in petro-spill bioremediation. Internation Biodeterioration & Biodegradation 65: 682-690.
4. Joo, H., Hirai, M., and Shoda, M. (2006) Improvement in ammonia removal efficiency in wastewater treatemnt by mixed culture of Alcaligenes faecalis No. 4 and L1. Journal of Bioscience and Bioengineering. 103(1): 66-73.
5. Kahveci, A., Asicioglu, E., Tigen, E., Ari, E., Arikan, H., Odabasi, Z., and Ozener, C. (2011) Unusual causes of peritonitis in a peritoneal dialysis patient: Alcaligenes faecalis and Pantoea agglomerans. Annals of Clinical Microbiology and Antimicrobials 10: 12.
8. Silver, S. & Phung, L.T. (2005) Genes and Enzymes Involved in Bacterial Oxidation and Reduction of Inorganic Arsenic. Applied and Environmental Microbiology 71(2): 599-608.
9. Luginbuhl, G.H., Rader, J.M., and Simmons, D.G. (1984). Evidence for plasmid DNA in Alcaligenes faecalis. Am J Vet Res 45(8): 1679-81.
10. Anderson, I.C., Poth, M., Homstead, J., Burdige, D. (1993). A Comparison of NO and N20 Production by the Autotrophic Nitrifier Nitrosomonas europaea and the Heterotrophic Nitrifier Alcaligenes faecalis. Applied and Environmental Microbiology 59(11): 3525-3533.
11. Austin, B., Colwell, R.R., Forns, J.M., and Rodgers, C.J. (1981). Alcaligenes faecalis subsp. homari subsp. nov., a new group of bacteria isolated from moribund lobsters. International Journal of Systematic Bacteriology 31(1): 72-76.
12. Muller, D., Médigue, C., Koechler, S., Barbe, V., Barakat, M., Talla, E., & ... Makita, Y. (2007). A Tale of Two Oxidation States: Bacterial Colonization of Arsenic-Rich Environments. Plos Genetics, 3(4): 53-66.
13. Murphy M.E., Turley S., Kukimoto M., Nishiyama M., Horinouchi S., Sasaki H., Tanokura M., Adman E.T. (1995). Structure of Alcaligenes faecalis nitrite reductase and a copper site mutant, M150E, that contains zinc. Biochemistry 34(38): 12107-17.
14. Bhimji, S. Peritonitis. PubMed Health. U.S. National Library of Medicine. Web. 20 Oct. 2011.
- Created and Edited by Kevin Wieczerza, Stephanie Freed, Amanda McKenzie, Hughes Burridge -- Students of Dr. Iris Keren
