Chronic Salmonella Typhi Infection and Gallbladder Cancer
Introduction
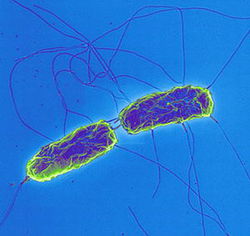
There is a strong correlation between chronic Salmonella Typhi (S. enterica enterica serovar Typhi) infection and gallbladder cancer.3 S. Typhi, a rod shaped, flagellated, aerobic, Gram-negative bacterium (Fig. 1) is a pathogenic serovar of the S. enterica enterica subspecies. Its pathogenicity is restricted to humans, and it is reported to cause 21 million acute cases of acute typhoid fever annually, with 200,000 fatalities.1 Bacteria invade the mucosal surface of the intestine but spread to deeper tissues such as liver, spleen, and bone marrow after phagocytosis by macrophages.1 They can also spread to the gallbladder via ducts from the liver during enterohepatic circulation.2
A small percentage of the individuals who suffer acute infection—between 3 and 5%—become asymptomatic carriers whose infections persist for many years following the acute illness.7 S. Typhi achieves this persistent carrier state by creating biofilms on cholesterol-based gallstones residing within the gallbladder of the infected individual. This carrier state requires both presence of cholesterol gallstones and S. Typhi infection of the gallbladder. Thus, few of the individuals who suffer acute infection are at risk for chronic carriage.2 The first widely described carrier was Mary Mallon, or “Typhoid Mary”, a food service worker who was both asymptomatic and highly contagious. S. Typhi bacteria are transmitted among individuals—with either acute or chronic infections—primarily through fecal contamination of food or water.7
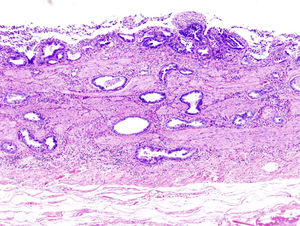
The carrier state can lead to chronic inflammation of the gallbladder, in which the bacteria metabolize primary bile acids to produce potentially carcinogenic toxins and metabolites. One such carcinogen producer is bacterial β-glucuronidase, a glycosidase whose action produces mutagenic intermediates. These compounds act in addition to the other secondary bile acids that are produced by bacterial enzyme processing then concentrated within the gallbladder.4 The result of toxic chemical environment is carcinoma of the gallbladder epithelium.3(Fig. 5). One defining feature of the gallbladder is its efficacy in concentrating not only bile salts but also toxins—an effect that amplifies their mutagenic effects. Thus, carcinoma develops here instead of in other organs that are implicated in chronic S. Typhi infection. Epidemiologically, the chronic typhoid carrier state has been demonstrated to be the single most important risk factor for gallbladder cancer in patients with cholesterol-based gallstones.3
Chemical characteristics of the gallbladder and bile salts
The gallbladder is an accessory organ that concentrates and stores the primary bile acids produced in the liver. Chemically speaking, primary bile salts mostly consist of variations of cholic and deoxycholic acids (Fig. 2) in complex with sodium. They are stored in the gallbladder and concentrated markedly, reaching levels exceeding 10-15% of total gallbladder contents.5
Though bile is a detergent that is generally toxic to bacteria in such high concentrations, S. Typhi has unique resistance to this toxicity and detergent action. This is due in part to the protection provided by genes such as the PhoP-PhoQ virulence factor. In addition to activating and repressing the production of both membrane and secreted proteins, this regulator has also been implicated in lipopolysaccharide (LPS) modifications. All of these contribute to a bile-resistant phenotype in vivo. This resistance applies also to secondary bile acid products, which include deoxycholic acid and conjugated forms of cholic and chenodeoxycholic acids.20(Fig. 2)
Adaptations of S. Typhi to the gallbladder niche

Role of cholesterol gallstones and biofilms in chronic S. Typhi carriage
S. Typhi colonizes the gallbladder and persists in an asymptomatic carrier state5 that is facilitated by formation of biofilms on cholesterol gallstones.6 These gallstones can be induced experimentally in mice with a lithogenic diet, which is supplemented with 1% cholesterol and 0.5% cholic acid. Significant amounts of S. Typhi can be recovered from cholesterol gallstones removed from infected individuals. The presence of bacteria on these gallstones is clearly visualized with electron microscopy (Fig. 3,6). Furthermore, bacteria can be recovered from stool samples of S. Typhi infected mice with cholesterol gallstones—but not infected mice without gallstones—for over a year.6 This is due to the continuous shedding of planktonic cells from the sessile, matrix-bound population within the biofilm. Epidemiologically speaking, this is the mechanism of contagion through subsequent contamination of food and water, particularly in less developed countries.7
Significantly, it also took much longer for mice with cholesterol gallstones to recover from symptoms of acute S. Typhi infection. Consistent results demonstrating S. Typhi persistence have been obtained on cholesterol substrates in vitro, affirming the specific role of cholesterol in this process.6
The O-antigen capsule is crucial to this specific binding affinity between S. Typhi bacteria and cholesterol, and it is required for biofilm formation. The O-antigen capsule is the key component of the exopolysaccharide (EPS) matrix, which provides rigidity to the biofilm and also protects the bacteria that it contains. The operon involved in the process of creating this capsule, yihVW, contains the yihP gene that codes for a symporter enzyme implicated in O-antigen production. This operon is furthermore upregulated when S. Typhi is grown in a concentrated bile environment. Thus, O-antigen capsule production is not only crucial to biofilm production but also bile induced.7
In the presence of bile, S. Typhi bacteria form loose, multilayer matrices on cholesterol surfaces. On heterogeneous gallstones, S. Typhi does not cover the non-cholesterol surfaces (Fig. 3), which further suggests binding specificity. Benefits conferred by biofilm formation include protection from antibiotic treatment as well as increased ability to tolerate the bile-rich gallbladder environment. Strains that are unable to form the O-antigen have been demonstrated to be unsuccessful in biofilm formation.19,8 The O-antigen capsule has accordingly been identified as a potential therapeutic target in multiple studies.7
Mutagenic and carcinogenic effects
Bile mutation of S. Typhi increases its fitness to niche
Bile salts are able to damage DNA, and the subsequently mutagenic effect on S. Typhi bacteria contributes to their adaptation to the gallbladder niche.9
One proposed mechanism of S. Typhi mutation relates to oxidative damage. Another involves the conjugation that characterizes primary bile salt chemistry.20 Primary bile acids are normally deconjugated during bacterial processing in the intestine. Deoxycholic and lithocholic acids, for example, are released following these processes (Fig. 2). They have been linked to tumor genesis in humans when present in abnormally high levels. S. Typhi, when in a concentrated bile environment, produces multiple altered proteins and becomes apoptosis resistant in response to deoxycholic acid and its derivatives.20(Fig. 4).
S. Typhi survival within the gallbladder niche environment can thus be increased through genome rearrangements and polymorphisms that select for changes resulting in increased fitness of S. Typhi.9 Analysis of the frequency of S. Typhi genome rearrangements following exposure to bile salts shows that these mutations tend to occur in several common locations and have characteristic manifestations. For example, specific S. Typhi gene targets that bile mutates under very high concentrations in vivo include DNA adenine methylase. This enzyme normally works to decrease bile sensitivity and reduce the rate of mutation in S. Typhi bacteria. Thus, its mutation can lead to further mutations and resilience of damaged bacteria.10
S. Typhi metabolizes bile to produce compounds that are carcinogenic to human cells
Although bile is not generally characterized as a strong mutagen in an absolute sense, the high concentration and long exposure time associated with chronic carriage contribute demonstrably to mutagenic potential and lead to cancerous changes of the gallbladder epithelium.10(Fig. 5) By metabolizing primary bile salts prior to their secretion into the small intestine, in addition to the cholesterol from the gallstones, S. Typhi bacteria themselves are capable of producing compounds that mutate the gallbladder epithelium.9 Increased deoxycholate levels, for example, have been reported to change S. Typhi protein production. This secondary bile salt itself also leads to pathogenicity in human epithelial cells.20
Chemical mechanisms of S. Typhi mutagenic action
Chemical mechanisms have been proposed for the bile salt metabolism that produces carcinogenic compounds in long-term S. Typhi carriers. For example, β-glucuronidase enzyme action can lead to deconjugation of conjugated primary bile acids, rendering toxic metabolites that are carcinogenic to the host and also present in high concentrations.4 The final products are non carcinogenic, but the process includes a high-energy, active intermediate compound. This intermediate acts by binding to DNA and has mutagenic potential in human epithelial cells.12,13
S. Typhi also produces two other key genotoxic compounds with potentially carcinogenic roles: cytolethal distending toxin B (CdtB)—which is the functional unit of cytolethal distending toxin (CDT)—and cytotoxic necrotizing factor 1 (CNF1). CdtB is a DNAase homolog, and it is the functional unit necessary for CDT expression. Through the CDT mechanism, S.Typhi is able to create DNA lesions in target cells—including, potentially, cells of human hosts—that result in pathogenesis.4,14 CNF1 blocks cytokines, thus leading to inflammation and inhibition of the cell cycle. Furthermore, it modifies proteins from the Rho family, which normally act to terminate transcription in prokaryotes.4
In summary, bacterial enzymes found in S. Typhi act on primary bile acids to produce toxic metabolites, as well as secondary bile acids, in high concentrations. This process leads to pathology of the gallbladder epithelium. A main concern here is high concentration of biliary deoxycholate, a secondary bile acid demonstrated to be present in significantly elevated levels in gallbladder carcinoma patients.4
Another proposed mechanism of S. Typhi mutagenicity relates to interactions with the cholesterol that actually forms the structural basis of the gallstones. These bacteria do not only alter bile salts to secondary bile forms but also convert the cholesterol itself into carcinogenic compounds, including cholesterol 5alpha,6alpha-epoxide, which causes pathogenic changes in epithelial cells.11(Fig. 5) Further studies demonstrate that S. Typhi bacteria are capable of metabolizing primary bile acids to similarly mutagenic cholic acid derivative forms in the presence of bile and cholesterol substrates.12
Epidemiological links between chronic S. Typhi carriage and gallbladder cancer
Studies performed in both endemic and non-endemic typhoid regions
The relationship between chronic S. Typhi carriage and gallbladder cancer has been researched and characterized in sites worldwide, including endemic typhoid regions such as India and Mexico as well as non-endemic regions such as Scotland and the United States. These data establish a link among cholesterol-based gallstones, S. Typhi carriage, and biofilm presence. One major case-control study performed in India—which has a very high typhoid incidence—found that gallbladder cancer patients had significantly higher incidences of S. Typhi than controls and cholelithiasis patients did, at 29.4%. Furthermore, the risk of developing gallbladder carcinomas in these typhoid carriers was 8.47 times higher than it was in non-carriers.15 Furthermore, as found in a study conducted in Mexico, typhoid carriage and biofilms were identified in 4.9% of surgically removed gallstones, but neither was present without the other—thus affirming that the mechanism of carriage is biofilm formation.6(Fig. 6)

The specificity of the niche gallbladder environment and the long infection time are key aspects of S. Typhi mutagenicity. A study conducted in Scotland thirty years following the 1964 typhoid outbreak found that 16% of acutely infected individuals had become chronic carriers. Furthermore, these individuals were 167 times more likely to develop gallbladder cancer than were patients who had suffered acute infections but not chronic carriage. Although risk of developing other cancers of digestive system organs was also elevated in this population, this elevation was less intense by several orders of magnitude on a logarithmic scale, thus stressing the specificity of carcinogenicity to the gallbladder.16 An earlier study conducted with diverse American populations corroborates this specificity, suggesting furthermore that variations of bile salts act as carcinogens within the gallbladder, bile duct, and small bowel. The most marked finding of this study, however, was that individuals identified as chronic typhoid carriers died of hepatobiliary cancer six times more often—a significant difference—than the control subjects did.17
Epidemiological links among S. Typhi carriage, secondary bile levels, and gallbladder carcinoma
There is a clear epidemiological link between degradation of primary bile acids in the gallbladder and S. Typhi carriage in gallbladder carcinoma patients. When patients presenting with gallbladder carcinoma were compared to patients presenting only with gallstones, a study conducted in India identified S. Typhi bacteria in the bile of 40% of gallbladder carcinoma patients and 30% of cholelithiasis patients. However, cancer patients but not cholelithiasis patients had significantly elevated secondary bile acid levels—specifically, lithocholate and deoxycholate, which are both linked to carcinogenicity in humans when present in atypical locations or at high concentrations.18 This finding affirms the role of highly concentrated secondary bile acids—which S. Typhi can produce—in gallbladder carcinoma.
Conclusion
Long-term S. Typhi carriers are uncommon and asymptomatic but have a significantly elevated risk of developing gallbladder carcinoma.3 S. Typhi bacteria survive in the gallbladder niche by forming biofilms on cholesterol gallstones. This survival is favored by structural defenses such as the O-antigen capsule, which facilitate biofilm formation, as well selective pressure exerted by mutagenic effects of bile salts on S. Typhi itself over longer periods of time.7 Finally, S. Typhi itself has a mutagenic effect on the gallbladder epithelium by metabolizing bile salts into carcinogenic secondary bile compounds and other genotoxins.10 This connection has been characterized pathologically and epidemiologically by studies performed worldwide, in which the chronic S. Typhi carrier state constitutes a key risk factor for gallbladder carcinoma.
References
1 Vladoianu IR, Chang HR, and Pechere JC. (1990). Expression of host resistance to Salmonella typhi and Salmonella typhimurium: bacterial survival within macrophages of murine and human origin. Microb. Pathog. 8: 83–90.
2 Hornick RB, et al. (1970). Typhoid fever: pathogenesis and immunologic control. N. Engl. J. Med. 283, 686–691
3 Dutta U, Garg PK, Kumar R, and Tandon RK. (2000). Typhoid carriers among patients with gallstones are at increased risk for carcinoma of the gallbladder. Am. J. Gastroenterol. 95:784-787.
4 Nath G, Gulati AK, and Shukla VK. (2010). Role of bacteria in carcinogenesis, with special reference to carcinoma of the gallbladder. World J. Gastroenterol. 16:5395-5404.
5 Gonzalez-Escobedo G, Marshall JM, and Gunn JS. (2011). [http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3255095/ Chronic and acute infection of the gall bladder by Salmonella Typhi: understanding the carrier state. Nature Rev. Microbiol. 9:9-14.
6 Crawford RW, et al. (2010). Gallstones play a significant role in Salmonella spp. gallbladder colonization and carriage. Proc. Natl. Acad. Sci. U.S.A. 107:4353-4358.
7 Crawford RW, Gibson DL, Kay WW, and Gunn JS. (2008). Identification of a bile-induced exopolysaccharide required for Salmonella biofilm formation on gallstone surfaces. Infect. Immun. 76:5341-5349.
8 Gibson DL, et al. (2006). Salmonella produces an O-antigen capsule regulated by AgfD and important for environmental persistence. J. Bacteriol. 188:7722–7730.
9 Prieto AI, Ramos-Morales F, and Casadesús J. (2006). Repair of DNA damage induced by bile salts in Salmonella enterica. Genetics. 174:575-584.
10 Prieto AI, Ramos-Morales F, and Casadesús J. (2004). Bile-induced DNA damage in Salmonella enterica. Genetics 168:1787-1794.
11 Chipman JK. (1982). Bile as a source of potential reactive metabolites. Toxicology. 25:99-111.
12 Connor TH, Forti GC, Sitra P, and Legator MS. (1979). Bile as a source of mutagenic metabolites produced in vivo and detected by Salmonella typhimurium. Environ. Mutagen. 1(3):269-276.
13 Kinoshita N and Gelboin HV. (1978). Beta-glucuronidase catalyzed hydrolysis of benzoapyrene-3-glucuronide and binding of DNA. Science. 199:307-9.
14 Haghjoo E and Galán JE. (2004). Salmonella typhi encodes a functional cytolethal distending toxin that is delivered into host cells by a bacterial-internalization pathway. Proc. Natl. Acad. Sci. USA. 101:4614-4619.
15 Shukla VK, Singh H, Pandey M, et al. (2000). Carcinoma of the gall bladder is it a sequel of typhoid? Dig. Dis. Sci. 45:900-903.
16 Caygill C, Hill M, Braddick M, and Sharp J. (1994). Cancer mortality in chronic typhoid and paratyphoid carriers. Lancet. 343:83-84.
17 Welton JC, Marr JS, and Friedman SM. (1979). Association between hepatobiliary cancer and typhoid carrier state. Lancet. 313(8120):791-794.
18 Pandey M, Vishwakarma RA, Khatri AK, et al. (1995). Bile bacteria and gall bladder carcinogenesis., J Surg. Oncol. 58:282-283.
19Prouty AM, Schwesinger WH, and Gunn JS. (2002). Biofilm formation and interaction with the surfaces of gallstones by Salmonella spp. Infect. Immun. 70:2640–2649.
20van Velkinburgh JC and Gunn JS. (1999). PhoP-PhoQ-regulated loci are required for enhanced bile resistance in Salmonella spp. Infect. Immun. 67:1614–1622.
21Langridge GC, Phan MD, Turner DJ, Perkins TT, Parts L, et al. (2009). Simultaneous assay of every Salmonella Typhi gene using one million transposon mutants. Genome Res. 19:2308–2316.
Edited by Hannah Moore, a student of Nora Sullivan in BIOL187S (Microbial Life) in The Keck Science Department of the Claremont Colleges Spring 2013.