Bacterial ecosystems of Deepwater Horizon Oil Spill Site
Introduction
Since the “Deepwater Horizon (DWH) drilling rig explosion” incident occurred in the Gulf of Mexico on April 20, 2010, this largest marine “oil spill” in the world has imposed great dangers to the environment and to humans [1] . The oil discharged was estimated at approximately 206 million gallons [2] . Human harm, caused by “oil spills” can be acute or long-termed. Acute exposure to polycyclic aromatic hydrocarbons, such as benzene and toluene can cause skin, respiratory, and neurological issues [2] . Chronic toxic chemical enters the food chain and ultimately affects humans through seafood consumption [3] . To solve this environmental disaster, researchers have launched microbial studies centered on bioremediation using microorganisms.
Physical Environment
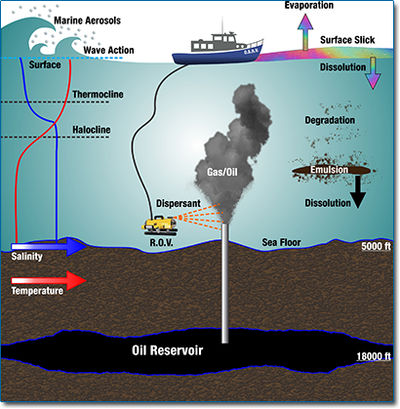
Due to the “oil spills”, the polluted sites in general showed an increase in hydrocarbon concentrations followed by increases in microorganisms and decreases in the oxygen content of the water [6] . The concentration of compounds from the “crude oil” decreased with their respective distances to the spill site. The rate of the bacterial degradation process depended on nutrients, oxygen, temperature, and pressure [8] .
The hydrocarbon compounds in crude oil are composed mostly of n-alkanes, polycyclic aromatic hydrocarbons ( “PAHs” ), alkylated PAHs, “BTEX” [4] [5] . At spill site, a large amount of methane was also released [6] and the water concentration of trace metals, especially Al, Fe and Mn, increased significantly [4] .
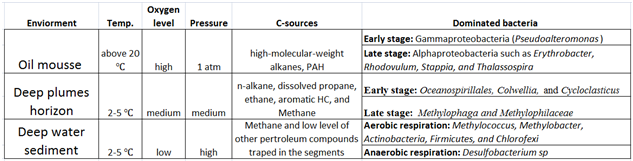
On the sea surface, hydrocarbon existed in the form of oil mousse [7] in an oxygen rich environment. Petroleum hydrocarbons were subjected to high temperature and strong irradiance [9] . Low-molecular-weight alkanes compound were vaporized quickly and most of the remaining pollutants were high-molecular-weight alkanes compounds (n>20) and “PAHs” [4] . Since the petroleum hydrocarbon was preferentially degraded aerobically [4] , oil compounds were degraded rapidly on the sea surface. Subsurface Deep hydrocarbon plumes at depths of 900−1300 m formed, consisting of dissolved gases (C1−C5 alkanes) and monoaromatics, buoyant oil droplets that retained some insoluble hydrocarbon fractions [10] . This increase in hydrocarbons as energy sources caused a blooming of the bacterial population in the deep plume horizon [11] and had lower PO43- , NO3-and dissolved O2 concentrations, and slightly higher NH4+ concentrations [13] . The temperature below 700m was always around 2-5 ℃in the Gulf of Mexico [14] . For deep waters and sediments, oil was exposed to different physical conditions with low temperature, low oxygen and high pressure [12] .
Ecosystem Process
Sea Surface
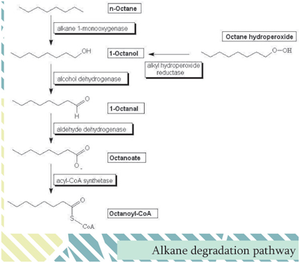
At Sea surface, evaporation and dissolution appeared to be more important than biodegradation for low molecular petroleum molecules [14] . Volatile organic compounds such as BTEX and low-molecular-weight alkanes quickly evaporated into the atmosphere leaving high-molecular-weight alkanes (n>20) and PAHs behind [4] [14] . The alkanes and PAHs were degraded aerobically by aerobic organisms involving the enzyme, “oxygenase” [8] . Often oxygenase and subsequent reactions yielded alcohols and dihydro- aromatics such as catechol which are destabilized compounds for further degradations.
Deep Plumes Horizon
At the deep plumes horizon, the major process was illustrated to be also the aerobic degradation of hydrocarbons based on the dominance of aerobic bacteria in these ecological niches [11] . After hydrocarbon was released, the bacterial community degraded them in the following order: n-alkane, dissolved propane, ethane, aromatic HC, and Methane [11] .
Water Sediment
In deep water sediment, both the aerobic and anaerobic degradation process presented at a much slower rates [12] . For the anaerobic process, different terminal electron acceptors could be used, such as nitrate, sulphate, and Fe (III) [16] .
Key Microorganisms
Sea Surface
In the oil mousses, aerobic bacteria that are tolerant to high temperatures and strong irradiance dominated. The majority were Gammaproteobacteria (93% “Pseudoalteromonas” ) at the spill site [12] . As oil was weathered gradually moving away from the accident site, Alphaproteobacteria such as “Erythrobacter” , Rhodovulum, Stappia, and Thalassospira became more dominant at the later stage of oil degradation [12] .
Deep Plumes Horizon
In the deepwater plume, psychrophilic aerobic Gammaproteobacteria dominated soon after the “oil spill” [11] . “Oceanospirillales” , Colwellia, and“Cycloclasticus” degraded alkanes, [propane, ethane, and higher alkane-derived intermediates] and aromatics respectively [11] . “Methylotrophs” , Methylophaga and Methylophilaceae, increased in number after the concentration of alkanes, alkane-derived intermediates, and aromatics significantly decreased [11] .
Water Sediment
The bacterial communities in oil-contaminated sediments were highly diversified [12] .The relatively enrichment of the Methylococcus, Methylobacter, Actinobacteria, Firmicutes, and Chlorofexi bacteria resembled the bacteria communities found in nature cold-seep sediments with gas hydrates [12] . Desulfobacterium sp., a sulfate-reducing bacterium also presented in sediments suggested that there were anaerobic degradation of petroleum hydrocarbons [12] .
Major Findings
The “DWH oil spill” was very different from many other “oil spills” in terms of oil volume, the nature of the oil, and the environments impacted. Therefore, it was an unique model for field studies. Many Researches focusing on monitoring biodegradation and improving bioremediation arose from the “DWH oil spill” .
The field and laboratory tests also gave many valuable data for bioremediation application. In terms of toxicity studies, Kujawinski EB et al. in 2011 investigated the toxicological effects of the injection of 771,000 gallons (2,900,000 L) of chemical dispersant, “DOSS” , for the purpose of bioremediation near the sea floor at the “oil spill” site on pelagic biota and showed that the concentrations and dispersant-to-oil ratios were lower than the toxicity threshold [18] . It showed that the dispersant treatment at DWH did not likely poison bacteria and slow down degradation as some suspected.
Other studies have focused on overcoming the rate-limiting environmental growth conditions for more efficient bioremediation. As an example, Boopathy, R et al. in 2012 demonstrated that anaerobic bacteria were viable for bioremediation of oil contaminated marsh sediments. Under mixed electron acceptor conditions, there was significant degradation of oil [19] . Warr, LN et al. in 2013 focused on developing the new technique using clay flakes to more effectively apply the fertilizer to the open water spill site for bioremediation, claiming it could significantly reduce the duration of oil spill by slowly releasing the nutrients into water [17] . All of the research projects undertaken have enriched our understanding about the conditions that optimize bacterial petroleum degradations. In the future, we can develop better strategies to cope with marine oil spills.
References
(1) Wikipedia. (1 November 2013)."Deepwater Horizon explosion". Retrieved from: http://en.wikipedia.org/wiki/Deepwater_Horizon_explosion
(2) Ramseur, JL. and Hagerty, CL.. “Deepwater Horizon Oil Spill: Recent Activities and Ongoing Developments.” Congressional Research Service, January 31, 2013. Retrieved from: http://www.fas.org/sgp/crs/misc/R42942.pdf (3) Arnold, C. “Studies seek to find answers about Gulf oil spill legacy” Lancet. 2013 Aug 24; 382(9893):673-4.
(4) Liu, Z. et al. “The weathering of oil after the Deepwater Horizon oil spill: insights from the chemical composition of the oil from the sea surface, salt marshes and sediments” Environ. Res. Lett. 2012 7 035302 doi:10.1088/1748-9326/7/3/035302
(5) Sammarco, PW. et al. “Distribution and concentrations of petroleum hydrocarbons associated with the BP/Deepwater Horizon Oil Spill, Gulf of Mexico” Mar Pollut Bull. 2013 Aug 15;73(1):129-43. doi: 10.1016
(6) Kessler JD. A Persistent Oxygen Anomaly Reveals the Fate of Spilled Methane in the deep Gulf of Mexico.” Science. 2011 Jan 21;331(6015):312-5. doi: 10.1126/science.1199697
(7) Wikipedia. (1 November 2013)."Deepwater Horizon oil spill". Retrieved from: http://en.wikipedia.org/wiki/Deepwater_Horizon_oil_spill
(8) MicrobeWiki. (1 November 2013). "Oil spills". Retrieved from: http://microbewiki.kenyon.edu/index.php/Oil_spills#Nutrients
(9) King SM. et al. “Photolytic and photocatalytic degradation of surface oil from the Deepwater Horizon spill.” Chemosphere. 2013 Oct 16. pii: S0045-6535(13)01300-3. doi: 10.1016
(10) Dubinsky EA. et al. “Succession of Hydrocarbon-Degrading Bacteria in the Aftermath of the Deepwater Horizon Oil Spill in the Gulf of Mexico.” Environ Sci Technol. 2013 Oct 1;47(19):10860-7. doi: 10.1021
(11) Valentine DL. et al. “Dynamic autoinoculation and the microbial ecology of a deep water hydrocarbon irruption.” Proc Natl Acad Sci U S A. 2012 Dec 11;109(50):20286-91. doi: 10.1073
(12) Liu Z. et al. “Evaluating bacterial community structures in oil collected from the sea surface and sediment in the northern Gulf of Mexico after the Deepwater Horizon oil spill.” Microbiologyopen. 2013 Jun;2(3):492-504. doi: 10.1002
(13) Hazen TC. et al. “Deep-sea oil plume enriches indigenous oil-degrading bacteria.” Science. 2010 Oct 8;330(6001):204-8. doi: 10.1126
(14) Atlas RM and Hazen TC. “Oil Biodegradation and Bioremediation A tale of the Two Worst Spills in U.S. History.” Environ Sci Technol. 2011 Aug 15;45(16):6709-15. doi: 10.1021
(15) Vladislav Lavrovsky (17 November 2013). "Directed Evolution of Alkane Degrading Enzymes". Retrieved from: http://www.odec.ca/projects/2004/lavr4v0/public_html/objectives.htm
(16) Peixoto RS. et al. “Petroleum-degrading enzymes: bioremediation and new prospects.” Enzyme Res. 2011;2011:475193. doi: 10.4061/2011/475193
(17) Warr LN. et al. “Bioremediating oil spills in nutrient poor ocean waters using fertilized clay mineral flakes: some experimental constraints.“ Biotechnol Res Int. 2013;2013:704806. doi: 10.1155/2013/704806
(18) Kujawinski EB. Et al. “Fate of Dispersants Associated with the Deepwater Horizon Oil Spill.” Environ Sci Technol. 2011 Feb 15;45(4):1298-306. doi: 10.1021
(19) Boopathy R. et al. “Biogradation of crude oil from the BP oil spill in the marsh sediments of southeast Louisiana, USA” Appl Biochem Biotechnol. 2012 Jul;167(6):1560-8. doi: 10.1007