Listeriosis

Etiology/Bacteriology
Taxonomy
Kingdom: Bacteria
Phylum: Firmicutes
Class: Bacilli
Order: Bacillales
Family: Listeriaceae
Genus: Listeria
Species: monocytogenes
Description
Listeria are Gram positive, facultative anaerobes that are observed to be bacilli in short chains. If a direct sample is observed under the microscope, Listeria can resemble cocci and can be mistaken for streptococci. All species produce flagella at room temperature, but do not express flagella at 37° C. These non-spore forming, catalase positive bacteria are found in soil, sewage, stream water, and animal carriers.
Pathogenesis
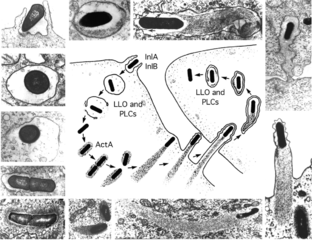
The human pathogen, L. monocytogenes, infects through intracellular mechanisms once ingested, thus bypassing the host’s immune system. D-galactose is expressed on the surface of the pathogen and is recognized by pattern recognizing receptors on the host macrophage. This causes phagocytosis of Listeria which becomes incased in a phagolysosome inside the cell. While inside the vacuole, L. monocytogenes secretes listeriolysin effectively lysing the vacuole membrane. The bacteria can then replicate using host mechanisms, and extract nutrients from the host cell. Since the body has an internal temperature of 37° C, flagella are unable to be produced, so Listeria uses direct polymerization of actin through transmembrane proteins to cause movement of the host cytoskeleton in various directions. Once inside the cell, the bacteria is able to express virulence factors. Proteins clustered around the ribosome binding site denature and allow transcription activator PrfA to bind. Translation then proceeds causing virulence factors to be expressed by the pathogen. These virulence genes are controlled by thermoregulation and are not expressed at temperatures below 37 degrees Celsius. The virulence factors aid in the formation of an actin tail on the pathogen. Actin monomers are collected from the cytoplasm of the cell and combined to form a flagella like tail. Gelsolin is an actin binding protein that is specifically localized in the tail causing and acceleration of motility throughout the cell body. Using actin motility, Listeria pushes against the cell membrane causing filopods. Adjacent cells recognize these filopods and the bacterium is engulfed. The process occurs again in the new cells and infection spreads. Once Listeria is initially phagocytosed by a macrophage it is never again extracellular in the host body.
Transmission
Listeriosis can be transmitted by eating food contaminated with the strain Listeria monocytogenes . Due to the long incubation period of 1 to 4 weeks, many animals that provide meat and dairy can be infected with this disease but not show any symptoms. This is the reason why most humans become infected by eating raw or undercooked meat products. Foods that come packaged ready to eat, such as cold cuts and cheeses, can become adulterated with Listeria after the cooking or pasteurization process, but before shipping out. Listeria is able to grow in such a large range of temperatures even a refrigerator can provide an environment for reproduction. With this risk being high, pregnant women are warned to stay away from these foods because if they contract the disease they can pass it on to their baby. Very rare, but reported cases of Listeria have been acquired through a stay at the hospital. [1]
Infectious Dose, Incubation, and Colonization
Listeria incubation period occurs anywhere from 3-70 days after infection. However symptoms of the disease are not seen until 1-4 weeks after infection. The variability of time for the disease to show symptoms makes it hard to diagnose Listeria soon after exposure.Infections dose is 1,000-1 million microbes. Colonization can occur in the human body at site such as the nervous system, gasterointestinal tract, and epithelial cells in various areas of the body. Biofilms are formed on food products infected with the bacteria and can cause resistance on production lines. Different species of Listeria cause variation in biofilm formation.
Epidemiology
The ingestion of food contaminated with Listeria is the number one cause of infection worldwide, with pregnant women and individuals with a compromised immune system being most susceptible.[5] Due to new found hospital acquired listeriosis, healthy patients with normal immune systems have become infected. The CDC indicates that 1600 people are infected with listeriosis each year in the United States and 260 cases end in death.
During a ten year period from 1998-2008 there were 24 confirmed listeriosis outbreaks in the United States. The literature on these outbreaks indicates that 359 people were infected, 215 were hospitalized, and 38 deaths. Majority of these outbreaks occurred in communities that consumed foods from the same location, but there were also 2 outbreaks that developed in hospitals from the food. All of these outbreaks were attributed to infected, unpasteurized and pasteurized dairy products, produce found in salad fixings, as well as deli meats and hotdogs. [3]
Virulence Factors
1. Adhesion to the surface of mammalian cells. Close interaction between host cells and L. monocytogenes makes invasion possible. Numerous surface adhesion factors allow the pathogen to connect to host cells such as:
- Lap adhesion protein uses an alcohol acetaldehyde dehydrogenase interaction with the host cell receptor, Hsp60, to adhere to intestinal cells. This Lap protein is required for full virulence.
- Ami protein associates to the bacterial surface via its C-terminal cell wall-anchoring (CWA) domain. The CWA allow adhesion to host epithelial cells.
- The gene segment dltA integrates D-alanine-polyphosphoribitol into lipoteichoic acids (LTAs)which contributes to cell adhesion.
- FbpA is expressed on the bacterial surface and binds to stationary host fibronectin. FbpA also helps expression of LLO and InlB (other virulence factor proteins).
- The C-terminus of InlJ has an LPXTG segment which bonds LPXTG-containing proteins to the cell membrane of the host.
2. Invasion of L. monocytogenes also contributes to its virulence. The pathogen can enter into macrophage cells or non-phagocytes.
- InlA and InlA are protiens that assist Listeria entry into non-phagocytes. InlA is a critical player for virulence.
- InlA and E-cadherin interaction allows for epithelial cell invasion because it causes cytoskeletal reorganization.
- Vip protein is present in all pathogenic species and absent in all non-pathogenic strains. Vip mediates Listeria invasion into epithelial cells. The host receptor, Gp96 interacts with Vip and this interaction is important for the entry of eukaryotic cells.
- GtcA mediates glycosylation of host cell wall teichoic acids.
- Listeriolysin O (LLO) helps invasion of host cells because it causes influx of calcium ions which allows for a more effective invasion of Listeria into the cell.
3.Vacuole escape occurs before invasion of non-phagocytic cells and involves the lysis of vacuoles.
- Listeriolysin O (LLO) has critical role in the escape of L. monocytogenes from vacuoles. This secreted toxin is a cholesterol-dependent cytolysin (CDC). LLO works best in acidic conditions, which occurs inside the vacuole. When LLO is inside the cytoplasm (at neutral pH), activity decreases which lessens host cell damage. Enzymes plcA and plcB work with LLO to enhance lysis of the vacuoles.
- PrsA2 stabilizes and catalyzes activity of LLO which allows for optimal activity during vacuole escape.
- SipZ assists in the secretion of LLO
- SvpA also promotes pathogen escape from the phagosomes.
4. Motility and spread allows Listeria to spread once inside the host.
- ActA polymerizes actin filaments that forms a comet-like tail. This tail enables the pathogen to propel and move in the cytosol. The ability to propel itself within the cytosol allows the pathogen to invade neighboring cells (cell-to-cell spreading).
- VASP protein is a protein that helps with motility by interacting with ActA, recruiting an active monomer-binding protein which starts actin polymerization, and reducing actin filament branching (allows for longer filaments to be created).
- InlC protein allows for the formation of protrusions of the cell wall by inhibiting host Tuba and N-WASP protein activity. Tuba and N-WASP help control the structure of epithelial cells[2]
Clinical Features and Diagnosis
Symptoms of infection from Listeria include:
- Fever
- Muscle Aches
- Nausea
- Diarrhea
Secondary symptoms; these are incredibly severe, as they usually indicate that the Listeria has now invaded ones nervous system:
- Headaches
- Stiff Neck
- Delayed Reactions
- Muscle Spasms
If a baby is born with Listeria common symptoms include:
- Loss of appetite
- Cantankerousness
- Vomiting
- Fever
Listeria will only be tested for if there is reasonable evidence to indicate on has the infection. The only tests used to diagnosis the presence of this bacteria is a sample of sterile body fluids, such as blood, urine, or spinal fluids. These bodily fluids are then tested for the presence of a Listeria bacteria.[1]
Treatment
Listeria is most commonly treated through the use of antibiotics. However, if mild or no symptoms are presented, then no intervention is needed. If pregnant women are diagnosed with "Listeriosis" an effective regiment of antibiotics is immediately prescribed to prevent it from spreading to the fetus. [1]The current antibiotic of choice is ampicillin or a cocktail of ampicillin or gentamicin, although due to allergies and each case being individual, various other antibiotics are efficient in killing this disease. [4]
Prevention
The FDA (Food and Drug Administration) has provided the public with guidelines to prevent Listeria contamination.
- Rinse all produce thoroughly under hot water before consuming, preparing, and even cooking. Even if the skin is going to be peeled off, it needs to be washed to prevent the spread of bacteria.
- Scrub all produce that is hard, such as melons and potatoes, with a clean brush under hot running water.
- Dry all rinsed produced with clean towels or paper towels.
- Separate produce from raw bovine, porcine, and avian meats from cooked meats, produce, and pre-packaged, no preparation needed foods.
An important step in keeping Listeria free is properly cleaning one's kitchen and all areas raw meats and produce have been stored.
- So that one does not create a fomite (object that carries disease) all objects that have touched raw meats and produce need to be washed.
- Keep the refrigerator under 4°C and the freezer less than -18CF to prevent Listeria from being able to grow in that environment.
- All leakages from raw meats, spoiled produce, and prepackaged foods should be immediately cleaned up with disinfectants and hot water.
Food Safety is an easy way to prevent infection from Listeria using the following methods.
- Cooking all bovine (63°C or if ground 71°C), porcine (63°C), and avian meats (74°C) to the proper FDA specified temperatures. [6]
- Do not let prepackaged food sit in the refrigerator for long periods of time, it is best to ingest these items as close to the date purchased as possible. Examples include hot dogs which unopened can be eaten within 2 weeks of purchase, but if opened should not sit longer than 1 week. Cold cuts should only be eaten within 2 weeks if it is left unopened, but packages that have been opened or cut at the deli need to be eaten no later than 3 to 5 days after purchase.
- If left overs are to be store in the refrigerator need to be placed in thin bowls that allow for the food to be cooled evenly. If not eaten within 3 to 4 days, these foods need to be thrown away.
- Stay away from unpasteurized dairy products. [1]
Host Immune Response
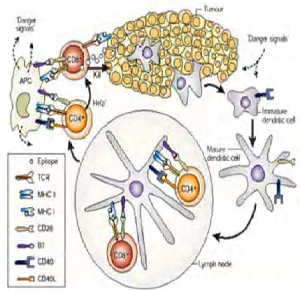
L. monocytogenes has been a model for study for the immune system since the 1960s. The response that Listeria causes in its infected host can be broken down into innate and adaptive responses.
Innate Respone
Listeria monocytogenes enters the body through infected food products that are able to survive the stomach acid and being to colonize the large intestine. Once the bacteria begin to invade the host, macrophages recruit neutrophils to aid in the phagocytosis of these pathogens. The bactericidal mechanisms of macrophages with this particular disease is of large interest due to Listeria replicating within these immunological cells. The macrophages attempt to clear out the bacteria, along with neutrophils, and natural killer cells that were signaled in by cytokines released by the macrophages. These same cytokines that recruit natural killer cells to the site of infection also trigger the host inflammatory response.
Adaptive Response
Dendritic cells link the innate and adaptive immune responses. DCs (dendritic cells) help activate the T cell’s response. CD4 and CD8 T cells play a major role for the adaptive immune response. L. monocytogenes antigens is presented to different T cells depending on the type of cell it infects. For example, if Listeria infects the cytosol of any cell, it will secrete proteins and these proteins are bound to MHCI molecules and presented to CD8 T cells. When Listeria is destroyed in lysosomes, antigen-presenting cells use antigen from this destruction and present it on MHC2 molecules to CD4 T cells. Memory T cells are used to protect the host from re-infection of Listeria.
L. monocytogenes antigens are commonly presented to T cell receptors by MHC Ib. The amount of bacteria specific cells peaks 5-6 days after infection. If a second exposure to L. monocytogenes occurs the immune response reacts similarly but increases its proliferation of MHCIa receptors and limits the production of MHCIb. However the adaptive response cannot perform adequately without MHCIb. Once Listeria has become intracellular, it becomes hidden from the immune response. The adaptive immune response can then fight back using a monoclonal antibody which targets LLO virulence factors. This pathway allows for the immune system to target the bacteria inside the cell as if it were a virus. The body also aims to trap the bacteria and anitgens in the peripheral lymphoid organs with B cells and antibodies so T cells can recognize the antigen as they are being developed. [7]
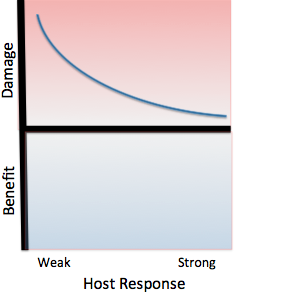
Damage Response Framework
Listeria monocytogenes can be classified as a Class 2 pathogen, based on the Damage Response Framework classification system, because it causes a response in host cells in the setting of normal host immune responses, and more serious damage in host cells with a weak immune response. A normal host immune system will respond to L. monocytogenes by stimulating phagocytosis of the pathogen into a phagosome, which the pathogen then dissolves.
Normal host immune systems have also begun to mutate and develop the ability to recognize the presence of L. monocytogenes and signal Natural Killer cells to destroy the infected host cells before the pathogen can spread. In this way, a strong host response would limit the damaging effects of L. monocytogenes.
In hosts with a weak immune response, L. monocytogenes would cause more serious damage, because the host’s immune system would be less capable of recognizing and responding to the presence of L. monocytogenes.
References
1. http://www.cdc.gov/listeria/
2. D Cabanes, et al. The Arsenal Of Virulence Factors Deployed By Listeria Monocytogenes To Promote Its Cell Infection Cycle. Virulence 2.5 (n.d.): 379-394. Science Citation Index. Web. 14 July 2013.
3. Emily J., Kelly A. Jackson, Shacara D. Johnson, Lewis M. Graves, Benjamin J. Silk, and Barbara E. Mahon. "Listeriosis Outbreaks and Associated Food Vehicles, United States, 1998-2008: Outbreaks and Associated Food Vehicles Medscape
4. Pharmacother, Ann. "Treatment of Listeriosis." National Center for Biotechnology Information, U.S. National Library of Medicine
5. Listeria The Chinese University of Hong Kong
6. "Safe Eats - Meat, Poultry, & Seafood." U.S. Food and Drug Administration
7. L.A. Zenewicz & H. Shen. Innate and adaptive immune responses to Listeria monocytogenes: A short overview
8. Casadevall, A., & Pirofski, L. (n.d.). The damage-response framework of microbial pathogenesis. Nat Rev Micro Nature Reviews Microbiology 2003. 17-24.
Created by Jennifer Gallup, Naomi Quillin, and Danielle Vinnedge, students of Tyrrell Conway at the University of Oklahoma.
Edited by Danielle Lewis, Erika Cummings, and Elizabeth Fish, students of Tyrrell Conway at the University of Oklahoma
![thumb] [4]](/images/thumb/f/fd/Mail.jpg/150px-Mail.jpg)