Pharmacokinetics of botulinum neurotoxin serotypes: Implications for Infant Botulism
By Sarah R. Hall
Introduction
Clostridium botulinum is a rod-shaped, anaerobic, spore forming, gram-positive bacilli and is responsible for botulism (Slonczewski et al. 2009). In contrast to the genus Bacillus, the genus Clostridium does not show a highly similar DNA sequence, and the Clostridium species vary greatly throughout the Firmicutes phylogeny. The spores of Clostridium cells develop differently than that of Bacillus. The endospore swells at one end of the cell, which visually takes the shape of a “drumstick” (Slonczewski et al. 2009). The common habitat of Clostridium spores is the soil and water and will begin to germinate in anaerobic and substrate-rich environments (Slonczewski et al. 2009).
C. botulinum is able to produce seven different botulinum neurotoxins (BoNT/A to G) (Rossetto et al. 2006) that are responsible for four different types of human disease: wound botulism, food-borne botulism, adult intestinal colonization, and infant botulism (Fox et al. 2005). Only BoNT/A, B, E, and F cause disease in humans (Fox et al. 2005). There is an average of 110 cases of botulism reported in the U.S. per year, and infant botulism, first reported in 1976, is the most commonly reported botulism form contributing to about 70% of the reported cases. BoNT/A and B account for more than 90% of the reported cases of infant botulism to the Centers for Disease Control and Prevention (Keet et al. 2005). Infant boys and girls are affected equally with a mean age of onset of 13 weeks, with a range from 1-63 weeks (“Botulism: Epidemiological Overview for Clinicians”). The risk factors for infant botulism are living in a rural environment, having a parent who works in the soil industry, and the consumption of honey (Fox et al. 2005). In fact, 59% of reported cases in Europe are associated to honey. While only 20% of U.S. soil samples contain botulinum spores, most reports of infant botulism originate in California, Pennsylvania, and Utah (Fox et al. 2005).
Clostridial organisms infect, grow, and germinate within the gastrointestinal tract in infant botulism and adult intestinal colonization types and in the wound for the wound botulism type, and then begin to produce the toxins that are absorbed by the body (Fox et al. 2005). In food-borne botulism, on the other hand, the toxin is already formed, and ingestion of this preformed toxin is how the disease occurs.
The reason for the high incidence of infant botulism compared to the other three types of the human disease is that infants are much more susceptible to the colonization and growth of clostridia within the gut. Throughout the age window of 1-63 weeks, most infants are weaned from breast milk and introduced to much different foods. This change in nutrient type is paralleled with changes in the gut flora in the infant, and this altered gut flora presents an opportunistic environment for the clostridial spores to colonize the intestine (Fox et al. 2005). Animal studies support this view. It has been found that adult mice that were originally resistant to clostridial colonization became susceptible after administered antibiotics (a mixture of erythromycin and kanamycin sulfate), which changed the gut flora of the mice (Burr et al. 1982).
Human botulism immune globulin (Baby-BIG) was approved in October of 2003 by the Food and Drug Administration to be used to treat infant botulism (Fox et al. 2005). Prior to Baby-BIG, treatment largely consisted of nutritional care and respiratory ventilation. Baby-BIG is composed of pooled plasma of adults immunized with pentavalent botulinum toxoid with neutralizing antibodies against BoNT/A and B. This treatment has achieved good results, with a reduced length of mechanical ventilation (0.7 vs. 2.4 weeks), a reduced duration of tube feeding (3.6 weeks vs. 10 weeks), and significantly reduced stay in the hospital (2.6 weeks vs. 5.7 weeks) (Fox et al. 2005).
The clostridial spore germinates within the colon, and the BoNTs produced from C. botulinum travel from the intestinal lumen to the circulatory system, and finally to their specific targets, the peripheral cholinergic nerve terminals (Schechter 1999). In summary, BoNTs cause persistent, but reversible, inhibition of acetylcholine release and muscle paralysis. Inhibition of acetylcholine release occurs at the neuromuscular junction and autonomic nervous systems. Constipation is usually one of the first signs of a botulism infection in an infant (Fox et al. 2005). This is followed by a descending, symmetric motor weakness and paralysis. The autonomic nervous system functioning declines over the time period of hours to several days, affecting first the cranial nerves, then the trunk, extremities, and finally the diaphragm (Fox et al. 2005).
The diagnosis of infant botulism is usually electrophysiology, since it is the quickest method (Fox et al. 2005). Infant botulism is suspected when the amplitude of the M-wave is small, and when there is an incremental response to rapid repetitive stimulation. A longer test is laboratory identification of botulinum toxins or cells in the serum or stool(Fox et al. 2005). However, since constipation is usually a common side-effect, samples of the stool are more difficult to obtain. Further, serum testing has its own draw-back in that it is a less sensitive test in comparison to stool testing.
BoNTs are highly specific due to two reasons: First, the toxin is lethal at pico- to femtomolar concentrations (Rossetto et al. 2006). Particularly, in the very small body weight of infants, minuscule amounts of toxin (nanogram/kilogram body weight) can cause devastating consequences (Schechter 1999). Second, the ratio of the cholinergic presynaptic terminals to the entire exposed cell surface within the body is a very tiny ratio, making it evident that BoNTs are extremely specific (Rossetto et al. 2006). All seven serotypes of BoNTs are composed of two polypeptide chains: a light (L) chain of 50 kDa linked by one disulfide bond (Fox et al. 2005) to a heavy (H) chain of 100 kDa (Rossetto et al. 2006). The L-chain disulfide bond is linked to the first half of the H-chain (HN or N-terminal domain of the H-chain). HN is responsible for entry into the cell, while the HC domain (C-terminal domain of the H-chain) plays a role in the specific interaction of the neural binding (Rossetto et al. 2006).
The HC domain can be separated into the N-terminal half, which folds like the carbohydrate binding proteins of the legume lectin family, and the C-terminal half, which folds like the trypsin inhibitor family proteins (Rossetto et al. 2006). This bipartite structure of the HC has been presumed to account for the high affinity, specificity, and rapidity of neuronal binding. The HN domain is composed of two antiparallel α–helices, which are highly homologous among the BoNTs. This common structure in the HN domain among the BoNTs is translated into a common function, where the L-chain in all BoNTs undergoes transmembrane translocation at a low pH. The catalytic L-chain functions as a zinc-dependent endopeptidase (Fox et al. 2005) with a cleft shaped active site where a catalytic zinc atom is coordinated with one glutamate and two histidine residues (Rossetto et al. 2006).
Within the neuron, three proteins: VAMP (vesicle-associated membrane protein), SNAP-25, and syntaxin, all assemble to form a trimeric complex that is usually abbreviated as the SNARE complex (Rossetto et al. 2006). It is the collection of SNARE complexes in a rosette shape that facilitates the movement of the synaptic vesicle to the cytosolic side of the presynaptic membrane. Therefore, SNARE approximates the synaptic vesicle filled with neurotransmitter close to the neuron terminal membrane so that the vesicle membrane can fuse and discharge the neurotransmitter (in this case acetylcholine) into the synaptic cleft. Only the proteolysis of a single SNARE protein prevents the formation of a functional SNARE complex. BoNT/B, D, F, and G cleave only VAMP (VAMP and Sbr or synaptic vesicle protein are used interchangeably), where most of its cytosolic domain is lost. BoNT/A and E cleave SNAP-25 only within its C-terminus. BoNT/C cleaves SNAP-25 and syntaxin (Rossetto et al. 2006).
Quantification of the half-lives of toxin inhibition and the speed of SNARE reformation in BoNTs/A, B, C, E, and F
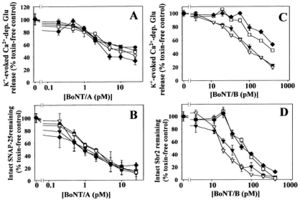
BoNTs do not cause neuronal death in vivo or in vitro, unless excessively high concentrations are applied (Rossetto et al. 2006). It was previously thought that BoNT binding was irreversible. Recovery was possible through the development of new motor end plates and regeneration of terminal motor neurons through new axon sprouts (Fox et al. 2005). More recent evidence suggests that neuronal function recovers with time by the inactivation of the BoNTs (Rossetto et al. 2006). The SNARE peptides that are the products of BoNT hydrolysis are removed and replenished by de novo synthesis occurring within the motor end plate (Rossetto et al. 2006). Even though all botulinum serotypes result in a nonfunctional SNARE complex and subsequent prevention of the release of neurotransmitter, the duration of neuromuscular paralysis is very different among the BoNTs. Recent evidence has suggested that BoNTs/A, B, and C have a longer inhibitory effect on nerve terminals than BoNTs/E and F (Foran et al. 2003). Almost all of the infant botulism cases are caused by BoNTs/A and B. Type A is reported to occur at a higher frequency in the western half of the United States, while most cases of BoNT/B occur in the eastern half the of the United States (Fox et al. 2005). Foran et al. (2003) used rat cerebellar neurons in order to quantify the half-life of each BoNT’s effect and the rate at which the SNARE substrates were made functional again. The cerebellar neurons were bathed in a culture medium with certain concentrations of BoNT/A (Figure 1A; 0, 0.1, 10 pM) or BoNT/B (Figure 1C; 0, 10, 100, 1000 pM) for 24 hours. In order to measure neurotransmitter release, the amounts of Ca2+-dependent [14C]glutamate released into basal and K+-stimulated samples was calculated and expressed as a percentage of the total cell content (Figures 1A and C). Further, in order to quantify the relative speed at which the SNARE substrates were made functional again in response to increasing concentrations of BoNT/A (Figure 1B) and BoNT/B (Figure 1D), equal amounts of neuronal protein were immunoblotted using SMI-81 Ig (SNAP-25) and anti-HV62 (for Sbr2) in order to measure the degree of SNARE proteolysis.
Interestingly, it was found that BoNT/A showed a concentration-dependent inhibition of neurotransmitter release to about a maximum of 65% blockade (Figure 1A) and corresponded to the cleavage of approximately 90% of the SNAP-25 (Figure 1B). BoNT/B, on the other hand, induced a dose-dependent inhibition of neurotransmitter release of about 80% blockade (Figure 1C) and was associated with proteolysis of about 100% of Sbr2 (Figure 1D). It is of further mention to note that only 10 pM of BoNT/A resulted in a 50% blockade of neurotransmitter release, whereas it took 100 pM of BoNT/B to induce a 50% blockade of neurotransmitter release. Even though BoNT/B showed a much greater inhibition of neurotransmitter release and more extensive proteolysis of the substrate, the removal of BoNT/A and BoNT/B by washing, suggested that BoNT/B shows a shorter duration of inhibition than BoNT/A. The disinhibition of neurotransmitter release and replenishment of the Sbr2 substrate for BoNT/B occurred in a time-dependent fashion for the neurons at 2, 7, 25, and 28 days. The time-dependent recovery of release and SNAP-25 replacement for BoNT/A was not seen (Figures A and B).
Follow-up experiments confirmed a lack of any significant recovery of neurotransmitter release a month after BoNT/A incubation, whereas BoNT/B had a much faster recovery period. Exponential decay calculations obtained inhibitory half-lives (t1/2 INH) for BoNT/A exceeding 31 days, while the mean t1/2 INH for BoNT/B was approximately 9.84 days (Table 1). The same concentration dependency studies were used to calculate the t1/2 INH for BoNT/C, E, F, and tetanus toxin (TeTx) as shown in Table 1. Therefore, BoNT/A is the longest-lasting, while BoNT/E and F are very short lasting and BoNT/B can be considered intermediate in duration.
Production of BoNT/F in Clostridium baratii and BoNT/E in Clostridium butyricum
The clinical course of infant botulism follows a slowly progressive course over a number of days, culminating in a long recovery period. The recovery of neuromuscular function has been reported to occur more rapidly than autonomic function (Fox et al. 2005). Infants that do not receive Human Botulinum Immunoglobulin and who need mechanical ventilation typically are dependent on the ventilator of a mean of 23 days and a mean hospital stay of 44 days (Keet et al. 2005). The symptoms and clinical spectrum can vary depending on the severity of the infection. Interestingly, recent evidence suggests that the type of clostridia strain colonizing the patient, in addition to the BoNT serotype produced (as mentioned above), has a large impact on the clinical syndromes (Fox et al. 2005).
One recent case of infant botulism is an example of the different clinical picture presented when a different clostridia strain colonized the infant. The female patient was only 54 hours old when she was diagnosed with infant botulism (Keet et al. 2005). It was commonly thought that the incubation period for infant botulism was at least 3 days (Keet et al. 2005). Thus, it is difficult to conceptualize in this case study how ingestion of the spore, sporulation, toxin formation, and toxin absorption could all occur in this short time period. The symptoms of the female infant, too, were quite different. She exhibited no preceding constipation. Instead of the progressive course of symmetrical, descending motor weakness, as typically perceived in infant botulism cases, this female patient exhibited “complete flaccid paralysis within 12 hours.” Like her quick onset, her recovery was very rapid, and she regained most neuromuscular function by 3 days after symptom onset, and respiratory endeavors were normal by 5 days (Keet et al. 2005). On the day of her discharge when she was 22 days old, the mouse neutralization bioassay test was reported and was positive for type F botulinum toxin. The agent that caused the disease was reported to be Clostridium baratii, and was hypothesized to have infected the patient immediately following her birth and upon exposure to Clostridium baratii spores in dust (Keet et al. 2005). Ingestion of the preformed toxin is not likely, since the infant was only fed breast milk, and stool samples of the infant’s mother and father did not test positive for BoNT or Clostridia. Further, in utero infections have never been reported.
Only two other cases of type F producing Clostridium baratii have ever been reported, and the cases were unique in the very early age of onset (9 and 14 days old are the only other two cases to have been reported), the rapid onset, and the rapid recovery (Keet et al. 2005). This is consistent with the half-life of BoNT/F inhibition of only about 1.76 days (Table 1).
Only two cases of infant botulism that was caused by Clostridium butyricum have ever been reported. In both of these cases, the infants were 16-week-old females in Italy who were thought to ingest the preformed toxin through honey (Fox et al. 2005). Their stool samples tested positive for BoNT/E, and both infants need mechanical ventilation. Full respiratory recovery was achieved in 8-12 days (Fox et al. 2005). As with Clostridium baratii, the rapid onset and recovery of Clostridium butyricum are typical of the half-lives of neurotransmitter blockade associated with BoNTs/E and F. Clostridium botulinum, Clostridium baratii, and Clostridium butyricum are all genetically and phenotypically distinct clostridial species, but all are now considered causative organisms of infant botulism (Fox et al. 2005).
Dual Toxin-Producing Strains of Clostridium botulinum
Most Clostridium botulinum strains produce a single toxin, as were described above. However, more recently, rare and unusual strains of C. botulinum have been identified that synthesize two types of the toxins (Barash et al. 2004), such as Ba, Ab, Bf, and Af (the capitalized letter in each pair denotes the principal toxin type), all of which have been identified. Researchers and clinicians usually determine the specific serotype of BoNT by toxin neutralization with the homologous “type-specific monovalent antitoxin” (Barash et al. 2004).
A total of three Bf dual toxin-producing C. botulinum was reported: Strain 3281 was isolated from an infant in New Mexico in 1980, Strain 4013 was isolated from an infant from California in 1981, and a third strain that was isolated from an infant in England in 1989 (Barash et al. 2004). Probably the most interesting characteristic of this strain is insight that the BoNT serotype produced by the bacteria depends on the temperature at which the organism germinates and grows. Barash et al. (2004) performed a mouse neutralization bioassay to determine the dual BoNT produced by a unique strain 93-197 in California and the effect of temperature on the predominant toxin synthesis.
Mice were injected with a 1:20 dilution of untreated toxic culture filtrate of strain 93-197, all of these mice died (Table 2, Unheated filtrate). Neutralization of the toxic culture filtrate did not occur when Type B monovalent antitoxin was used by itself (Table 2, Unheated filtrate + antitoxin B); Type F monovalent antitoxin also failed to neutralize the toxic culture filtrate (Table 2, Unheated filtrate + antitoxin F), since all of these mice died. However, all mice that were pretreated with Type B and F monovalent antitoxins combination and then injected with the culture filtrate survived (Table 2, Unheated filtrate + B-F antitoxin mixture). The finding that the B-F antitoxin mixture was needed in order for neutralization suggests that the strain 93-197 produces two distinct serological BoNTs, types B and F. Further, single-colony isolates from the initial stool sample from 1993 (Isolate no. 2-6) replicated the neutralization observed with the index isolate (Isolate no. 1), leaving out the potential that these findings were from a culture that contained both C. botulinum type B and C. botulinum type F (Barash et al. 2004).
The effect of temperature on the type of toxin produced by the 93-197 strain was characterized by incubating the Bf strain at three different temperatures (30°C, 35°C, and 37°C) and testing for the predominant toxin produced. Interestingly, it was found that BoNT/B was produced primarily at 37°C, while BoNT/F was exclusively produced at 30°C, while both BoNT/B and F were produced at 35°C (Table 3, Botulinum toxin type detected). Going along with this logic, the mice that were pretreated with antitoxin B prior to injection with the three culture filtrates incubated at different temperatures would be expected to survive the culture filtrate incubated at 37°C, since BoNT/B was predominantly produced. This was exactly what was found (Table 3, Filtrate + antitoxin B), and the only mice that survived from those pretreated with antitoxin F, were those injected with the filtrate at 30°C (Table 3, Filtrate + antitoxin F). The intermediate 35°C temperature produced both BoNTs/B and F at concentrations that were lethal to mice.
Conclusion
While the disease of botulism is considered rare, the pathophysiology of BoNTs on the inhibition of acetylcholine release via proteolysis of the SNARE complex is very complex. The complexities carry over into the very broad clinical spectrum of infant botulism. Recent evidence now suggests that the clinical picture is even wider then previously thought. The differences in age of onset, length of onset and recovery, and symptoms are now suggested to be due to differences in the serotype of BoNT produced by C. botulinum, the strain of organism producing the different BoNT serotype (C. baratii, C. butyricum), and the temperature-dependent alternating BoNT serotypes produced in a single organism.
References
[http://jcm.asm.org/cgi/content/full/42/4/1713?view=long&pmid=15071029 Barash, J.and Arnon, S. “Dual toxin-producing strain of Clostridium botulinum type Bf isolated from a California patient with infant botulism.” Journal of Clinical Microbiology. 2004. Volume 42. p. 1713-1715.]
[http://www.jbc.org/cgi/content/full/278/2/1363 Foran, P. Mohammed, N., Lisk, G., Nagwaney, S., Lawrence, G., et al. “Evaluation of the Therapeutic Usefulness of Botulinum Neurotoxin N, C1, E, and F Compared with the Long Lasting Type A.” Journal of Biological Chemistry. 2003. Volume 278. 1363-1371.]
[http://www.jmedcbr.org/issue_0501/Reiss/Reiss_01_07.html Reiss, J., and Reiss, K. “Recent Advance in the Detection and Identification of Botulinum Neurotoxins (BoNTs).” The Journal of Medicinal Chemical, Biological, and Radiological Defense. 2007. Volume 5.]
Schechter, R. “Infant Botulism: A Brief Overview.” Anaerobe. 1999. Volume 5. 161-164.
Slonczewski, J. and Foster, J. Microbiology: An Evolving Science. New York: W.W. Norton & Company, 2009.
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2009, Kenyon College.
