Saccharopolyspora spinosa
Classification
Domain: Bacteria
Phylum: Actinobacteria
Class: Actinobacteria
Order: Actinomycetales
Family: Pseudonocardiaceae
Genus: Saccharopolyspora
Species
Saccharopolyspora spinosa
|
NCBI: [1] |
Related species:
Saccharopolyspora hirsuta
Saccharopolyspora erythraea
Description and Significance
Discovery and Description
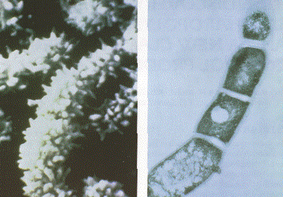
In 1982, researchers vacationing in the Virgin Islands discovered the actinomycete bacteria, Saccharopolyspora spinosa, in soil collected at an inoperative sugar mill rum still (Mertz and Yao 1990, Thompson et al. 2000). Similar to other species in the Saccharopolyspora genus, S. spinosa are aerobic, gram-positive, and have mycelium (Mertz and Yao 1990). The mycelium are made up of pale yellowish-pink aerial hyphae, and yellow to yellow-brown substrate hyphae (Mertz and Yao 1990). These hyphae are long bead-like spore chains (with 50+ spores) that are surrounded by spiny (='spinosa') spore sheaths (Mertz and Yao 1990). Spores are oblong and approximately 1.1μm by 1.5μm (Mertz and Yao 1990). Aerial hyphae have a hook, loop and incomplete spiral arrangement that are described as 'rectus-flexibilius' (Mertz and Yao 1990). These bacteria are most similar to S. hirsuta and S. erythraea in morphology, but have different physiological characteristics compared to other species in this genus (Mertz and Yao 1990). Furthermore, since the discovery of S. spinosa, many research cultures have developed different strains of S. spinosa that have distinct characteristics. Strains of S. spinosa are convoluted in scientific literature, but a few including the type strain: A83543.1 are listed on StrainInfo.net.
Significance
Saccharopolyspora spinosa produce metabolites named 'spinosyns' that are economically, ecologically, and agriculturally important. It has been found that spinosyns have pesticidal properties that are effective for many mites and insects (Salgado 1998, Bond et al. 2004, Cloyd 2009, Thompson et al. 2000, Watson 2001, Hertlein et al. 2011). Interestingly, the discovery of these properties led to a patent (US5631155) on S. spinosa and the byproducts produced by the organisms (Turner et al. 1995). Spinosyns are generated by S. spinosa in an aerobic fermentation broth consisting of necessary metabolic nutrients and the S. spinosa organisms. The spinosyns extracted from the broth make up a class of insecticides that are sold under trade names such as Conserve® and Entrust®. Because the active ingredients in these pesticides are biological byproducts, the pesticides are marketed as "natural," and less toxic compared to other available chemicals. Furthermore, the spinosyns have two modes of action that help prevent development of resistances. Therefore, pesticides containing spinosyns are attractive options for pest management in agriculture.
Genome Structure
The genome of Saccharopolyspora spinosa has been drafted by the Beijing Genomics Institute. It was produced using a whole-genome shotgun strategy with an Illumina genome analyzer as well as Roche 454 pyrosequencing. The entire genome was found to be at 8,581,920 base pairs with 8,302 predicted coding sequences. G+C content was found to be 67.94%. The Illumina pair-end reads produced an assembly of 22 contigs. An estimated 82.8% of nucleotides are predicted to encode proteins (Pan et al. 2011). This organism is still under study and its genome is in the process of being fully mapped.
A 108-kb genomic DNA region of S. spinosa NRRL 18395, producer of spinosyns, has been cloned, sequenced and analyzed to reveal clustered genes encoding a type I polyketide synthase (PKS) complex. PKS genes in the organism are flanked by those that encode homologs of enzymes involved in the urea cycle, valine, leucine and isoleucine biosynthesis and energy metabolism (Zirke et al. 2004).
Cell Structure, Metabolism and Life Cycle
Cell Structure
The cell wall of Saccharopolyspora spinosa is mainly composed of a thick layer of peptidoglycan, and lipoteichoic acids, that serve as chelating agents and also for adherence. The individual peptidoglycan molecules are cross-linked by pentaglycine chains using a DD-transpeptidase enzyme. Spores of S. spinosa are round to oval or oblong and covered by a sheath. The cell wall of this organism, just as with other members in its genus is characterized by the absence of mycolic acids (long-chain α-substituted Β-hydroxylated fatty acids) (Alexander et al. 2000).
Metabolism
Saccharopolyspora spinosa is a chemoorganoheterotroph that decomposes organic matter to produce energy. It may utilize a list of organic compounds for carbon and energy. Some of these include: acetate, butyrate, citrate, formate, lactate, malate, proprionate, pyruvate, and succinate (Mertz and Yao 1990). This species also reduces nitrogen. During metabolism S. spinosa generates metabolites two of which, spinosyn a and d, are the most notable. These metabolites are mainly generated in a production setting for the purposes of extracting them for the production of active ingredients in pesticides.
Life Cycle
Since the discovery of S. spinosa, there have been no reports in the scientific literature of the organism being re-recovered in nature. However, it is likely that the life cycle of S. spinosa in nature is similar to other soil actinomycetes. The aerial hyphae of S. spinosa likely serve as a means of spore dispersal. In actinomycetes, spores germinate and a mycelium network grows. Aerial and substrate hyphae containing spore chains grow from the mycelium. Genetic information among spores is acquired through conjugation (Whalen and Sampedro 2010). Fragmentation of S. spinosa hyphae in liquid (Mertz and Yao 1990) may also serve as a means of spore dispersal.
Biology and Significance
Habitat
Saccharopolyspora spinosa is kept in culture. Culture medias that have demonstrated good growth conditions for S. spinosa include actinomycete isolation agar, Bennett agar, tomato paste-oatmeal agar (Waksman 1961 via Mertz and Yao 1990, Mertz and Yao 1990), and BHI (Brain Heart Infusion) agar (Waldon et al. 2001). The optimal temperature for growth in media are 15-37°C (Mertz and Yao 1990). These organisms are also grown in fermentation broths (Waldon et al. 2001).
Ecology
The interrelationships between S. spinosa and other organisms has not been thoroughly investigated. However in a study using potato cyst nematode, Globodera rostochiensis, bioagents obtained from S. spinosa caused significant inhibitory effect on multiplication of the nematode, suggesting a possible antagonistic relationship with this organism in nature (Trifonova 2010). It is also possible that S. spinosa would generate available nitrogen (via nitrogen reduction) for other organisms in natural settings.
Environmental and Applied Significance
The discovery of S. spinosa has led to a novel family of environmentally friendly compounds with many applications in integrated pest management programs. The metabolites produced by S. spinosa make up the low-risk insecticidal chemical called 'spinosad'. This natural pesticide consists of a mixture of tetracyclic macrolide neurotoxins, spinosyn A and D that kill many insect pests and have no known toxic effect on humans (Bond et al. 2004, Pan et al. 2011). Studies have demonstrated that spinosad is not carcinogenic, mutagenic or terotogenic to mammmals (Thompson et al. 2000, Kirst 2010). Furthermore, spinosyns also have low toxicity and reduced risk toward many aquatic and avian fauna (Kirst 2010). Spinosad is an attractive pest management tool because it degrades quickly via photolysis (West 1997, Thompson et al. 2002, Kirst 2010).
Low toxicity to non-target organisms and rapid degradation are characteristics that pose many opportunities for pest management in a variety of settings. For example, spinosad has been demonstrated to be a good option for mosquito management in domestic water supplies (Bond et al. 2004). It has also been marketed as a treatment for headlice under the pharmaceutical name Natroba. In general, spinosad is effective for wide range of mites and insects (Thompson et al. 2000). Some of these are major crop pests such as European Corn Borer, Colorado Potato Beetle, Western Flower Thrips, and Grape Berry Moth (Thompson et al. 2000).
Spinosads can persist for much longer periods in the absence of light. In dry grain storage bins this compound remains viable for six months and up to two years (Hertlein et al. 2011). Therefore, the chemical is used in several countries to protect stored grain from pests. The effectiveness of this compound for these purposes is still being explored. It is reported that several factors such as insect species, pest life stage, grain type, and grain variety influence the strength of spinosad (Hertlein et al. 2011).
Mode of Action of Spinosyns
Contact and ingestion of spinosad are two means of exposure to the pesticide (Thompson et al. 2000). Spinosyns have a unique mode of action involving the postsynaptic nicotinic acetylcholine and GABA receptors (Bond et al. 2004, Watson 2001). Spinosyn A, the major active component of spinosad, causes involuntary muscle contractions and tremors by widespread excitation of neurons in the central nervous system of insects at low concentrations. Prolonged spinosyn-induced hyperexcitation results in paralysis associated with neuromuscular fatigue and death (Salgado 1998).
References
Alexander, M., Bloom R.B., Hopwood, D.A., Hull, R., and B.H., Iglewski (Ed). Encyclopedia of Microbiology. Second Edition. 2000. Elsevier Science.
Bond J.G., Marina, C.F., and Williams, T. "The naturally Derived Insecticide Spinosad is highly Toxic to Aedes and Anopheles Mosquito Larvae. Medical and Veterinary Entomology. 2004. Volume 18. p 50-56
Cloyd, R. "Western Flower Thrips Managment: Have We Reached An Impasse?". OFA Bulletin. 2009. Volume 918. p. 24-29.
Hertlein M.B., Thompson, G.D., Subramanyamb, B., and Athanassiou, C.G. "Spinosad: A New Natural Product for Stored Grain Protection". Journal of Stored Products Research. 2011. Volume 47. p. 131-146.
Kirst, H.A. "The spinosyn family of insecticides: realizing the potential of natural products research". The Journal of Antibiotics. 2010. Volume 63. p 101–111.
Mertz, F.P., and Yao, R.C. "Saccharopolyspora spinosa sp. nov. Isolated from Soil Collected in a Sugar Mill Rum Still". International Journal of Systematic Bacteriology. 1990. Volume 40(1). p. 34-39.
Pan, Y., Yang, X., Li, J., Zhang, R., Hu, Y., Zhou., Y., Wang, and B Zhu . "Genome Sequencing of the Spinosyns-Producing Bacterium Saccharopolyspora Spinosa NRRL 18935". Journal of Bacteriology. 2011. Volume 193(12). p. 3150-3151.
Salgado, V.L. "Studies on the Mode of Action of Spinosad: Insect Symptoms and Physiological Correlates". Pesticide Biochemistry and Physiology. 1998. Volume 60. p 91–102.
Thompson, G.D., Dutton, R., and Sparks, T.C. "Spinosad - a case study: an example froma natural products discovery programme". Pest Management Science. 2000. Volume 56. p. 696-702.
Thompson, D. G., Harris, B. J., Lanteigne, L. J., Buscarini, T. M. & Chartrand, D. T. "Fate of spinosad in litter and soils of a mixed conifer stand in the Acadian forest region of New Brunswick". J. Agric. Food Chem. 2002. Volume 50 790–795.
Trifonova, Z.T. "Studies on the Efficacy of some Bacteria and Fungi for Control of Globodera restochiensis". Journal of Agricultural Sciences. 2010. Volume 55 (1). p. 37-44.
Turner, J.R., Huber, M.L.B., Broughton, M.C., Mynderse, J.S., Martin, J.W. United States Patent: US5631155 for Saccharopolyspora spinosa strain. 1995.
Waldon, C., Matsushima, P.,Rosteck, P.R.J., Broughtonb, M.C., Turnerb, J., Madduria, K., Crawforda, K.P., Merloa, D.J., and Baltz, R.H. "Cloning and analysis of the spinosad biosynthetic gene cluster of Saccharopolyspora spinosa". Chemistry & Biology. 2001. Volume 8(5). p. 487–499.
Watson, G.B. "Actions of Insecticidal Spinosyns on γ-aminobutyric Acid Receptors from Small Diameter Cockroach Neurones". Pesticide Biochemistry and Physiology. 2001. Volume 71. p. 20–28.
West, S. D. "Determination of the naturally derived insect control agent spinosad and its metabolites in soil, sediment, and water by HPLC with UV detection". J. Agric. Food Chem. 1997. Volume 49. p. 3107–3113.
Whalen, J.K., and Sampedro, L. "Soil Microorganisms" in Soil Ecology and Management. 2010. Cambridge University Press, Cambridge.
Zirke, R., Black T.A., Gorlach, J., Ligon, J,M. and I. Molnar."Analysis of a 108-kb Region of the Saccharopolyspora spinosa Genome Covering the Obscurin Polyketide Synthase Locus". DNA Seq. 2004. Volume 15, No. 2 p.123–134.
Author
Page authored by Emily Pochubay, Matt O'Grady, Placid Mike Mpeketula, and Demetrious Parker students of Prof. Jay Lennon at Michigan State University.
<-- Do not remove this line-->