The Mycorrhizal Network: Interactions, Communication & Transport
Introduction

By Amanda Carroll
Mycorrhizal networks are one of nature's most fascinating and lesser-known symbionts. These fungi help to facilitate communication between plants via mycorrhizal networks underground. Specific kinds of mycorrhizal include ectomycorrhiza and arbuscular mycorrhiza. These fungi help to enhance growth, nutrient uptake, and the physiological function of plants and plant communities. The purpose of this page is to discuss the function of the mycorrhizal networks, the specific types of mycorrhizal fungi, mycorrhiza helper bacteria, and specific examples of these interactions at work. The goal is to present a holistic view of these incredible underground networks.
[1] [2] [3] [4] [5] [6] [7] [8] [9] [10]
Mycorrhizal Networks
Mycorrhizal networks exist in all terrestrial ecosystems linking the roots of plants together via mycelium. This network allows for the building of fungal colonies and permits nutrient and resource transfer to take place. The interplant transfer of nutrients that takes place enhances the survival of the plants, creates defense mechanisms, and improves the growth of plants across ecosystems [6]. It has been found that the networks can influence the diversity within the plant community and most importantly the influence of interplant interactions [6]. These networks serve plants in helping to facilitate the cycling of carbon, nutrients, and water. On a broader scale, these networks are a key player in an
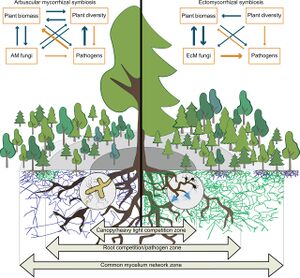
ecosystem as a whole as it helps to maintain ecological stability and evolution [6]. There exists a general lack of specificity between plants and fungi which suggests that plant species that share compatible mycorrhizal fungi lead to the building of plant communication communities [6]. Ecosystems that facilitate this symbiotic relationship between plant and fungi include temperate and boreal forests, woodlands, grasslands, Arctic tundra, woodland savannah, Mediterranean and Sclerophyllous woodlands, and tropical forests and woodlands [6]. The colonization of mycorrhizal networks is dependent upon soil environmental factors, alternate hosts, host plant stress, carbon allocation patterns, fungal genotype, plant genotype, and interactions between microbial communities in the soil [6].
The most important factor in the functioning of the mycorrhiza networks is the allocation of carbon from host plants. Fueled by carbon, the mycorrhizal networks power to act as avenues for the transfer of nutrients among plants within a community. Specifically, their transportation of materials allows for protection against disturbances in soil structure, soil microbe competition for resources, competition amongst plants, and chemical absorption of important nutrients in the soil [6]. The pathways of these networks seem to work with the mass flow of materials from a source-sink gradient which can be initiated by fungal growth or differences in nutrient levels across plants [6]. Water is another important material that is transported through these pathways. Not only do the extensive networks allow for a greater surface area which helps plants absorb more water, but it also provides great assistance during times of drought where the soil needs to be scavenged for water. Furthermore, resources can be carried by the water and transported to other plants as well through multiple interplant pathways within the mycorrhizal networks [6]. It can be seen that plants that live in stressed environments or have smaller route systems greatly benefit from these extensive networks.
The two major mycorrhizal fungi that form these networks include ectomycorrhiza and arbuscular mycorrhiza. These mycorrhizae are distinguished by the ecosystems they tend to be a part of. The ectomycorrhiza exists in temperate zone forests and boreal ecosystems [4][5]. Where areas arbuscular mycorrhizae exist in almost all terrestrial plants in their respective ecosystems [4][5]. A major difference between the two types of fungi is the nutrients in which they allocate to plants the most. The ectomycorrhiza seems to specialize in acquiring nitrogen for their plants were areas the arbuscular mycorrhiza seems to specialize in acquiring phosphorous for their host plants. Though both mycorrhizae facilitate the transport of the same micro and macronutrients, they differ in the emphasis on quantities of specific nutrients.
Ectomycorrhiza
In the broader group of mycorrhizae fungi, ectomycorrhiza is an important division that colonizes the roots of the target host plants and allows for the facilitation of nutrient uptake. Ectomycorrhizal fungi form a mutualistic relationship with their host plants which are primarily trees in temperate zones and boreal ecosystems [5]. The fungi which form these relationships mainly include Basidomycetes and Ascomycetes, but there are a wide variety of fungal species that fall under the ectomycorrhizal category [5]. Specifically, these fungi help to improve a plant's access to nutrients such as nitrogen and in turn receive photosynthetic carbon [1]. It has even been found that plants will specifically detect, discriminate and reward the fungi who present the greatest amount of nutrients with more carbohydrates [10]. The nutrient acquisition process that occurs between root and fungi is essential in aiding nutrient cycling as well as key soil processes, but most importantly the nutrient exchange operation is essential to the proper functioning of forest ecosystems [1]. It is also important to note that ectomycorrhiza fungi help their hosts during droughts and also help to create salinity tolerance.
Structure
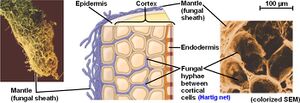
Structurally, the ectomycorrhiza is concentrated around the lateral roots of their hosts intertwining themselves in between root epidermal cells. Specifically, they form mycelial structures which surround the epidermal cells in an interlacing pattern within the root [1]. Otherwise known as the Hartig net, this structural setup of the fungi helps to create a larger surface area for the symbiotic partners which in turn allows for more nutrient acquisition to occur for both host and fungi [1]. The fungus encloses the root with a mantle from which hyphae extend into the surrounding soil [5]. Within the root, the hyphae place themselves between epidermal and cortical cells which forms what is known as the Hartig net [5]. The Hartig net is the nutrient exchange site between fungi and plant cells [5]. As mentioned before, these fungi receive photosynthetic carbon outputs from their host in exchange for limiting nutrients like macro and micronutrient’s which these fungi obtain for their hosts beyond the nutrition depletion zone surrounding their roots [1]. This is why the maximal surface area that the ectomycorrhizal creates for their host’s roots is incredibly crucial for nutrient uptake.
Nutrient Uptake
Speaking further on the topic of nutrient uptake, ectomycorrhizal fungi play an essential role in supplying plants with a key nutrient, nitrogen. The fungi can acquire the nitrogen through specific transporters they encode which take in nitrate and ammonium ions from the soil [1]. Furthering this process, the fungi also have specific enzymes in addition to the transporters which make use of the available organic and inorganic nitrogen sources [1]. Inorganic nitrogen intake is centered around ammonium as it is less energetically expensive to process compared to nitrate. Transporters AMT1 and AMT2 are characteristic of many of the ectomycorrhizal fungi species which are essential to the transport of ammonium [1]. The transport of nitrate into the fungi is a far more complicated process than the import of ammonium and requires a completely separate enzymatic pathway [1]. Organic nitrogen acquisition is the result of decomposing organic matter. The fungi have various mechanisms in which they have adapted to perform decomposition which primarily include oxidative mechanisms [1]. A common mechanism is Fenton chemistry, which breaks down lignin from plant cell walls to gain nutritional elements [1]. Interestingly, despite the availability of carbon in the organic matter, the ectomycorrhizal fungi only assimilate nitrogen and instead utilize the host-supplied carbon [1]. Additionally, another method of organic matter decomposition includes Class II peroxidases. It is important to note that the decomposition of organic matter to obtain nitrogen by the fungi is dependent upon the host’s ability to provide carbohydrates to the fungi, incentivizing the process [1].
Arbuscular Mycorrhizae
Within the general realm of mycorrhizal fungi, arbuscular mycorrhizae are a particular group that specifically helps its host to survive under stressful environmental conditions. Arbuscular mycorrhizae can also be classified as endomycorrhiza as they insert themselves into the cells of a root. Fungi that are included in the arbuscular mycorrhizae category are primarily from the Glomeromycotan phylum [5]. Approximately 70%-80% of terrestrial plants interact with this species of mycorrhizal fungi in the natural environment [4]. This fungus helps to aid in enhancing photosynthetic rate, water uptake, and gas exchange processes. Environmental stressors that they can help their host combat include salinity, temperature stress, drought, presence of metals in the soil, and fungal disease [2]. Generally, like all mycorrhizal fungi, arbuscular mycorrhizae enhance the surface area of their host’s root system which increases the uptake of important components plants gain from the soil. Furthermore, these fungi are a key endosymbiont for the facilitation of plant productivity which in turn allows for a higher functioning ecosystem around the plant [2]. The relationship between host and fungi is a classic example of a mutualistic relationship. Arbuscular mycorrhizal fungi are also highly regarded for their role in sustainable crop improvement [2].
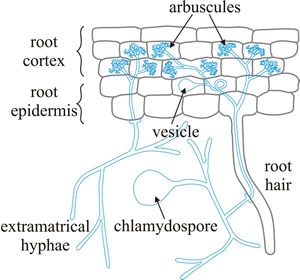
Structure
Structurally, this mycorrhizal fungi involves itself in the root system by forming hyphal networks. To form this hyphal network, its basis is mycelial which extends the root system and increases surface area immensely. The fungi specifically invade root cortical cells and form points of material exchange. Furthermore, vesicles and arbuscules are formed in conjunction with the hyphae [2]. Additionally, hyphae and spores are also a part of the hyphal network which exists in the rhizosphere [2]. This extensive network is the explanation of why plants who are in a mutualistic relationship with this fungi experience improvement in both plant growth and plant nutrition.
Nutrient Uptake
Nutrient uptake is significantly increased with the presence of Arbuscular mycorrhizal fungi. They help to enhance photosynthate production by concentrating higher amounts of macro and micronutrients around the root system which allows for the plant to significantly increase its biomass [2]. Most importantly, this fungi specifically increases the inorganic nutrient, phosphate, for its host plant. The arbuscles formed by the fungi aid in the exchanging of carbon and phosphorous which are essential to the plants' function and can help in environments where phosphorous is limited [2]. This essential nutrient exchange comes at a price for the fungi’s host as it is a two-way relationship in a mutualistic relationship. The host plants provide carbohydrates which are essential for the growth and survival of the fungi [3]. It has been found that the symbiont quality of the relationship between host and fungi is dependent upon the cost of carbohydrates transferred to fungi and phosphorous transferred to plant [3].
Specifically breaking down the phosphorous transport in arbuscular mycorrhizae, the pathway that does so is incredibly effective because of the increased surface area that the fungi provide for the root system which allows for larger volumes of soil to be scavenged [4]. The phosphorous is taken up and translocated by the hyphae in the hyphal network and delivered to cortical cells [4]. This phosphorous is then transferred to the plant by way of arbuscular mycorrhizae phosphorous transporters [4]. This specific phosphorous intake pathway prevents the depletion of phosphorous in the rhizosphere which allows for the continuous cycling of obtaining phosphorous for plant nutrition and growth.
Mycorrhizae Helper Bacteria (MHB)
Though mycorrhizal fungi are the facilitators are nutrient and content intake from the soil, the driving force behind the plant-fungi relationship is the symbiotic relationship that exists between the mycorrhizae and mycorrhizae helper bacteria (MHB). Mycorrhizae helper bacteria are bacteria that aid in the symbiotic relationship between root and fungus. Bacteria strains that promote arbuscular or ectomycorrhizal symbioses are nonspecific [5]. Bacteria that have been recorded that aid in the symbiotic process is gram-negative bacteria
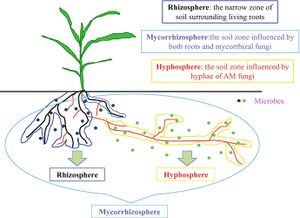
like Proteobacteria and gram-positive bacteria such as Firmicutes or Actinomycetes [5]. The MHB’s promote three components of the fungi and their interactions with their hosts: (1) it lessens environmental stressors on the mycelium of the mycorrhizal fungi, (2) it stimulates the extension of mycelial, and (3) it helps to facilitate the contact and colonization between root-fungus [5]. Also, specific activities that these bacteria perform that relate to plant survival specifically include the mobilization of nutrients from soil minerals, the protection of plants from root pathogens, and the fixation of nitrogen from the atmosphere [5]. These MHB activities establish why the bacteria are in symbiosis with the fungi which can then be in the symbiotic relationship with plants.
Specifically, it has been found that the bacteria associated with ectomycorrhiza work with and complement the fungi’s external mycelium which aid in the mobilization of nutrients [5]. Additionally, bacteria associated with ectomycorrhiza help to promote nitrogen fixation in areas of poor nitrogen levels which in turn help to improve plant nutrition [5]. In both relationships between ectomycorrhizal and arbuscular mycorrhizal fungi to mycorrhiza helper bacteria, help to protect root systems against root pathogens [5]. Furthermore, the mycorrhiza bacteria helpers for ectomycorrhiza and arbuscular mycorrhiza aid in the processing of soil minerals into nutrients that plants can absorb. Specifically, some bacteria associated with ectomycorrhizas can process iron from minerals and modify the pH of the rhizosphere [[5]. Some bacteria associated with arbuscular mycorrhizae have been found to enhance the process of phosphorous uptake from rocks that contain phosphate [5].
It is important to note that the study of mycorrhiza helper bacteria is a relatively new field, and a lot is still to be learned about the biological interactions that go on in the mycorrhizosphere. Initial studies have found general understandings, but a lot is hypothesized about the specific relationships between these bacteria and fungi.
Interplant Communication
Adaptive behaviors in plants can be altered through the linkage to other plants via mycorrhizal fungi. Some behaviors that can be changed include physiological aspects, defense responses, and gene regulation [7]. Inter-plant
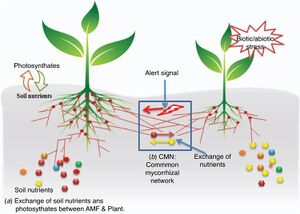
communities are built on the mycelial which vastly expand the soil and facilitate the linking of plants of the same or different species into mycorrhizal networks. This network allows for the interaction of plant and fungi which creates an adaptive social network capable of providing feedback and adaptation to the environment. Furthermore, these networks can be both evolutionary and ecologically significant as they increase the fitness of the plants and fungi [7]. These networks can be inclusive or exclusive, with the ability to have multiple plants and fungi species interacting together or singular species interacting with each other [7]. These specific interactions between plants, use the mycorrhizal networks for interplant resources exchange and as well as signal flux exchanges which can alter plant behavior [7]. The sources that tend to circulate within these networks include water, carbon, phosphorous, nitrogen, micronutrients, macronutrients, and stress chemicals [7]. The exchange of interplant resources is regulated by source-sink relationships that exist in the mycorrhizal network [7]. Specifically, a plant that is recognized to be rich in nutrients can take on the role of a donor for a plant that is poor in nutrients (the sink) [7]. This exchange is driven by mass flow as a result of the source-sink gradient created by plants having different nutrient levels [7].
Tomato Plant Defense Systems
As mentioned previously, the mycorrhizal networks are facilitators of changes in plant behavior by the transfer sources and signal fluxes between plants [7]. Specifically, the exchange of information (signals) such as the release of stress chemicals or defense responses can be spread through the mycorrhizal networks. Tomato plants have been found to demonstrate this phenomenon and signal to neighboring tomato plants that there is a pathogen infestation. Furthering these concepts, a study was done that examines the mycorrhizal network as an information channel that relays defense signals between connected plants via the mycelia of Glomus mosseae [8]. The study found that when plants connected by way of mycorrhizal networks to plants that were the pathogen-infected ones, they exhibited higher levels of production of the defense-related enzymatic processes and gene expression [8]. This proves that these networks of fungi can serve as allocation networks for nutrients, water, and defense networks within the plant communities [8]. The researchers ultimately demonstrated that mycorrhizal communication networks are facilitators of the transferring of defense signals and disease resistance between the pathogen-infected plants and the neighboring healthy plants [8]. To conclude, the tomato plant model further exemplifies mycorrhizal networks as transfer networks for plants and ultimately proves that plants can communicate information within communities.
Interplant Cycling of Phosphorous
Studies have shown that within a mycelial network, individuals that are connected can be involved in preferential nutrient cycling, such as the cycling of phosphorous [9]. One mode which causes the cycling of nutrients, like phosphorous, comes from when the dying roots of one plant send sources to another plant [9]. A specific study examined the transfer of phosphorous between dead roots of a grass plant (with had arbuscular mycorrhiza) to roots of three deciduous trees (some trees have arbuscular mycorrhiza, and some have ectomycorrhiza), the cycling of nitrogen and phosphorous between grass and ectomycorrhizal and arbuscular mycorrhizae trees, and the transfer of phosphorous between heathland plants [9]. It was ultimately found that trees with specific mycorrhizal obtained greater amounts of phosphorous allocated from the roots of the dying grass plants which dictates one mode in which phosphorous can be shared among plants [9]. Further findings indicated that among heathland plants, acquisition of phosphorous came from dying roots of neighboring plants as well the normal cycling of nutrients [9]. Overall the study illustrates that nutrient cycling in plant communities depends on soil nutrient availability, turnover of plants or plant parts, physiological imbalances in plants (nutrient concentrations), and strong mycelial networks.
It is important to note that other nutrients, like nitrogen, water, and other soil minerals are cycled between plants just like phosphorous.
Conclusion
Thinking about the holistic relationship of the plant and fungi, plants are linked together by their mycorrhizae symbionts. The extension of fungal hyphae in the soil extends the surface area of roots which helps to exploit the soil’s mineral nutrients and water shared between the host plant and fungi. Both arbuscular mycorrhiza and ectomycorrhiza are the key types of mycorrhizal fungi involved in the creation of these beautiful communication networks underground. They provide nutrients to plants such as nitrogen, phosphorous, and other minerals alongside water and even information. Important signaling comes from these systems as well such as those of defense or chemical stressors. These incredible fungi construct underground communities that may not be seen but have the greatest impact on the world above the soil horizon. They play a key role ecologically in ecosystems everywhere.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 [Stuart EK and Plett KL (2020) Digging Deeper: In Search of the Mechanisms of Carbon and Nitrogen Exchange in Ectomycorrhizal Symbioses. Front. Plant Sci. 10:1658. doi:10.3389/fpls.2019.01658]
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 [Begum N, Qin C, Ahanger MA, Raza S, Khan MI, Ashraf M, Ahmed N and Zhang L (2019) Role of Arbuscular Mycorrhizal Fungi in Plant Growth Regulation: Implications in Abiotic Stress Tolerance. Front. Plant Sci. 10:1068. doi: 10.3389/fpls.2019.01068]
- ↑ 3.0 3.1 3.2 [By E. Toby Kiers, Marie Duhamel, Yugandhar Beesetty, Jerry A. Mensah, Oscar Franken, Erik Verbruggen, Carl R. Fellbaum, George A. Kowalchuk, Miranda M. Hart, Alberto Bago, Todd M. Palmer, Stuart A. West, Philippe Vandenkoornhuyse, Jan Jansa, Heike Bücking Science12 Aug 2011 : 880-882. DOI: 10.1126/science.1208473]
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 [Sally E. Smith, Iver Jakobsen, Mette Grønlund, F. Andrew Smith, Roles of Arbuscular Mycorrhizas in Plant Phosphorus Nutrition: Interactions between Pathways of Phosphorus Uptake in Arbuscular Mycorrhizal Roots Have Important Implications for Understanding and Manipulating Plant Phosphorus Acquisition, Plant Physiology, Volume 156, Issue 3, July 2011, Pages 1050–1057, https://doi.org/10.1104/pp.111.174581]
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 [Frey‐Klett, P., Garbaye, J. and Tarkka, M. (2007), The mycorrhiza helper bacteria revisited. New Phytologist, 176: 22-36. https://doi.org/10.1111/j.1469-8137.2007.02191.x]
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 6.8 6.9 [Suzanne W. Simard, Kevin J. Beiler, Marcus A. Bingham, Julie R. Deslippe, Leanne J. Philip, François P. Teste. Mycorrhizal networks: Mechanisms, ecology and modelling. Fungal Biology Reviews. Volume 26, Issue 1, 2012, Pages 39-60, ISSN 1749-4613, https://doi.org/10.1016/j.fbr.2012.01.001.]
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 7.8 7.9 [Gorzelak, M. A., Asay, A. K., Pickles, B. J., & Simard, S. W. (2015). Inter-plant communication through mycorrhizal networks mediates complex adaptive behavior in plant communities. AoB PLANTS, 7, plv050. https://doi.org/10.1093/aobpla/plv050]
- ↑ 8.0 8.1 8.2 8.3 8.4 [Song, Y. Y., Zeng, R. S., Xu, J. F., Li, J., Shen, X., & Yihdego, W. G. (2010). Interplant communication of tomato plants through underground common mycorrhizal networks. PloS one, 5(10), e13324. https://doi.org/10.1371/journal.pone.0013324]
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 [Eason, W.R., Newman, E.I. & Chuba, P.N. Specificity of interplant cycling of phosphorus: The role of mycorrhizas. Plant Soil 137, 267–274 (1991). https://doi.org/10.1007/BF00011205]
- ↑ 10.0 10.1 [Marc-André Selosse, Franck Richard, Xinhua He, Suzanne W. Simard. Mycorrhizal networks: des liaisons dangereuses? Trends in Ecology & Evolution. Volume 21, Issue 11, 2006, Pages 621-628, ISSN 0169-5347, https://doi.org/10.1016/j.tree.2006.07.003.]
