Persister Cells in E. coli: Difference between revisions
No edit summary |
No edit summary |
||
| Line 2: | Line 2: | ||
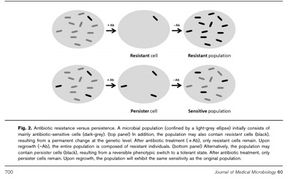
Bacterial communities form dormant, antibiotic tolerant cells called persister cells.<sup>5</sup> These cells are not mutants, but phenotypic variants of the wild type. <sup>5</sup> Persisters are selected to increase chances of survival in fluctuating environments. <sup>2</sup>. The diminished (or in some cases, overproduction) activity of key molecules in the cell, as well as non-growing or reduced growth rate, make cells in dormant state distinguishable. <sup>2</sup> Persisters are found in all phases of cell growth and are non-dividing with a reduced metabolism. <sup>2,10</sup> The slow growth of the persister cells protects them from antibiotics.<sup>1</sup> Cells in the persister state are highly immune to bacterial antibiotics, which interfere with processes of key molecules. <sup>1</sup> Persisters may represent the long-looked-for explanation for biofilm tolerance to antibiotics. <sup>6</sup> <i>Escherichia coli</i> does not make persister cells in early-exponential growth, which indicates presence of specific persister genes. <sup>5</sup> | Bacterial communities form dormant, antibiotic tolerant cells called persister cells.<sup>5</sup> These cells are not mutants, but phenotypic variants of the wild type. <sup>5</sup> Persisters are selected to increase chances of survival in fluctuating environments. <sup>2</sup>. The diminished (or in some cases, overproduction) activity of key molecules in the cell, as well as non-growing or reduced growth rate, make cells in dormant state distinguishable. <sup>2</sup> Persisters are found in all phases of cell growth and are non-dividing with a reduced metabolism. <sup>2,10</sup> The slow growth of the persister cells protects them from antibiotics.<sup>1</sup> Cells in the persister state are highly immune to bacterial antibiotics, which interfere with processes of key molecules. <sup>1</sup> Persisters may represent the long-looked-for explanation for biofilm tolerance to antibiotics. <sup>6</sup> <i>Escherichia coli</i> does not make persister cells in early-exponential growth, which indicates presence of specific persister genes. <sup>5</sup> | ||
==Types of Persistence== | ==Types of Persistence== | ||
| Line 13: | Line 12: | ||
Type II persisters are repetitively produced without environmental triggers and stressors and are slow growing. <sup>1</sup> These “bet-hedging” strategies of persistence are believed to have evolved from antibiotic stressors and stress imposed on bacteria by prophages. <sup>7</sup> | Type II persisters are repetitively produced without environmental triggers and stressors and are slow growing. <sup>1</sup> These “bet-hedging” strategies of persistence are believed to have evolved from antibiotic stressors and stress imposed on bacteria by prophages. <sup>7</sup> | ||
<br> <br> | <br> <br> | ||
| Line 25: | Line 23: | ||
The first candidate persister gene identified was the hipA gene encoded by the hipBA locus. HipA is a protein kinase that makes EF-Tu nonfunctional by phosphorylation. This inhibits protein synthesis and leads to tolerance of many drugs, possibly even persister formation <sup>5</sup>. | The first candidate persister gene identified was the hipA gene encoded by the hipBA locus. HipA is a protein kinase that makes EF-Tu nonfunctional by phosphorylation. This inhibits protein synthesis and leads to tolerance of many drugs, possibly even persister formation <sup>5</sup>. | ||
Revision as of 21:02, 17 March 2014
Bacterial communities form dormant, antibiotic tolerant cells called persister cells.5 These cells are not mutants, but phenotypic variants of the wild type. 5 Persisters are selected to increase chances of survival in fluctuating environments. 2. The diminished (or in some cases, overproduction) activity of key molecules in the cell, as well as non-growing or reduced growth rate, make cells in dormant state distinguishable. 2 Persisters are found in all phases of cell growth and are non-dividing with a reduced metabolism. 2,10 The slow growth of the persister cells protects them from antibiotics.1 Cells in the persister state are highly immune to bacterial antibiotics, which interfere with processes of key molecules. 1 Persisters may represent the long-looked-for explanation for biofilm tolerance to antibiotics. 6 Escherichia coli does not make persister cells in early-exponential growth, which indicates presence of specific persister genes. 5
Types of Persistence
Persistence is triggered by environmental and intracellular stressors. While the process of differentiation is not completely understood, there is some evidence suggesting how and why cells become persistent. There are two different types of persistence.
Type I persisters are triggered to differentiate and become physiologically antibiotic tolerant, using strategies to resist persistence unless exposed to stressful conditions, such as starvation. This suggests that the bacteria sense these stressful environments and in preparation, push part of the population to become dormant. The rest of the population is left to thrive and risk death. 1 Type I persisters are generated at the stationary phase of bacterial growth. 2
Type II persisters are repetitively produced without environmental triggers and stressors and are slow growing. 1 These “bet-hedging” strategies of persistence are believed to have evolved from antibiotic stressors and stress imposed on bacteria by prophages. 7
Genetic Mechanisms
There are at least 37 known types of Toxin-Antitoxin (TA) systems in Escherichia coli, which are thought to be the genetic basis for the formation of persisters from normal cells. 10 These toxin mechanisms usually consist of two genes encoding for stable toxins that disturb essential processes, and an unstable antitoxin that remedies the effects of the toxin. 10 Toxin genes always code for proteins and the antitoxins code for either protein or RNA. 3
Under type I conditions, the antitoxins are degraded by proteases in the cell, ClpXp or Lon, and the toxins are released to do their typical function in the cell 3 The most well depicted system is the Type II TA system, made up of a two-gene operon which encodes a stable toxin and an unstable antitoxin, MqsR/MqsRA. This TA system produces proteins that come together to form a complex that is not toxic. The antitoxin mqsA can be found just below the mqsR in the same operon, and encodes a slightly longer amino acid protein that neutralizes mqsR toxicity. 3 Evidence suggests that the mqsRA TA system plays an important part in many of Escherichia coli cellular processes. Deletion of mqsR and the mqsRA operon decreased persister cell formation, while mqsR overexpression increased persister formation. This is important because mqsA has also been shown to serve as a transcriptional regulator of several Escherichia coli genes, including general response regulators, as well as the mqsRA operon. 3
The first candidate persister gene identified was the hipA gene encoded by the hipBA locus. HipA is a protein kinase that makes EF-Tu nonfunctional by phosphorylation. This inhibits protein synthesis and leads to tolerance of many drugs, possibly even persister formation 5.
Section 3
Include some current research in each topic, with at least one figure showing data.
Further Reading
[Sample link] Ebola Hemorrhagic Fever—Centers for Disease Control and Prevention, Special Pathogens Branch
References
Edited by (your name here), a student of Nora Sullivan in BIOL168L (Microbiology) in The Keck Science Department of the Claremont Colleges Spring 2014.