Amphibacillus xylanus: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{Uncurated}} | |||
<h2> <span class="mw-headline" id="Classification">Classification</span></h2> | <h2> <span class="mw-headline" id="Classification">Classification</span></h2> | ||
<p><ul> | <p><ul> | ||
Latest revision as of 02:01, 13 August 2013
Classification
- Kingdom - Bacteria
- Phylum - Firmicutes
- Class - Bacilli
- Order - Bacillales
- Family - Bacillaceae
- Genus - Amphibacillus
Species
|
NCBI: Taxonomy |
Amphibacillus xylanus
Description and Significance
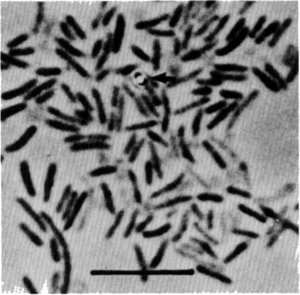
Amphibacillus xylanus is a Gram positive, spore-forming, and chemoorganotrophic bacterium that was isolated from composts of manure with grass and rice straw. They are also a facultative anaerobic organism that can grow in several different environments. However, it lacks cytochrome, quinone, and catalase. This helps explain the method of metabolism that Amphibacillus xylanus relies on.
Amphibacillus xylanus is also flagellated and motile, and it maintains many of the characteristics known to Amphibacillus. For example, the cells are rod-shaped and are 0.3 to 0.5 micrometers in diameter and are 0.9 to 1.9 micrometers long. The spores are oval in shape and heat resistant. The sporangia, where the spores are formed, are lysed and the spores are then released. These spores are formed under both aerobic and anaerobic conditions. Amphibacillus xylanus does not grow in nutrient broth, but it produces ethanol, acetic acid, and formic acid under anaerobic conditions and acetic acid under aerobic conditions from glucose. When colonies are grown on glucose agar, the colonies are small, circular, smooth, convex, entire, and white after only one day of incubation.
The microorganism grows ideally at pH values between 8 and 10, but it cannot grow at pH 7. The major cellular fatty acids included,saturated straight chains, anteiso-branched chains and iso-branched chains.
Genome Structure
The sequencing of the Amphibacillus xylanus genome was completed in October, 2012 and it is under review for publishing, but the sequence can be found here. Amphibacillus xylanus has a circular genome with 2569486 nucleotides and within the genome there are approximately 2411 protein genes and 78 RNA genes. It has been found that Amphibacillus xylanus is not related to representative species of the genera bacillus, which include clostridium and sporolactobacillus. In fact, little of its genome has matched any other bacterium. In the SinIV, 15abb, and EpO1<font: super>T, the DNA genome had a 36-38 cytosine + guanine percentage.
Very little of Amphibacillus xylanus' genome has been found to match the genome of other bacteria, and even though Gram-positive microorganisms are taxonomically distant from Gram-negative organisms, the amino acid sequence of A. xylanus' NADH oxidase was found to share 51.2% similarities with the alkyl hydroperoxide reductase from S. typhimurium. Another protein in Amphibacillus xylanus is found to have a 64.3% similarity to the AhpC of S. typhimurium.
Metabolism and Life Cycle
Amphibacillus xylanus is distinct due to its multiple metabolic pathways. It is capable of digesting xylan aerobically and anaerobically. In anaerobic cultures, the reducing agent was titanium (III) citrate and the fermentation product of the reaction was formic acid, acetic acid, and ethanol. Therefore, under anaerobic conditions, Amphibacillus xylanus is a heterofermentor. This means that the bacterium produces more than one major end product. Under aerobic conditions, the reducing agent was shaking and the fermentation product was found to be acetic acid. One of the bioenergetics properties of Amphibacillus xylanus is the fact that the flavoprotein NADH oxidase from Amphibacillus xylanus consumes oxygen to produce hydrogen peroxide. Typically, a microorganism would utilize a cytochrome system to consume oxygen, but since Amphibacillus xylanus does not have a respiratory system available, it instead utilizes NADH oxidase. However, the use of NADH oxidase explains how the microbe can grow effectively in both anaerobic and aerobic conditions. Furthermore, it is believed that NADH is in control of the intracellular redox balance of the cell under aerobic conditions. Despite the fact that Amphibacillus xylanus lacks a respiratory system and hemeproteins, catalase, and peroxidase, it is still able to grow efficiently and it has the same growth rate under both anaerobic and aerobic conditions. It is able to do so because it has aerobic and anaerobic pathways that produce similar amounts of ATP.
Amphibacillus xylanus is also able to utilize D-xylose, L-arabinose, D-ribose, D-glucose, D-fructose, esculin, salicin, maltose, sucrose, cellobiose, trehalose, soluble strach, and pectin, along with the xylan obtained from oat spelt.
A. xylanus' NADH oxidase has been found to have unique functional properties that are different from other NADH oxidases. NADH oxidase was found to catalyze the electron transfer between NADH and DTNB (requiring 6 electrons), and this leads to the idea that there are redox centers in addition to the FAD found in the protein. Once further tests were run, the two disulfides that served as the redox centers were found to be the cys-337 and cys-340 within the NADH oxidase. Amphibacillus xylanus is able to perform alkyl-hydroperoxide reductase activity in the presence of AhpC. This activity is very dependent on ionic strength as well as salt concentration. The absence of ammonium sulfate lowers the activity. For the salt concentrations, the stimulation was greatest with phosphate and the least with chloride. This pattern suggests that the activity by salts is due to enhancing the hydrophobic interaction between the flavoprotein (NADH oxidase) and the simple disulfide protein (AhpC).
Ecology and Pathogenesis
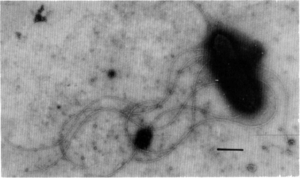
Amphibacillus xylanus was first isolated from alkaline compost of manure with grass and rice straw and it can survive in a variety of environments because of its multiple metabolic pathways. It is a chemoorganotroph and can be found in both anaerobic and aerobic conditions because it is a facultative anaerobic organism. Even though Amphibacillus xylanus does not have a respiratory chain, the microbe has the same cell growth rate and cell yield under both strict anaerobic and aerobic conditions. This is because similar high amounts of ATP are produced under both conditions. Despite the microbe’s ability to grow equally in both aerobic and anaerobic conditions, Amphibacillus xylanus requires other environmental conditions in order to survive and grow in a habitat. First, Amphibacillus xylanus can grow in temperatures between 25 ° C and 45 ° C. However, it cannot grow at a temperature of 50 ° C and above. The microorganism can also grow in the presence of 3% NaCl, but not in the presence of 6% NaCl. The range of pH that Amphibacillus xylanus can tolerate is from pH 8 to 10. However, it cannot grow effectively at a pH of 7.
References
Author
Page authored by Lauren Jackson and Hillary Albert, students of Dr. N. Walker at Michigan State University.
