Application of Wolbachia in Invertebrate Vector Control
By: Chinagozi Ugwu
Introduction
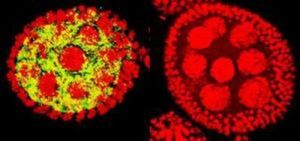
The race to decrease, or even totally eliminate, the persistence of vector-borne infectious diseases has been ongoing for years, if not generations. A parasitic new champion has been found in the endosymbiont bacterium by the name of Wolbachia pipientis. The α-proteobacteria, Wolbachia, is a gram-negative intracellular parasite. Wolbachia is naturally present in over 20% of all insects. [1] It was first discovered in the ovaries of Culex pipiens mosquito. Wolbachia forms either an obligate or a facultative relationship with its host, and these interactions have many effects. This endosymbiont bacterium is in the family of Rickettsiacea. However, Wolbachia has not been found to be directly pathogenic to humans because there is no transmission pathway in humans. Wolbachia is primarily transmitted within invertebrates either vertically, which is transmission of disease directly from mother to child, or horizontally by contact with members of an infected species. [2] The ovaries of fruit flies with the infection fluoresces yellow because the bacteria infects the flies eggs for vertical transmission (Fig.1).
Many invertebrates are infected by Wolbachia, and the bacteria’s success may be credited to the diverse phenotypes that result from infection. The phenotypes range from mutualism to reductive parasitism.[3]Invertebrates such as nematodes form mutualistic relationships with Wolbachia whereas, fruit flies and mosquitoes are involved in reductive parasitic relationships[4].While the first relationship is necessary for nematodes[5],the parasitic relationships with the arthropods may result in early death or exclusion of some male and older members of a population by Wolbachia.[6] Inheritance of this bacterial infection mainly occurs by maternal transmission through the cytoplasm of eggs, therefore male arthropods are not necessary for the successful transmission of Wolbachia. The need for females and not males results in Wolbachia having many physiological and reproductive effects, which may reduce male presence of the host species. This bacteria can feminize infected males so that they can produce more eggs with bacterial cells. [7]
The facultative endosymbiont relationship between Wolbachia pipientis and host invertebrate organisms allows for increased bacterial propagation and persistence in host populations, but Wolbachia is hardly ever found to be beneficial to these host organisms. In arthropods and many other invertebrates, Wolbachia infection can modify host reproduction for a variety of species in such ways as; reproductive incompatibility, thelytokous parthenogenesis, male-killing, and feminization.[8]
Recently, Wolbachia, was found to be medically important for biologically combating vector-borne infections.[9] These bacteria could be used for population replacement and suppression of vector organisms such Aedes aegypti mosquito which spreads dengue and yellow fever, Culex pipiens mosquito which spreads West Nile Virus, and anopheles mosquito that act as the vector organism for malaria. [10]
Physiological and reproductive Effects of Wolbachia
The parasitic endosymbiont Wolbachia pipientis induces a variety of reproductive disorders in their host organisms. These disorders are often specie specific and include: parthenogenesis in Hymenoptera and Thysanoptera; feminization in isopod crustaceans; male-killing in Lepidoptera; Drosophila and Coleoptera; and sperm competition and cytoplasmic incompatibility in a diverse array of arthropods. Wolbachia, also has many physiological and reproductive effects on arthropods, such as mosquitoes. The deleterious effects of Wolbachia on mosquitoes is very important as mosquitoes have several species that act as vectors for deadly infectious diseases such as Dengue Fever [11].
Thelytokous parthenogenesis is a form of asexual reproduction, in which, females develop from eggs that are unfertilized. While most microbe-associated thelytokous are reversible, Wolbachia induced thelytokous parthenogenesis, however, is irreversible [12] . Wolbachia bacteria induces irreversible parthenogenesis by causing the reduction and loss of genes: for sperm maturation; for mating behavior of females; for a major muscle in female sperm reservoir; and the spermatheca. If females infected by parthenogenesis-inducing Wolbachia are successfully cured by using antibiotics to remove the bacteria, they can then produce male offspring. Most often, however, the male offspring are not sexually functional, and the formerly infected females become unable or unwilling to mate [13].
Wolbachia induces feminization in isopod crustaceans through conversion of genetic males into functional females. [14] These converted females produce higher female densities in the population. This feminization process is mediated by Wolbachia feminization strains that are transferred through eggs. Male-killing strains of Wolbachia are elicited in organisms of the order Lepidoptera, Drosophila and Coleoptera. The reduction of the male population in a species by Wolbachia is to increase the chances of the bacterial cells being transmitted down the generational line.
[15]To Wolbachia the males in a population are unimportant because the male sperm cells are too small to harbor the bacterial cells for propagation. In contrast the female egg cells are important, therefore, females are favored. Wolbachia infected males in some host species experience decreased sperm competition in comparison to uninfected males of the same species. Significantly fewer sperm is produced by males that are infected with Wolbachia, due to the reduction in sperm competitive ability. [16] Another type of reproductive manipulation, which will be discussed next is cytoplasmic incompatibility and this is the most prominent feature used for reproductive manipulation by Wolbachia.
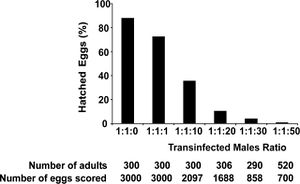
The endosymbiont, Wolbachia, imposes cytoplasmic incompatibility in many arthropods, resulting in embryonic mortality. When an infected male mates with an uninfected female, unidirectional cytoplasmic incompatibility arises; however, uninfected males can successfully mate with infected females, as can two infected parents. [17] At the other end of the spectrum, bidirectional cytoplasmic incompatibility occurs between individuals infected with different Wolbachia strains, when they mate. Wolbachia affects mitotic division as a result of spermatogenesis paternal chromosome modification. This modification thus causes a loss in mitotic synchrony. The importance of cytoplamsic incompatibility induced by Wolbachia is for use in stopping mosquito vector disease transmission. Wolbachia can manipulate reproduction by causing a form of sterility known as cytoplasmic incompatibility (CI), which prematurely stops the development of early embryos. When a Wolbachia infected male mates with an uninfected female or a female infected by a different strain mates, then cytoplasmic incompatibility occurs. Some Wolbachia strains stimulate an extremely high cytoplasmic incompatibility (CI) which leads to increased population invasion and also increased mortality and suppression in the mosquito population as shown in figure 3. Strains such as wRi Wolbachia strain spread at a rate of 100km/yr as a consequence of cytoplasmic incompatibility. The increased mortality of vector mosquitoes has no impact on the Wolbachia strains because the host organism reproduce prior to death. [18] The life-shortening strain spreads, due to the benefit of higher reproductive success that arises from cytoplasmic incompatibility of individuals with Wolbachia bacterial infection.
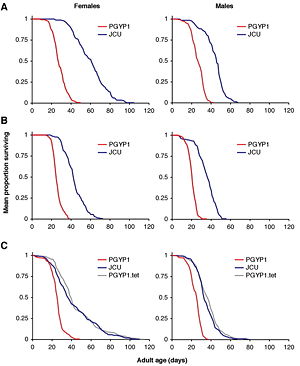
The most virulent Wolbachia strain, popcorn (wMelpop) associated with brain degeneration in its host fly, was isolated from the organism D. melanogaster. This strain, wMelpop, reduces the life-span of the infected flies to about 50% compared to uninfected flies.[19] Density of wMelpop strain in different tissues, at the adult stage, is drastically higher than during the earlier stages of life, such as the larval and pupal stages. Bacterial cells accumulate at such high densities, in different tissues, resulting in early death because of the overwhelming host cell pathology.
There are several physiological factors that influence how effective wMelpop is at shortening the life-span of infected organisms. Temperature is a very important physiological factor. At higher temperatures, the virulence of wMelpop increases while the life-span of the bacterial host organism drastically decreased. For instance in the fly D. melanogaster, wMelpop had little to no effect on the life-span at 19 °C, however, at 25 °C there was a considerable decrease in life-span (fig. 2). It is of great medical interest to employ Wolbachia strains, such as wMelpop, for reduction of Dengue Fever transmission by altering the age structure and life-span of Ae. aegypti. Since only adult, female, Ae. aegypti mosquitoes are capable of pathogenesis the introduction of wMelpop strains would eliminate the part of the mosquito population responsible for transmitting Dengue Fever. Theoretical models revealed that transinfection with wMelpop strains of Wolbachia could significantly reduce transmission of disease about 90% or more.
Wolbachia Transmission Methods
The most widely known natural means of Wolbachia pipientis infection is through vertical transmission, into the cells in the female egg. Vertical transmission from mother to offspring, ensures that Wolbachia infection is passed down in generations. This form of transmission is important and effective because of the bacterium’s intracellular nature. Vertical form of Wolbachia transmission occurs only within populations, in a species. Reduction in mitochondrial variation as a result of Wolbachia selecting for females can only occur if vertical transmission is the mechanism of Wolbachia infection [20]. In addition,it is also possible for Wolbachia bacterial infection to be transmitted horizontally.
In horizontal transmission, Wolbachia infection may occur between individuals or even between species. Horizontal transmission of Wolbachia unlike vertical transmission weakens the association between Wolbachia and the host's mitochondria. A study by Viljakainen et al in 2008 revealed that horizontal transmission occurs naturally and more frequently than expected. Two common strains of Wolbachia were discovered to infect a group of wood ant species, Formica rufa and F. exsecta. The persistence of the Wolbachia infections, in the Formica wood ant species, may be contributed to the combination of horizontal and vertical transmission.
The use of Wolbachia to combat vector-borne diseases is favored because insecticides and pesticides have failed. Chemical agents have failed because, after a period of time the insects become resistant. This constant development of resistance by pest and vector invertebrates has influenced the need and research for a biological agent. Vector mosquitoes are the primary targets for the Wolbachia led combat because of the potent pathogens vector-borne disease they carry. Even though Wolbachia is present in many other species of mosquitoes, most vector mosquitoes do not have naturally occurring Wolbachia infections.
The Anopheles mosquito species were assayed and, in over 30 species examined, Wolbachia was absent in all. Chromosomal testing was done on Anopheles gambie to determine whether Wolbachia had once upon a time been present in this species of Anopheles mosquitoes. Researchers found results that are indicative of Wolbachia's presence in this species of vector mosquito.
The chromosome 3R region coding for Wolbachia type proteins was identified. The salivary gland transcriptome data suggest that ancestral Anopheles were infected with Wolbachia via horizontal transmission of the bacteria. However, because horizontal transmission is not as stable as vertical transmission, Wolbachia infection could not persist in Anopheles mosquitoes. The researchers hypothesized that the current resistance of Anopheles against Wolbachia could be accredited to the Wolbachia-like transcripts. The transfer of the highly pathogenic Wolbachia strain, wMelpop, from its natural host, D. melanogaster, into A. aegypti was done by bacterial adaptation. This was possible by implementing serial passage into A. aegypti cell culture for 3 years. The culturing of Wolbachia in A. aegypti caused a genetic adaptation, so that virulence decreased when the strain was transferred back to Drosophila.
Several different methods are employed for transinfection of Wolbachia into different organisms. A variety of techniques are used even for studying the effects of different Wolbachia strains on vector mosquitoes. Wolbachia strain, wAlbB from Aedes albopictus mosquito was successfully infected into An. gambiae using the modified shell-vial technique. Other in vivo methods for Anopheles infection with Wolbachia include embryonic micro injection in Drosphila, to obtain that organisms strain of Wolbachia. The embryonic microinjection protocols may also be used to infect Anopheles with bacteria from Aedes aldopictus and Aedes aegypti. Transfer protocols for transfer of Wolbachia by directly injecting into adult Aedes mosquito and Drosphila has also been developed. The transfer of Wolbachia into the malaria causing mosquito, Aedes gambiae is being done with both the embryonic and adult injection protocols.
Therefore, Wolbachia’s successes and persistence depends on the fitness of its host.
Wolbachia further insures its own success by tremendously increasing the number of offspring infected females produces, compared to the number of offspring that uninfected females produce. Female mosquitoes with Wolbachia bacterial infection can mate with both infected and uninfected males successfully, however, the reverse of this is unsuccessful. That is, when an uninfected female mosquitoes attempt to mate with Wolbachia-infected males, some or all of the uninfected female’s fertilized eggs die. This Wolbachia induced reproductive behavior will undoubtedly increase the population of mosquitoes with Wolbachia infection, which in turn, maintains life-shortening Wolbachia in the populations of such vector- mosquitoes as those that transmit dengue fever.
Pathogen Exclusion in Vector Mosquitoes with Wolbachia
The endosymbiont Wolbachia is capable of forming many types of relationships with invertebrates. In some instances this intracellular α-proteobacterium interacts as an obligatory mutualist and still with other invertebrates and at other instances it acts as a facultative parasite. The relationship between filarial nematodes is that of obligatory mutualism. Infection of mosquitoes, especially transinfection of vector-mosquitoes results in a parasitic interaction between Wolbachia and the arthropod.
The only instance in which there exists an obligate relationship between Wolbachia and a host, is in the case of filarial nematode.In this case the removal of the Wolbachia strain present, with antibiotics, in pathogenic nematodes is disastrous for the nematode. Without the intracellular bacteria, the filarial nematode loses most of its reproductive and pathogenic function amongst other things. Filarial nematodes are parasites that are responsible for many diseases. Wolbachia has been discovered in filarial namatodes of Onchocercidae family. This family of filarial nematodes includes human pathogens such as elephantiasis and lymphatic filariasis caused by Wuchereria bancrofti, river blindness caused by Onchocerca volvulus and heart worms in dogs. These nematodes depend upon Wolbachia for development and fertility. They have established such long and stable relationship with Wolbachia through vertical transmission. Treatment of filarial caused diseases with antibacterial drugs removes Wolbachia [21]. Filarial nematode treated with tetracycline successfully kills the bacteria and also affects the nematode parasite. Antibiotics are effective treatments for filarial diseases because the removal of Wolbachia also negatively affects the nematode parasites. Abnormalities occur in growth of nematodes treated with tetracycline, decreased fecundity, shortened life-span and moulting are some of the abnormalities.

Treatment of non-Wolbachia infected nematodes with tetracycline does not show similar results, therefore strengthening the hypothesis of mutual interaction between pathogenic nematodes and Wolbachia. The relationship between Wolbachia and other organisms that may be pathogenic to humans are not obligate. Arthropods such as many vector- mosquitoes do not have Wolbachia. When these mosquitoes interact with Wolbachia the results are not favorable for the arthropod.
Mosquitoes are flying insects in the Culicidae family, with more than 2000 different species. The males and females mosquitoes both feed on nectar but the females of some species of mosquito are capable of sucking blood from humans and animals. The hematophagic activity of female mosquitoes is essential for production of eggs and has made them one of the deadliest known disease vectors that claim millions of lives each year.
Mosquitoes act as vectors for a variety of parasites and pathogens. Many mosquito-borne diseases such as West Nile Virus, Dengue Fever, Malaria, Yellow fever are transmitted by different species of mosquitoes. Yellow Fever, West Nile Virus and Dengue Fever are all caused by a family of viruses called Flaviviridae. Culex pipiens mosquitoes act as the primary vectors for the West Nile Virus in America while female Aedes aegypti mosquitoes are the disease vectors that transmit Dengue Fever and Yellow Fever. The vector-borne infectious disease, malaria can be caused by one of four types of Plasmodium parasites that can infect humans, with Plasmodium falciparum causing the most dangerous infection to humans. Malaria is transmitted by infected female Anopheles mosquito.
Many mosquito species have naturally occurring Wolbachia bacterial infections which can induce strong CI. Major disease vector mosquitoes like the Anopheles and Ae. aegypti mosquitoes, however, do not have naturally occurring Wolbachia bacterial infections. Since Ae. aegypti mosquitoes do not have Wolbachia the wAlbB strain was transferred from Ae. Albopictus to Ae. Aegypti. The successful transinfection of Ae. Aegypti. with blastoderm stage embryo method demonstrates that Ae. aegypti is a stable Wolbachia host. Adult Ae. aegypti mosquitoes were also successfully double infected with wAlbA and wAlbB/i> from Ae. Albopictus using the adult microinjection technique. The embryonic microinjection technique was most recently used to successfully transfer into the Ae. aegypti mosquito is wMelpop.
Recent collaborative research demonstrated that infection of Aedes aegypti mosquitoes, the vector for Dengue Fever and Yellow Fever, with a maternally inherited Wolbachia bacteria strain, wMelpop, shortened the life span of adult mosquitoes under laboratory conditions. The Flaviviridae viruses like most other pathogens, need a long time to develop in their vector-mosquitoes. Wolbachia induces cytoplasmic incompatibility which combined by the high frequency of Wolbachia’s inheritance, will allow suppression and eventually replacement of the natural population, thus reducing or eliminating Dengue and Yellow Fever. In order to stabilize the wMelpop strain within Ae. aegypti’s intracellular environment, an in vitro infection in RML-12 was established prior to wMelpop transfer into Ae. Aegypti. Temperature was also an important factor in determining the effect of Wolbachia as wMelpop virulence depended on temperature. Virulence increased dramatically at higher temperatures. Virulence fluctuations due to temperature that were observed are similar to the range of temperatures that might be experienced by Ae. aegypti. The effect of wMelpop on Ae. aegypti life-span is more pronounced during summer months when temperatures increase. Consequently the infection of wMelpop on Ae. aegypti populations, decreases during cooler months.
Possible Environmental Consequences of Wolbachia in Vector-Mosquitoes
Wolbachia pipientis is a gram-negative bacterium belonging to α-proteobacteria. The infection of this intracellular bacterium is transmitted ovarially and forms various types of relationships with invertebrates. In most arthropods, Wolbachia has a facultative-mutualism role however in parasitic nematodes the relationship with Wolbachia is that of obligatory mutualism.

The Wolbachia pipientis bacterium is responsible for reproductive alterations in a wide range of invertebrate organisms. Different strains of Wolbachia induce different reproductive manipulation techniques. Some strains induce cytoplasmic incompatibility, other strains are male-killers while others cause thelytokous parthenogenesis and still other Wolbachia strains induce sperm competition. The reason for such disparity in reproductive manipulation across different Wolbachia strains is still unclear. These endosymbionts may be transmitted vertically from mother to egg cells and can also be transmitted horizontally among individuals across different species. Laboratory transinfection methods have been successfully developed to infect different invertebrates
The ecological benefits of artificially infecting vector organisms are vast. These pioneering techniques,if used, have the potential of wiping out vector-borne infectious diseases such as malaria, dengue fever, yellow fever and West Nile Virus. Wolbachia cuts the life span of these mosquitoes short, preventing the mosquitoes from hosting the viruses and protists that cause these diseases. As a biological weapon, Wolbachia may permanently reduce or remove these diseases that claim millions of life each year.
References
7. J. L., Rasgon. Insect Symbiosis: Wolbachia and Anopheles mosquitoes. (2008)]
22. Parthenogenesis
24. Mosquito
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2009, Kenyon College.
