Bacillus thuringiensis toxin in C. elegans
By Sarah Adrianowycz
Bacillus thuringiensis as an organism
Bacillus thuringiensis is prevalent in the soil, and is a gram –positive, spore-forming member of the Bacillus ceres group. The Bacillus ceres group is a collection of seven genetically similar species, three of which are so alike that they are occasionally considered the same organism, except for their vastly different pathogenic outcomes (Kho et al. 2011). It is these different pathogens, or external expression of the organism’s phenotype, which are often used by researchers to determine which species are distinct. In the case of B. thuringiensis the organism is typified by the production of specific invertebrate related pathogens (Ceuppens et al., 2013). However, the issue with using phenotypic differences to differentiate among these organisms is that the differences frequently stem from virulence factors contained on plasmids, which are extremely variable and transient. These genetic elements separate from the primary genome can be exchanged through horizontal gene transmission, lost in the environment, or selected against during culturing in the laboratory. Despite the issues with distinguishing strains based upon their ability to cause disease, B. thuringiensis is often distinguished by the ability to form crystal proteins. These products are insecticidal in nature and referred to as cry genes for the genes that code their expression (Ceuppens et al.). In the wild, most strains of B. thuringiensis are capable of producing anywhere from 1-4 types of cry proteins (Kho et al., 2011).The ability to produce multiple cry genes may be explained because of the synergistic effect that some of these proteins have in taking over a host when working together, or because different potential hosts have different levels of susceptibility to specific cry proteins (E. Schnephf, 1998).
It is the ability to produce these crystal proteins that gives B. thuringiensis value as an insecticide and has made it especially interesting to humans who are looking into biological applications of the cry proteins. These toxins have been utilized both in direct application on to crops and as a source of transgenic elements that provide resistance against the orders Lepidoptera, Diptera, and Coleoptera (E. Schnepf, 1998) when incorporated into the genomes of genetically modified organisms. The method of action within this subset of proteins makes them especially effective against insects but hasn’t produced evidence of negative effects against either humans or other higher trophic level organisms, as is typically the case for insecticides. Because of these perceived benefits and the lack of obvious repercussions, the utilization of plants with cry proteins has become common place since 1996, when genetically modified potatoes, corn, and potatoes with versions of cry genes were made commercially available (Schnepf).
Cry Genes in Bacillus thuringiensis
The genome of B. thuringiensis has between 2.4 – 5.7 million base pairs and is supplemented extensively by plasmids, both linear and circular in nature. The largest of these additional genetic elements are responsible for the encoding of the crystals released during sporulation (Schnepf).
Cry genes are expressed during the stationary phase of B. thuringiensis’s life cycle, and to such an extensive degree that 20-30% of the dry weight of the organism’s spore is made up of the resulting crystals. The regulation of crystal formation and its relationship with the stationary phase are monitored at three levels of regulation: transcriptional, posttranscriptional, and posttranslational, but not all cry genes are dependent upon the process of spore formation (Schnepf). The geometric structure of the resulting crystals differs based upon the type of cry protein being expressed, ranging from bipyramidal to spherical, although certain cry proteins require accessory proteins to assemble into their correct crystal structure (Schnepf).
The crystals produced by cry genes are made up of potential toxins that become active only when consumed by a susceptible organism. Susceptible organisms are those that have alkaline conditions in a portion of their digestive system, as is the case for the midgut of insects and the intestines of nematodes. Once the crystal has entered the alkali conditions, the toxin begins to work after being made soluble, cleaving by enzymes involved in protein catabolism, and then being inserted into the membrane of the epithelium where it is able to form pores (Schnepf). The success of various cry proteins can sometimes be explained by differences in solubility, and decreasing solubility has been suggested as a reason for increasing insect resistance to the toxin. Once processing is completed, cry proteins are capable of two main functions, the ability to bind to specific receptors on the target organism and to function as an ion channel. Two methods of pore function have been suggested, one where the pore allows non-specific ion transport and another where the controlled transport of specific ions results in undermining of the membrane potential that is necessary for the cell’s normal functioning (Schnepf). The reason that there is debate over the selectivity of the pore is because of the wide range of experimental conditions in which the cry proteins have been tested has resulted in conflicting results. But this inconsistency may be evidence of the pore responding to differing environmental conditions instead of improper scientific experimentation (Schnepf).
There have been recent attempts to systematically alter the structure of the cry proteins in order to improve their functioning from the point of view of those trying to increase the toxin’s insecticidal nature. Although decreasing solubility is the evolutionary means suggested by which insects decrease their susceptibility to the toxins, human efforts have been focused on increasing the amount of irreversible binding, a process typically associated with insertion of the toxin into the epithelial cells of the organism, in order to create more lethal varieties of cry proteins (Schnepf).
The Relationship between B. thuringiensis Cry Proteins and Nematodes
B. thuringiensis is widespread in the soil and allocates a large amount of energy and resources to the production of cry toxins that need to be consumed to be activated, despite the fact that most of the insects its cry proteins have been researched to be effective against spend minimal time feeding directly from the soil (Wei et al., 2003).
In contrast, nematodes, specifically Caenorhabditis elegans, inhabit the soil under typical conditions and are a compelling model organism because not only are they likely to encounter cry toxins and other pathogens in their natural habitat, but they represent one of the over 100,000 species of nematodes that directly consume soil and B. thuringiensis (Wei et al., 2003). This similarity of habitat and the frequent interaction among these organisms means C. elegans may be one of the intended targets for cry proteins. Nematodes have a number of responses to deal with environmental threats, among them immune responses, although these are costly for the organism. Another more rapid and less energetically and metabolically expensive option is supplementation by behavioral changes to adapt in a rapidly changing environment to newly encountered threats (Luo et al.,2013).
Although the impact of cry proteins has been widely studied in insects that harm agricultural products and directly influence humans, less research has been done on the nematode model. It is important to determine the impact of these toxins on C. elegans to answer the question of by what method these proteins are toxic, how the two organisms might have coevolved, and the potential for larger environmental impacts if this stable soil inhabitant is negatively impacted by something that has found widespread application in industrial agriculture (Wei et al., 2003). Because of the inclusion of cry genes within staple food crops, nematodes, including C. elegans have had increasing contact with this pathogen in what were previously stable environments that contained the cry proteins as a result of B. thuringiensis but at levels lower than currently.
Structure of Cry5B
There are over 200 types of cry proteins that have so far been identified, and they range in genetic similarity from less than 20% to more than 90% (Hui et al., 2012). However, Cry5B is the most intensively researched cry protein with negative impacts on nematodes, and this toxin functions through binding to specific glycolipid receptors in the worm’s intestines (Hui et al. 2012). Although the sequence similarity between Cry5B and the cry proteins that are most effective against insects is limited to approximately 20%, the proteins share a similar three component structure. The toxins that are effective in insects that have so far been sequenced have a high degree of structural similarity, but the average difference between Cry5B and this group is even greater than for the most dissimilar insecticidal toxin, Cry2Aa (Hui et al., 2012).
The first domain of Cry5B is believed to be the active mechanism for pore formation, and because of this vital functional role it has the largest degree of similarity to other cry proteins which are all believed to function through pore formation (Hui et al., 2012). Although there is only 22% sequence similarity, once the complex is activated by the proteases in the nematode intestine the activated toxin becomes similar to that of Cry4B (Hui et al., 2012). The first domain is made up of five helixes grouped together with one focal helix that serves as initiator for pore formation. The central helix has been identified in all cry proteins that have so far been structurally investigated (Hui et al., 2012).
Domain two is the most structurally independent of the three components of Cry5B, and has greater structural similarity to a banana lectin than to other cry proteins because of the homodimer structure. Because of this structural difference from other cry proteins but the similarity to a manose binding structure, Cry5B is able to accomplish glycan receptor binding that the other cry proteins are not (Hui et al., 2012).
Domain three is conserved among cry proteins and all of the cry proteins structurally analyzed show marked similarity to molecules capable of binding carbohydrates in this domain, among which some of the mechanisms are known and well studied (Hui et al., 2012).
Impact of Cry5B on Caenorhabditis elegans
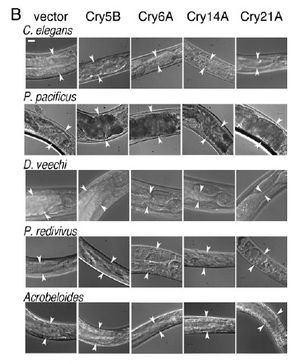
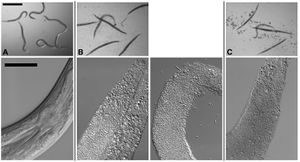
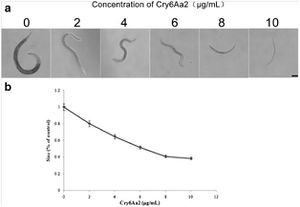
Cry5B, when produced by E. coli and fed directly to C. elegans results in the death of the nematodes after approximately six days at a lethal dose of approximately 8µg/ml because of cry protein intoxication, and no infection by the bacteria. In contrast, when Cry5B is provided for nematodes at a similar concentration but in the presence of B. thuringiensis death results in 24-48 hours as the bacteria take control, resulting in what the authors refer to as a “Bob” phenotype, or Bag of bacteria, where the internal organs of the nematode are digested and replaced by a combination of bacterial cells and spores surrounded by the cuticle of C. elegans (Kho et al., 2011). Bacterial infection requires both the presence of this pore forming protein (PFP) and the bacterial source, although it does not matter if the cry protein is produced by the bacteria or just available for uptake in the environment. C. elegans cultured in the presence of B. thuringiensis without Cry5B does not result in infection. The “Bob” phenotype can be seen in the accompanying image, Figure 1, as represented in panel B where the infected nematodes are completely linear, devoid of any internal structures, and lacking their typical color. (Kho et al., 2011).
A subsequent experiment determined that the Cry5B protein is only able to cause death in C. elegans when binding occurs between the toxin and a subset of receptors in the organism’s intestinal track. In mutant nematodes that were devoid of glycolipid receptors, the toxin was incapable of forming a pore, and as a result B. thuringiensis was unable to cause an infection, supporting the hypothesis that the establishment of the pore is a vital virulence factor that permits infection by B. thuringiensis (Kho et al., 2011).
Although C. elegans death is almost certain within 48 hours of exposure due to takeover by B. thuringiensis, it can take as few as fifteen minutes of exposure to the cry protein and the pathogenic bacteria for a high level of death to be observed, and there is no statistical difference in the percentage of C. elegans death for any time points tested between 15 minutes and 8 hours of exposure, even when organisms were subsequently washed of the excess cry protein and the bacteria (Kho et al., 2011).
Means of resistance to Cry5B in Caenorhabditis elegans
In order to understand the means through which some C. elegans individuals are able to remain resistant to cry proteins , Huffman et al. selected for mutants that were not killed in the presence of Cry5B and determined that surviving nematodes were defective in bre-1 through bre-5 functioning (2004). Bre (an abbreviation for Br-toxin resistant) mutants are unable to integrate the pore forming protein Cry5B into the epithelium of the intestine, possibly because the bre genes encode a receptor that permits the uptake of Cry5B, or possibly because the genes influence the level of binding that occurs in the nematode’s intestine. Either way, the result is limited binding of the protein and limited pore formation (Huffman et al., 2004).
