Bifidobacterium adolescentis: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{ | {{Curated}} | ||
===Classification=== | ===Classification=== | ||
Latest revision as of 17:54, 15 April 2011
Classification
Higher order taxa
Bacteria; Actinobacteria; Actinobacteridae; Bifidobacteriales; Bifidobacteriaceae; Bifidobacterium; Bifidobacterium adolescentis ATCC 15703
Genus
Bifidobacterium adolesecentis
Species
B. angulatum; B. animalis; B. asteroides; B. bifidum; B. boum; B. breve; B. catenulatum; B. choerinum; B. coryneforme; B. cuniculi; B. dentium; B. gallicum; B. gallinarum; B indicum; B. longum; B. magnum; B. merycicum; B. minimum; B. pseudocatenulatum; B. pseudolongum; B. psychraerophilum; B. pullorum; B. ruminantium; B. saeculare; B. scardovii; B. simiae; B. subtile; B. thermacidophilum; B. thermophilum; B. urinalis; B. sp.
Description and significance
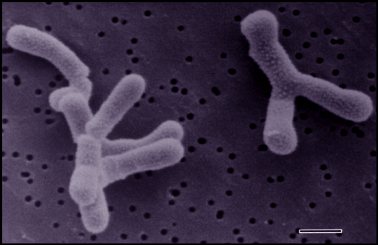
Bifidobacterium adolescentis are normal inhabitants of healthy human and animal intestinal tracts. Colonization of B. adolescentis in the gut occurs immediately after birth. Their population in the gut tends to maintain relative stability until late adulthood, where factors such as diet, stress, and antibiotics causes it to decline(2). This species was first isolated by Tissier in 1899 in the feces of breast-fed newborns. Tissier was the first to promote the therapeutic use of bifidobacteria for treating infant diarrhea by giving them large doses of bifidobacteria orally. Since then, their presence in the gut has been associated with a healthy microbiota (2,6). The correlation between the presence of bifidobacteria and gastrointestinal health has produced numerous studies focusing on gastrointestinal ecology and the health-promoting aspects that bifidobacteria are involved in. Obtaining more information about specific strains of bifidobacteria and their roles in the gastrointestinal tract have been on the rise as these probiotic organisms are being used as food additives, such as dairy products. Their name is derived from the observation that these bacteria often exist in a Y-shaped, or bifid form (2,4).
There are currently 29 species of Bifidobacteria that have been described; Five of these, including B. adolescentis, have interested dairy manufacturers in producing "therapeutic fermented milk products" (2)due to their higher survival rates in acidic products(1). Thus, along with other beneficial intestinal bacteria such as Lactobacillus acidophilus, Lactobacillus casei, Streptococcus thermophilus, and Enterococcus faecium, bifidobacteria have been added into dairy products, most notably yogurt and milk. These "probiotics" (literally, "pro life") make great residents in dairy products because dairy products need to be refrigerated and have a short shelf life, in addition to being a commonly consumed source of nutrients, enabling consumers to reap the many healthy benefits these bacteria provide in improving gastrointestinal health (6,11).
Genome structure
The genome of Bifidobacterium adolescentis averages 2.1 Mbp in length. Its form is either an elongated, thin irregular rod-, Y- or V-shape. B. adolescentiscontains one circular chromosome. B. adolescentis has 2,089,645 nucleotides, including 1,631 protein-encoding genes and 69 RNA genes (2). G-C content ranges from 42-67% (3), with an average range around 60% (2). To date, no plasmids have been detected in B. adolescentis(7), though contrasting experiments report B. adolescentis plasmid extraction for transinfection during experiments(10).
Cell structure and metabolism
B. adolescentis is a gram-positive organism, containing one cell membrane, and is not mobile (2,4). Each species of bifidobacteria contain different components in their cell walls; B. adolescentis' cell wall is made made primarily of murein, containing Lys- or Orn-D-Asp within its peptide chains. Its polysaccharide components include glucose and galactose. Myristic, palmitic, and oleic are the major fatty acids within the cell wall. Leipoteichoic acids on the cell wall's surface function to help the organism adhere to the intestinal wall (1). Bifidobacteria are anaerobes (though some can tolerate oxygen, using the enzymes superoxide dismutase and catalase in their defense against the toxic effects of superoxide and hydrogen peroxide)(1). The main sources of carbon used for energy by bifidobacteria are simple sugars which are readily used by the upper intestinal tract, as well as complex carbohydrates such as starches, cellulose, hemicellulose, xylan, pectins and gums that are not digested by the human gut (6).B. adolescentis, like all of the Bifidobacteria species, can ferment lactose and grow well in milk, as well as use many carbohydrates. Glucose is fermented using the fructose-6-phosphate pathway which requires the enzyme, fructose-6-phosphoketolase(F6PPK)(1). As a result of metabolizing various carbohydrates, bifidobacteria produce short-chain fatty acids such as propionate, butyrate, and acetate to be used as energy sources (1).
Nitrogen metabolism is also observed in bifidobacteria, using ammonium sulfate as its nitrogen source. As a result, bifidobacteria's ability to use ammonia as a nitrogen source may decrease that amount of ammonia in the colon(1).
Bifidobacteria have also been found to synthesize vitamins; B. adolescentispredominantly synthesizes cyanocobalamin and nicotine, as well as thiamin, folic acid, and pyridoxine. B. adolescentis' ability to produce vitamins plays a beneficial role in increasing the nutritional quality of fermented dairy products to which it is added (1,8).
Ecology
Bifidobacteria colonize and reside in the gastrointestinal tracts of humans as well as most mammals. Before birth, human fetuses are microbe-free, and thus do not contain in bacteria within their digestive tracts. However, almost immediately after birth, bacteria begin to colonize in the newborn's intestinal tract, forming the intestinal microflora (1,2,11). While various bacteria make up the microflora, bifidobacteria predominate as the main bacteria during the neonatal period of development, especially in infants that are breast-fed. Bifidobacteria's predominance in the intestinal tract has been confirmed by its high percentage (96%) of the bacterial content within infant fecal matter samples.(11)
As a resident within the human gut, bifidobacteria create a bacteria-host, symbiotic-like relationship, providing the human host with many health benefits(1,4,6). Healthful contributions by bifidobactia include improved intestinal functioning, by increasing the absorption of human milk protein by removing the casein in human milk. Improved amino acid metabolism is seen in bifidobacteria's production of various B vitamins. In infants, bifidobacteria play a significant role in preventing the loss of nutrients, by suppressing the growth of competing bacteria in the intestinal tract. Bifidobactia prevent constipation in the host by producing acids that stimulate peristalsis and promote normal bowel movements. This contribution is reinforced by the availability of Bifidus preparations for constipation sufferers, which work by moistening fecal material. Antibiotic activity is also carried by bifidobacteria, working against pathogens such as E.coli, Staphylococcus aureus, Shigella dysenteriae, Salmonella typhi, Proteus ssp., and Candida albicans by suppressing their growth in the host organism. Bifidobacteria also inhibit the growth of these enteric pathogens by breaking down conjugated bile acids into free bile acids(1).
In treating against liver damage, bifidobacteria (especially B. bifidum) and lactulose are added into the host's diet to re-establish normal intestinal microflora in in the advanced stages of cirrhosis. Rebalancing the intestinal microflora thus reduces concentration levels of ammonia and free phenols in the blood. Protein metabolism is also increased as bifidobacteria re-normalize intestinal function(1).
In the prevention against the initiation against colon cancer, bifidobacteria play a significant role. Recent studies have shown that bifidobacteria reduce the levels of various colonic enzymes which are used to convert procarcinogenic molecules into carcinogens (such as nitrosamines and secondary bile salts). Other studies have shown that the increased consumption of fermented milks (those containing bifidobacteria and other probiotics) decreases the risk of colon cancer(1).
Bifidobacteria have also been found to aid in antitumor activity in the host by stimulating the host's immune response upon injection into the tumorous region. Upon injection, bifidobacteria (especially B. adolescentis) help to inhibit tumor growth by delivering anticancer genes to the tumor area and stopping angiogenesis(1,10).
In regards to the host's immune system, bifidobacteria have been shown to stimulate the enlargement of lymph nodes in the spleen and increase the number of B lymph cells that are involved with humoral immunity and T lymph cells that are associated with cell-mediated immunity(1).
Lastly, bifidobacteria contribute to the host's gastrointestinal health by lowering cholesterol levels in the blood. A factor known as the hypocholesterolemic factor (which is present in fermented milks) has been found and is suggested to be what inhibits cholesterol synthesis in the body. However, more consistent research about this health contribution is still in the process(1).
Pathology
Bifidobacterium adolescentis is a non-pathogenic organism (1).
Application to Biotechnology
B. adolescentis synthesizes various B vitamins that are beneficial to the nutritional health of the host organism. Vitamins that are synthesized included thiamin(B1), pyridoxine(B6), folic acid(B9), nicotine, cyanocobalamin(B12), ascorbic acid(Vitamin C), biotin, and riboflavin (4).
Also, bifidobacteria contain phytase activity, enabling the dephosphorylation of phytic acid (myo-inositol hexaphosphate, IP6) and produce several myo-inositol phosphate intermediates, IP3, IP4, and IP5. IP6 has been shown to have antinutritional effects by limiting the dietary bioavailability of amino acids and minerals such as Ca2+, Z2+, and Fe2+. However, bifidobacteria reduce these effects by dephosphorylating IP6 into less phosphorylated products during food processing and gastrointestinal transit. Additionally, these less phosphorylated intermediates are also involved in regulating vital cellular functions (5).
In the process of manufacturing fermented milk products, new methods have been developed to preserve the bacteria during the manufacturing. To avoid problems during the de-aerating step of processing fermented milks, bifidobacteria are mixed with Streptococcus thermophilus, which has a high oxygen-utilizing ability(1). To keep oxygen permeability low, glass bottles or aluminum-laminated packages are used to distribute the product (1).
In the field of genetic engineering, some species of bifidobacteria have plasmids and have been cloned into E. coli vectors together with an antibiotic resistance marker and have resulted in the replication of bifidobacteria and E. coli by the generations of shuttle vectors(6).
[edit]
Current Research
1. Bifidobacterium adolescentis as a producer of folate in the colon: testing various strains of Bifidobacterium revealed their ability to produce folate. Experiments analyzing cultured samples of feces showed that the addition of B. adolescentis may increase the folate concentration in a colonic environment. Results provided positive insight into the use of probiotics in preventing folate deficiency in colonic epithelial cells as well as more efficiently protecting the colon against inflammation and cancer(8).
2. Dietary Factors in inflammatory disease: While the pathogenesis of inflammatory bowel diseases is not completely understood, it is a wide suspicion that the causes of ulcerative colitis and Crohn's disease are related to diet and dietary factors. Experiments and tests have shown that probiotic therapy may be a significant intervening factor in the process of inflammatory bowel diseases. However, due to the incomplete understanding about the role of bifidobacteria and other known probiotics on the gastrointestinal tract, more research is underway to determine whether probiotic therapy will become more than just an alternative approach to treating inflammatory bowel diseases(9).
3. B. adolescentis used as a transporter of anticancer genes into tumors: In experiments for gene cancer therapy, B. adolescentis was selected for use to deliver endostatin gene to solid tumors in tumor-bearing mice. B. adolescentis was chosen as the delivery vector because it is a non-pathogenic bacteria found in the human body, and thus does not produce endotoxins or toxins; it increases the immune response in the host and inhibits the growth of tumors in other various cancers; it can be easily killed by antibiotics or in the presence of oxygen.
B. adolescentis with the endostatin gene was injected into the mice through the tail veins, and upon analysis of the mice' tissues, B. adolescentis was only found in the tumor and not in any of the non-affected tissues. Inhibition of angiogenesis in the local tumor areas was also observed, thus preventing any tumor growth. This observation revealed that B. adolescentis selectively germinated and proliferated in the tumor tissue effectively delivering the endostatin gene. Since tumor growth is dependent on angiogenesis, the fact that B. adolescentis inhibited angiogenesis, and thus suppress tumor growth makes it a suitable and specific transporter for the endostatin gene in tumor tissue as a means of gene cancer therapy.(10)
References
1.[Arunachalam, Kantha D., et al. The Role of Bifidobacteria in Nutrition, Medicine, and Technology. Nutrition Research. Vol 19, No. 10, pp. 1559-1597, 1999. Elsevier Science Inc., USA.]
3.[Biavati, B. and Mattarelli, P. (2001) The family Bifidobacteriaceae. In: The Prokaryotes (Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.H. and Stackebrandt, E., Eds.), pp. 1–70. Springer, New York.]
4.Bifidobacterium (Wikipedia)
7.[http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1352255 Lee, Ju-Hoon, and Sullivan, Daniel. Sequence Analysis of Two Cryptic Plasmids from Bifidobacterium longum DJO10A and Construction of a Shuttle Cloning Vector. Appl Environ Microbiol. 2006 January; 72(1): 527–535. Copyright © 2006, American Society for Microbiology]
10. Xi Li, et al. Bifidobacterium adolescentis as a delivery system of endostatin for cancer gene therapy: Selective inhibitor of angiogenesis and hypoxic tumor growth. Cancer Gene Therapy. February 2003, Volume 10, Number 2, Pages 105-111. 2003 Nature Publishing Group.
11.[Yaeshima T., et al. Bifidobacteria: their significance in human intestinal health. Malaysian Journal of Nutrition 3: 149-159, 1997. Morinaga Milk Industry Co. Ltd., Nutritional Science Laboratory, Japan.]
Edited by student of Rachel Larsen and Kit Pogliano
