Clostridium difficile infection and fecal bacteriotherapy: Difference between revisions
No edit summary |
|||
| Line 1: | Line 1: | ||
{{Uncurated}} | |||
==Introduction: History and Significance== | ==Introduction: History and Significance== | ||
<br>By Rebecca Varnell<br> | <br>By Rebecca Varnell<br> | ||
Latest revision as of 18:44, 29 September 2015
Introduction: History and Significance
By Rebecca Varnell
Clostridium difficile is a Gram-positive, spore-forming Firmicute found abundantly in nature (Figure 1). The rod-shaped bacteria were first noticed and described by Hall and O’Toole in 1935 for their notable abundance in the intestines of infants (Poxton 2005). C. difficile can and often do live among the diverse microbiota of a healthy human and animal gut. They are found abundantly in soil as well as human and animal feces. However, in the absence of microbial competition,C. difficile can proliferate. This occurs in the intestines individuals whose intestinal microbial communities are compromised, such as in patients treated with heavy antibiotics. The bacterial lifecycle includes the formation of dormant spores, which can be passed from individuals infected with or carrying C. difficile by fecal-oral route. Spores released in human or animal feces can live on surfaces in hospitals or nursing homes then picked up and ingested by a healthy individual. Dormant spores are known for their resilience and longevity; spores can live on surfaces for many months despite heavy cleaning (Patel 2010). The presence and spread of C. difficile in hospitals and nursing homes is a major concern for doctors and patients alike.
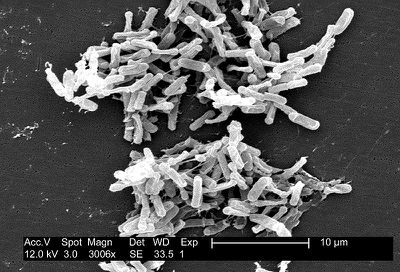
In 1978, researchers first isolated C. difficile from the stools of individuals suffering from antibiotic-associated pseudomembranous colitis (PMC), a severe inflammation of the colon exhibited by individuals treated with heavy courses of antibiotics (Patel 2010). In the following decades, a series of studies investigated these bacteria. Since a prominent study in 1990 by Borriello et al. articulated the pathogenesis and urgency of C. difficile, these bacteria have been studied extensively (Poxton 2005).
C. difficile infection (CDI) is the umbrella term that refers to colonization of C. difficile in a human host. CDI can present itself clinically in a diversity of ways, most notably antibiotic-associated diarrhea (AAD) and antibiotic-associated colitis (AAC) (Farooq et al. 2015). CDI is a major issue in the United States; it is one of the most common health-care associated infections in the US and most commonly affects hospitalized patients and individuals in nursing homes. Major CDI outbreaks in hospitals have caused healthcare providers to reevaluate liberal use of antibiotic treatment. Alternative treatment methods such as fecal microbiota therapy (FMT) that avoid the continued use of antibiotics may be the most sustainable, effective, and cost-efficient treatment for CDI (Boyle et al. 2015).
Life cycle and sporulation
Though little research into the mechanisms of C. difficile metabolism exists, it is a known obligate anaerobe, and coupled amino acid fermentation is its primary source of ATP (Bouillaut 2012). Because C. difficile cannot metabolize in the presence of oxygen, the bacteria produce metabolically dormant spores that transfers infection between patients in a hospital. The dormant spore is produced through the sporulation pathway initiated during infection. Certain strains incapable of producing dormant spores cannot be transferred between patients and thus remain confined to the gastrointestinal tract of the host. Spores, for the strains that can produce them, are resistant to antibiotics and immune responses from the host. Remarkably, the spores are also resistant to disinfectants commonly used in hospitals. This unique feature explains the rapid spread of CDI in healthcare facilities (Burns and Minton 2011).
The lack of homology between C. difficile spores and other spore-forming bacteria such as Bacillus subtilis profoundly limits scientists’ ability to understand the specific structural nuances that confer the bacteria’s incredible resilience. However, research related to the spore structure of C. difficile spores indicates that the structure can be divided into compartments; the innermost compartment is the spore core. The spore core contains spore DNA, RNA, and most enzymes (Paredes-Sabja et al. 2014). This core has an incredibly low water content, which contributes to its resiliency. A compressed inner membrane protein with low permeability surrounds the core. A germ cell wall, cortex, outer membrane, coat, and exosporium all surround the inner membrane. The many layers protect the spore DNA from harsh conditions (Figure 2).
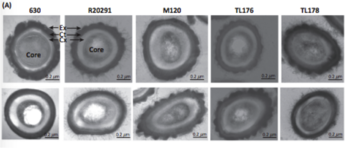
The formation of spores is essential to C. difficile spread between human hosts. Sporulation is induced by lack of environmental nutrients and consequently, cellular starvation (Weston 2008). A genome-wide approach conducted by Pettit et al. (2013) identified SpoOA as a positive regulator of sporulation. SpoOA encodes a transcriptional regulator, which dictates colonization and transmission. The regulator binds upstream of sporulation genes and is also associated with controlling toxin gene expression (Pettit et al. 2013). Thus, the presence of SPoOA in C. difficile is essential in its pathogenicity.
When sporulation is signaled, the cell stops growing and begins growing the spore (Paredes-Sabja et al. 2014). The cell divides asymmetrically, producing a forespore and larger mother cell. The mother cell produces the cortex as well as the inner and outer coats for the forespore. When the spore is mature, the mother lyses and releases the spore. When appropriate environmental conditions are met, an unknown signaling mechanism causes the spore to germinate. Germination is an essential feature of CDI progression, as the vegetative cells proliferate within the host to cause infection (Howerton et al. 2010).
The ingestion of spores does not necessarily lead to colonization and thus, infection. Certain environmental conditions must be met in order for germination and colonization to occur. Recent studies indicate the presence of primary bile acids such as taurocholate is necessary for the germination of C. difficile. Secondary bile acids, on the other hand, such as chenodeoxycholate, can inhibit the germination of the spores, thus preventing infection (Seekatz and Young 2014).
Structure and Pathogenicity
The toxins produced by certain strains of C. difficile during the stationary phase of vegetative growth bind to and colonize the gastrointestinal tract, causing the symptoms associated with CDI: diarrhea, inflammation, and colonic damage (Westin 2008). Strains without toxin-producing capabilities are nonpathogenic. The emergence of a virulent strain that produces both toxins was detected following severe outbreaks of CDI in 2000 and 2003. The strain is referred to as North American Pulsed Field type 1 (NAP1) and is still believed to be the primary cause of CDI (Kelly and LaMont 2008).
Conflicting studies have named toxin A and toxin B as the essential virulence factor of C. difficile, leading healthcare providers to prefer diagnostic testing that can identify the presence of either toxin (Farooq et al. 2015). Many virulent strains produce both exotoxin A and B. These toxins belong to the family of clostridial glucosylating toxins, which are named for their dependency on glucosyltransferase activity to confer toxicity (Kelly and LaMont 2008). Toxin A is associated with increased intestinal permeability and fluid secretion. Toxin B leads to colonic inflammation. Together, the two toxins cause the uncomfortable and dangerous symptoms of CDI. Based on the conserved N-terminal domain in the two genes that code for Toxin A and B, researchers hypothesize a gene duplication event (Voth and Ballard 2005).
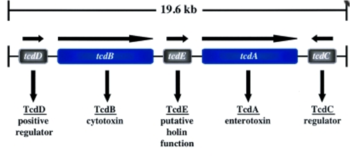
Five genes are encoded on the pathogenicity locus in total: two toxin genes and three regulatory genes (Figure 3). One regulatory gene, tdcC, codes for the protein TcdC, which inhibits transcription during the exponential growth phase of the bacteria. In patients affected by major outbreaks of C. difficile infection, deletion mutations in this gene significantly increased toxin production. Individuals infected by this mutant showed tremendously high toxin levels and correspondingly suffered acute symptoms of CDI. The gene tcdD codes for TcdD, which enhances the expression of the two genes coding for toxins A and B (Voth and Ballard 2005). The gene that encodes TcdE is positioned between tcdB and tcB. This protein is thought to be a positive regulator for its neighbor genes that code for toxins A and B. TcdE is also thought to allow for the release of toxin A and B by permeabilizing the C. difficile cell wall. The coordinate actions of these three regulatory gene products act to regulate toxin production and allow for its release out of the cell.
Toxin A and B show homology as they both feature a C-terminal receptor-binding domain and a central hydrophobic domain. The central domain allows the toxins to insert into the membrane of the endosome of a eukaryotic host cell (Jank et al. 2006). In both toxins, the TcdB binding domain interacts with cell-surface receptors, which induces endocytosis. The toxin undergoes a transformational change in the presence of a low pH environment within the cytosol, which allows it to disrupt the Rho GTPases through glucosylation (Jank et al. 2006). These intracellular changes disrupt the cytoskeleton and eventually lead to cell death, which causes damage to the mucosal epithelium and induction of an inflammatory response (Seekatz and Young 2014). The tight junctions between cells are also disrupted, allowing an inflow of neutrophils, which add to colonic inflammation.
In addition to toxins A and B, a third toxin of interest is C. difficile binary toxin (CDT), which can disrupt the cytoskeleton and possibly increase the strain virulence (Seekatz and Young 2014). The exact relationship between CDT and CDI continues to be investigated.
Interaction with Microbiota
The microbial composition of the human gut is well studied. Costello et al. (2012) advocates approaching the microbial community in the human gut from an ecological framework. The human body, understood in these terms, is a microbiome in which immense symbiotic interactions take place. The maintenance and balance of these interactions is crucial to human health. The microbial community living in the human gastrointestinal tract enables humans to derive energy from dietary substrates, promotes the differentiation of host tissues, stimulates the immune system, and protects the host from invading pathogens (Costello et al. 2012). Myriad conditions linked to disruption in the microbial community evidences its importance to human health. For example, Turnbaugh et al. (2006) linked obesity to the relative abundance of Bacteroidetes and Firmicutes. Though the development of the microbial community in humans is still poorly understood, it is acknowledged that individuals have idiosyncratic communities influenced by a range of genetic and environmental factors (Palmer et al. 2007).
A healthy adult colon can contain up to 1012 bacteria per gram of contents (Owens et al. 2008). The presence of diverse and abundant microbes in the human gut protects the host from overgrowth and colonization of certain bacteria. Competition for nutrients ensures that no particular bacterial strain dominates the community. A decline in microbial diversity increases an individuals’ susceptibility to CDI, which explains higher instances of CDI in individuals over 65 since elderly people typically have less diverse microbial communities (Seekatz and Young 2014). Antimicrobial therapy also reduces bacterial abundance and diversity; it may take a healthy adult up to two weeks to restore indigenous flora levels after antibiotic use. Thus, individuals taking antibiotics are commonly regarded as the population most susceptible to CDI. Other vulnerable populations include post-partum women, immunocompromised children, or individuals taking proton pump inhibitors.
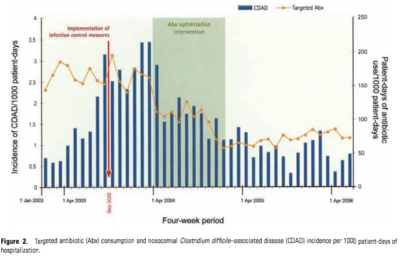
A study conducted in Quebec by Valiquette et al. (2007) following major outbreaks associated with the hypervirulent NAP1/027 strain indicated that restricting antibiotic usage in hospitals markedly declined the instance of CDI. The study measured the rate of CDI (referred to as CDAD - C. difficile associated disease) per 1000 patient-days of hospitalization as it relates to the rate of targeted antibiotic (Abx) use. Following the implementation of restrictive antibiotic use policies, the rates of CDI/CDAD decreased by 60% (Figure 4).
With increased understanding of the link between antibiotic use and instance of CDI, researchers have investigated which specific antibiotics are more strongly associated with CDI. A study conducted by Owens et al. (2008) describes data from an experiment on mice demonstrating that three categories of antibiotics that affected the risk of CDI. Antibiotics such as ceftriaxone disrupt the intestinal flora but are not active against C. difficile. These antibiotics are the most likely to create host conditions amenable to CDI. Other antimicrobials that inhibit C. difficile growth such as oral vancomycin may prevent CDI during antibiotic treatment. Despite this, following the cessation of these antimicrobials, patients may still be at risk for developing CDI if they are exposed to C. difficile during recovery. Lastly, antimicrobials that only cause minimal disruption of the anaerobic microflora such as aztreonam do not promote CDI (Owens et al. 2008). This indicates that maintaining indigenous levels of microbial abundance and diversity is critical in protecting against CDI. Patients face the highest risk of developing CDI if C. difficile is resistant to the microbial and the microbial is very disruptive to microbial flora.
The risk of developing CDI is associated with specific antimicrobials that meet the aforementioned conditions. When C. difficile is resistant and a certain antibiotic and that same antibiotic is very disruptive to microbial flora, the risk of CDI is very high. Because of these factors, Clindamycin, an antimicrobial used for treating infection with anaerobic organisms, is highly associated with CDI (Johnson et al. 1999). The predominant strain in the outbreaks in the early 1990’s showed a 100% resistance to clindamycin (Samore et al. 1997). Additionally, clindamycin has a profound effect on the indigenous microbial community of a patient, creating optimal conditions for CDI to develop. Hospitals heavily curbed their use of clindamycin following these outbreaks of CDI, which resulted in the decline of clindamycin-resistant strains. A similar phenomenon occurred in the late 1990s with cephalosporins, which were heavily used in hospitals. A notable instance of cephalosporin-resistant CDI outbreak was the Veterans Administration Hopsital in New York in 1994 (Johnson et al. 1999).
CDI outbreaks have most recently been attributed to fluoroquinolones, which were formerly thought to be effective against C. difficile (Kallen et al. 2009). The emergence of flouroquinolone-resistant strains has led many hospitals to limit their use of this formerly common class of antimicrobials.
Though antimicrobial use is most commonly associated with CDI, this is not always the case. Recent studies indicate the presence of CDI outside of hospitals and in individuals not recently treated with antibiotics. A study by Eyre et al. (2012) utilized whole-genome sequencing to track the exposure to C. difficile noted that one third of CDI cases occur outside of the hospital. Further, C. difficile has been detected in soil, water, and domesticated animals. Other studies indicate that infant microbial communities may contain a high amount of C. difficile though they do not frequently contract CDI. The mechanism behind this phenomenon is still unclear; it has been postulated that infants lack the receptor necessary for CDI to progress. Others theorize that compounds in human breast milk prevent toxin binding (Rolf and Song 1994).
Clinical Presentation and Treatment
As of 2014, 15% of patients treated with antibiotics in hospitals developed antibiotic-associated diarrhea and 20-30% of those cases were caused by C. difficile (Paredes-Sabja et al. 2014). CDI typically manifests as diarrhea with colitis, which is characterized by frequent diarrhea, abdominal pain, fever, and leukocytosis (LaMont et al. 2015). The influx of neutrophils into the mucosa of the colon and the continued activation of the host immune system can lead to the formation of pseudomembranes, which often causes the condition pseudomembranous colitis (PMC). Though PMC is associated with CDI, not all patients with CDI suffer from this condition (Farroq et al. 2015). Pseudomembranes form when endothelial damage causes small areas of necrosis, which present as yellow-white plaques on the mucosal surfaces of the colon (Figure 5).
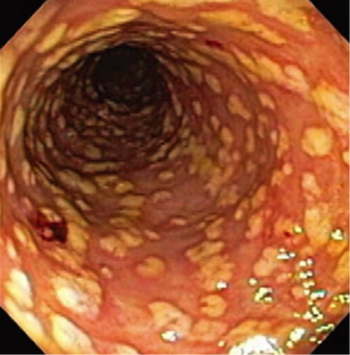
Fulminant colitis occurs in severe cases of CDI and is incredibly dangerous for patients. Toxic megacolon and bowel perforation are associated with fulminant colitis and can lead to sepsis (LaMont et al. 2015).
A high proportion of adults in hospitals of nursing homes are believed to be asymptomatic carriers (Carroll and Bartlett 2011). The immune system of a host and the microbial diversity of the gut is believed to determine the likelihood of infection for an individual carrying C. difficle. The prevalence of C. difficile in healthy adult gastrointestinal tracts have led researchers to classify this bacteria as an opportunistic pathogen only affecting individuals with compromised microbial diversity (Patel 2010).
CDI is diagnosed by the symptomatic presentation of severe diarrhea and a stool test positive for C. difficile toxins or a toxigenic strain. PCR testing is the preferred test for detecting the presence of C. difficile because of its high sensitivity and speediness (Kelly and Lamont 2008). Endoscopy is used in clinical settings when immediate diagnosis is required or antibiotic therapy has repeatedly failed. The ability to see pseudomembranes in the colon almost always indicates infection. Because of the risk of perforation in individuals with compromised colonic health, endoscopy is not the preferred method of diagnosis for C. difficile (Carroll and Bartlett 2011).
The standard therapy for individuals with CDI is metronidazole and/or vancomycin (or fidaxomicin) treatment. These are all known antibiotics that effectively target C. difficile. However, the rate of recurrence of CDI is relatively high (30-65%) after these antibiotic treatments (Bakken et al. 2011). The recurrence of CDI after treatment is incredibly dangerous, as it increases the risk of the emergence of resistant strains. The high proportion of C. difficile genome (11%) that consists of mobile genetic elements such as conjugative transposons allows the bacteria to quickly acquire antibiotic resistance (Sebaihia et al. 2006). Recurrent CDI (RCDI) has become increasingly more common with the emergence of a new hypervirulent strain that is less resistant to antibiotic therapy (Boyle et al. 2015). RCDI is incredibly dangerous and is the most common cause of death among patients with CDI.
Fecal microbiota therapy/transplant (FMT) is emerging as the most effective treatment for CDI. FMT is the infusion of a fecal suspension from a healthy donor into the gastrointestinal tract of a patient suffering from CDI. Though FMT is only recently being accepted as a viable treatment for CDI, physicians in the 17th century are reported performing FMTs on animals (Bakken et al. 2013). The first recorded human FMT was performed in 1958 via enema .The traditional method of FMT via enema is now largely obsolete. Currently, FMT can be performed via fecal infusion through nasogastric tube, colonoscopy, or self-administered enemas (Brandt et al. 2012).
Recent studies into the efficacy and safety of FMT are incredibly promising. A study performed in 2012 with individuals suffering from RCDI who had been previously treated by antimicrobials by Brandt et al. (2012) indicated positive long-term results. The cure rate within 90 days of initial FMT was 92%. Those with persistent CDI were treated again with FMT; the secondary cure rate was 98%. More recently, the Mayo Clinic’s review of 31 patients treated with FMT reported a 97% rate of substantial improvement among patients suffering from CDI. The study observed no negative effects of FMT and deemed it a safe, viable, and cost-effective treatment of CDI (Patel 2013).
The mechanism of FMT is relatively simple. The compromised microbial community of an individual suffering from CDI is supplemented by the microbial community of a donor who is typically a healthy family member or close friend. With increased microbial diversity, C. difficile growth is limited and normal microbial conditions are restored. The transmission of bacterial pathogens through FMT is a concern to researchers, though no instances of transmission have been reported (Brandt et al. 2012). A thorough screening process protects patients from a range of transmissible diseases.
Despite the high cure rates of FMT, patients remain resistant to the treatment. FMT remains a last-resort treatment option in many hospitals and patients are hesitant towards the treatment. Some researchers remain skeptical of possible side effects of FMT, such as the development of new disorders as a result of an altered microbial gut community (Boyle et al. 2015). Future studies confirming the efficacy and safety of FMT may increase the amount of patients suffering from CDI who are willing to undergo this cost-effective treatment.
Conclusion
C. difficile infection is relevant to current medicine and incredibly dangerous. This spore-forming bacteria's ability to withstand harsh conditions including bleach cleaning is incredibly concerning for the medical community. As the bacteria continue to be passed among patients in hospitals and individuals in nursing homes, they mutate into hypervirulent strains. Massive outbreaks of CDI evidence the danger inherent in the spread of C. difficile. This infection challenges researchers and physicians to rethink their conception of the human microbial community. Novel research indicates an increased emphasis on the benefits of a diverse microbiota and the deleterious effects of a compromised microbial community. While in the past, many physicians indiscriminately prescribed antibiotics, current research into the potential harm of antibiotic overuse is curtailing this practice.
While antibiotics are an indispensable asset to medicine, they are not without consequences. The movement away from liberal antibiotic use in hospitals could indicate a general shift in medicine towards holistic patient care as opposed to the isolated treatment of individual symptoms. Much of the literature dedicated to C. difficile infection management is spawning a new movement to view the human body as a dynamic and sensitive microbiome. The implications for this changing perception of the human body are immense. Experimental treatment methods such as fecal bacteriotherapy evidence the drive for new methods of care that push the boundaries of traditional medicine. As CDI increasingly becomes a public health crisis in developed countries, more treatments such as FMT that avoid antibiotic therapy will likely arise.
References
Johan S. Bakken et al. “Treating Clostridium difficile infection with Fecal microbiota transplantation,“ Clinical Gastroenterology and Hepatology 9 (2011): 1044-1049, accessed April 19, 2015, doi:10.1016/j.cgh.2011.08.014.
Laurent Bouillaut et al. “Proline-Dependent regulation of Clostridium difficile Strickland Metabolism,” Journal of Bacteriology 195 (2013): 844-854, accessed April 10, 2015, doi: 10.1128/JB.01492-12.
Miriam L Boyle et al. “Fecal Microbiota Transplant to Treat Recurrent Clostridium difficile Infections,” Critical Care Nurse 35 (2015): 51-64, accessed April14, 2015, doi: 10.4037/ccn2015356.
Lawrence J Brandt et al. “Long-term Follow-up of Clonoscopic Fecal Microbiota Transplant for Recurrent Clostridium difficile Infection,” The American Journal of Gastroenterology 107 (2012): 1079-1087, accessed April 16, 2015, doi: 10.1038/ajg.2012.60.
David Burns and Nigel Minton. “Sporulation Studies in Clostridium difficile,” Journal of Microbiological Methods 87 (2011): 133-138, accessed April 19, 2015, doi:10.1016/j.mimet.2011.07.01.
Andres F Carrion et al. “Severe colitis associated with docetaxel use: a report of four cases,” World Journal of Gastrointestinal Oncology, 10 (2010): 390-394, accessed April 17, 2015, doi: 10.4251/wjgo.v2.i10.390.
KC Carroll and JG Bartlett, “Biology of Clostridium difficile: implications for epidemiology and diagnosis, Annual Review of Microbiology 65 (2011): 501-521, accessed April 16, 2015, doi: 10.1146/annurev-micro-090110-102824.
Elizabeth K. Costello et al. “The Application of Ecological Theory Toward an Understanding of the Human Microbiome,” Science Magazine, 226 (6086): 1255-1262, accessed April 16, 2015, doi:10.1126/science.1224203.
David W Eyre et al. “A pilot study of rapid benchtop sequencing of Staphylococcus aureus and Clostridium difficile for outbreak detection and surveillance,” BMJ Open 2 (2012), accessed April 19, 2015, doi:10.1136/bmjopen-2012-001124.
Priya D. Farooq et al. “Pseudomemranous colitis,” Disease-a-month 61 (2015): 181-206, accessed April 14, 2015, doi: 10.1016/j.disamonth.2015.01.006.
Amber Howerton et al. “Mapping Interactions between Germinants and Clostridium difficile Spores,” Journal of Bacteriology 193 (2010): 274-282, accessed April 19, 2015, doi:10.1128/JB.00980-10.
Thomas Jank et al. “Rho-glucosylating Clostridium difficile toxins A and B: new insights into structure and function,” Glycobiology 17 (2007): 15R-22R, accessed April 18, 2015. doi:10.1093/glycob/cwm004.
Stuart Johnson et al. “Epidemics of Diarrhea Caused by a Clindamycin-Resistant Strain of Clostridium difficile in Four Hospitals,” The New England Journal of Medicine, 341 (1999): 1645-1651, accessed April 10, 2015, doi:: 10.1056/NEJM199911253412203.
Alexander J. Kallen et al. “Complete Restriction of Fluoroquinolone Use to Control an Outbreak of Clostridium difficile infection at a Community Hospital,” Infection Control and Hospital Epidemiology 30(3): 264-272, date accessed April 15, 2015, doi: http://dx.doi.org/10.1086/598167
Ciarán P. Kelly and J. Thomas LaMont. “Clostridium difficile – More Difficult Than Ever,” The New England Journal of Medicine 359 (2015): 1932-40, accessed April 15, 2015, DOI: 10.1056/NEJMra0707500.
J Thomas LaMont, “Clostridium difficile infection in adults: clinical manifestations and diagnosis,” UpToDate (2015), accessed April 19, 2015 .
Robert C. Owens et al. “Antimicrobial-Associated Risk Factors for Clostridium difficile Infection,” Oxford Journals: Clinical Infectious Diseases 46 (2008): S19-S31, accessed April 19, 2015. doi: 10.1086/521859.
Chana Palmer et al. “Development of the Human Infant Intestinal Microbiota” PLoS Biology 177 (2007), accessed April 16, 2015, doi:10.1371/journal.pbio.0050177.
Laura J Pettit et al. “Functional genomics reveals that Clostridium difficile SpoOA coordinates sporulation, virulence, and metabolism,” BMC Genomics 14 (2014): 160, accessed April 9, 2015, doi: 10.1186/1471-2164-15-160.
Nipa A Patel. “Clostridium difficile” Journal of Continuing Education Topics 12 (2010): 102, accessed April 20, 2015, ISSN: 1522-8606.
Ian R. Poxton. “Clostridium difficile” Journal of Medical Microbiology, 54 (2005): 97-100, accessed April 10, 2015, doi: 10.1099/jmm.0.45951-0.
Neal Patel, “Fecal Microbiota Transplant for Recurrent Clostridium difficile Infection: Mayo Clinic in Arizona Experience” Mayo Clinic Proceedings 88 (2013): 799-805, accessed April 19, 2015, doi: 10.1016/j.mayocp.2013.04.022.
Daniel Paredes-Sabja et al. “Clostridium difficile spore biology: sporulation, germination, and spore structural proteins,” Trends in Microbiology 22 (2014): 406, accessed April 15, 2015, doi: 10.1016/j.tim.2014.04.003.
Matthew Saramore et al. “Multicenter Typing Comparison of Sporadic and Outbreak Clostridium difficile Isolates from Geographically Diverse Hospitals” The Journal of Infectious Diseases 176 (1997): 1233-1238, accessed April 14, 2015, doi: 10.1086/514117.
Mohammed Sebaihia et al. “The multidrug-resistant human pathogen Clostridium difficile has a highly mobile, mosaic genome,” Nature Genetics 38 (2006): 779-786, accessed April 20, 2015, doi:10.1038/ng1830.
R. D. Rolfe and W. Song, “Immunoglobulin and non-immunoglobulin components of human milk inhibit Clostridium difficile toxin A-receptor binding,” Journal of Medical Microbiology, 42 (1995): 10-19, accessed April 18, 2015, doi: 10.1099/00222615-42-1-10.
AM Seekatz and VB Young, “Clostridium difficile and the microbiota,” Journal of Clinical Investigation, 124 (2014): 4182-9, accessed April 17, 2015, doi: 10.1172/JCI72336.
Peter J. Turnbaugh et al. “An obesity-associated gut microbiome with increased capacity for energy harvest” Nature 444 (2006): 1027-1031, accessed April 19, 2015, doi:10.1038/nature05414.
Louis Valiquette et al. "Impact of a Reduction in the Use of High-Risk Antibiotics on the Course of an Epidemic of Clostridium difficile-Asociated Disease Caused by the hypervirulent NAP1/027 strain" Oxford Journals: Clinical Infectious Diseases 25 (2007): S112-S121, accessed April 18, 2015, doi: 10.1086/519258
Daniel E. Voth and Jimmy D. Ballard, “Clostridium difficile Toxins: Mechanism of Action and Role in Disease,” Clinical Microbiology Reviews 18 (2005): 249-263, date accessed April 19, 2015, doi: 10.1128/CMR.18.2.247-263.2005.
Debbie Weston. Infection Prevention and Control: Theory and Practice for Healthcare Professionals. (Hoboken, Wiley-Interscience, 2008), 15-55.
Lois S. Wiggs/CDC (PHIL #6260), Scanning electron micrograph of C. difficile bacteria obtained from the CDC Public Health Image Library. 2004. Wikimedia Commons.
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2015, Kenyon College.
