Coccidioides immitis
A Microbial Biorealm page on the genus Coccidioides immitis
Classification
Higher order taxa (1)
cellular organisms; Eukaryota; Fungi/Metazoa group; Fungi; Dikarya; Ascomycota; Pezizomycotina; Eurotiomycetes; Eurotiomycetidae; Onygenales; mitosporic Onygenales; Coccidioides
Species
|
NCBI: Taxonomy |
Coccidioides immitis
Description and significance
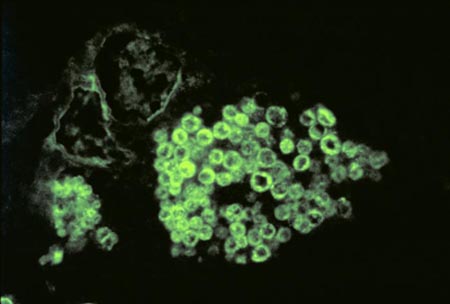
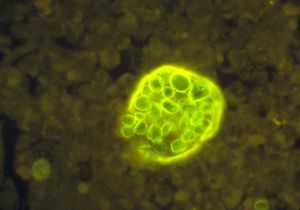
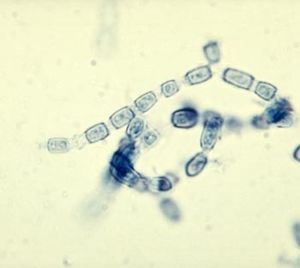
Coccidioides immitis is a dimorphic fungus exist in two distinct forms, saprophytic and parasitic. In the soil, the organism exists as a mold with septated hyphae resembling a shape of a barrel. When entering a host, the arthroconidia (spores) break off from the hyphae and evolve into round structures called spherules. Then when inside the host, spherules grows and undergo internal division, forming smaller structures called endospores. Large spherules can rupture to release packets of endospores, resulting in new foci of infection within the host (2). Coccidioides immitis main habitat is in soil (14)
Formal description of Coccidioides immitis was performed by Rixford and Gilchrist from a case observed in California in 1986 (3). However, the parasite was then still thought to be a protozoan. The correct taxonomic status of C. immitis as an ascomycete fungus was demonstrated by Ophuls and Moffit in 1900 (4) by culture on artificial of the fungal mycelia. It is important to have its genome sequences because Coccidioides immitis, along with its relative Coccidioides posadasii, can cause a disease called Coccidioidomycosis (Valley Fever), and it is a rare cause of meningitis, mostly in immunocompromised persons, and the disease can be fatal. Persons afflicted with HIV/AIDS are highly susceptible to Coccidioidomycosis and suffer a high mortality rate from the disease. (5).
Genome structure
The current total unique contig length of Coccidioides immitis genome is 28,876,843 base pairs; the estimated genome size is 29 Mb. Also, Coccidioides immitis strain RS was sequenced by way of sequencing each end of plasmids and Fosmids from randomly sheared fragments of 4 kb, 10 kb, and 40 kb average size. Coccidioides immitis strains contain either a MAT1-1 or a MAT1-2 idiomorph, which is 8.1 or 9 kb in length, respectively, the longest reported for any ascomycete species. MAT1-1 and MAT1-2 contain four or five genes more than are present in the MAT loci of most ascomycetes. Along with their cDNA structures, all genes in the MAT loci are transcribed (6). An analysis of 436 Coccidioides isolates from patients and the environment indicates that in both Coccidioides immitis and C. posadasii, there is a 1:1 distribution of MAT loci, as would be expected for sexually reproducing species. The current assembly contains 50 sequence contigs in 7 supercontigs (7).
Coccidioides immitis SOWgp gene was also isolated; SOWgp encodes an immunodominant, spherule outer wall glycoprotein that is presented as a component of a parasitic phase-specific, membranous layer at the cell surface. SOGwgp translates a 422-amino-acid (aa) polypeptide that contains 6 copies of a 41- to 47-residue tandem repeat enriched in proline (20.4 mol%) and aspartate (19.7%)(8). The recombinant polypeptide (rSOWp) was shown to bind to mammalian extracellular matrix proteins in an in vitro assay, suggesting that the parasitic cell surface antigen may function as an adhesion. Deletion of the SOWgp gene by using a targeted gene replacement strategy resulted in partial loss of the ability of intact spherules to bind to ECM proteins and a significant reduction in virulence of the mutant strain. The parasitic cell surface glycoprotein encoded by the SOWgp gene appears to function as an adhesion and contributes to the virulence of C. immitis.(8)
Cell structure and metabolism
Cells of Coccidioides immitis in the hyphal stage have thin, structure-less walls and a cytoplasmic membrane. Many nuclei, elongated mitochondria with both transverse and longitudinal cristae, and lipid particles are present. A large central accumulation of electron transparent polysaccharide appears in the spherule as the cell grows. The peripheral cytoplasm contains nuclei, each enclosed in a double-layered membrane, mitochondria, and small dense particles. Prior to cleavage the polysaccharide droplets are lost, while mitochondria become small and spherical. Endospores are formed and liberated when the spherule wall breaks. These begin to grow and repeat the cleavage cycle. (11)
Cell wall: Like most pathogenic fungi, the cell wall of Coccidioides immitis is rich in chitin and chitin metabolism is a reasonable target for the design of antifungal agents. The antigenic composition of an alkali-soluble, water-soluble cell wall extract of Coccidioides immitis is heterogeneous in composition, containing two distinct antigenic components. One is present as a polymer that is antigenically identical to a polymeric antigen in coccidioidin, designated antigen 2. The other component presented an unusual precipitin pattern in that a cathodal leg was demonstrable in the absence of an anodal leg (9). The fungal cell wall also contains mannoproteins and glucans. Any disruption in its integrity should affect growth. The cell wall provides a unique therapeutic opportunity for antifungal agents by targeting a structure not found in mammalian cells. The echinocandins are cyclic hexapeptides, members of a new class of antifungal agents. They appear to inhibit the synthesis of 1, 3-β-d-glucan, a major cell wall component which provides structural integrity and osmotic stability in most pathogenic fungi (10).
Metabolism: Of various carbohydrates and amino acids tested, glucose, mannose, fructose, and glutamate were the most efficient substrates metabolized by the endospores and spherules of Coccidioides immitis (12). Strain M-11 of Coccidioides immitis was found to utilize mannitol for growth in the mycelial form but not in the spherule form. Cell-free extracts of both forms, grown on glucose, were capable of reducing nicotinamide adenine dinucleotide with mannitol-1-PO but not with mannitol. The extracts accomplished a rapid oxidation of reduced nicotinamide adenine dinucleotide by fructose-6-PO, the expected product of mannitol-1-PO oxidation. (13)
Ecology
The distribution of Coccidioides immitis has been shown to be restricted to America through ecological and epidemiological studies. Coccidioides immitis has been known for its saprophytic nature, tolerance for high soil salinity in climates of high temperature and low rainfall (14). Epidemiological studies and case reports have extended the apparent range of this organism; however, almost all soil isolates in Arizona and California have been from areas with the physical and biotic parameters of this zone. The repeated isolation of C. immitis from the soil has been from small, well defined areas (14). C. immitis has been observed to grow in the soil if naturally infected soil is simply enriched with sterile water. The ease with which it can be grown is paradoxical to its naturally restricted distribution to soils in endemic areas (15). Animals, although not the major natural reservoir, cannot be ignored as possible factors in the ecology of C. immitis.
Pathology
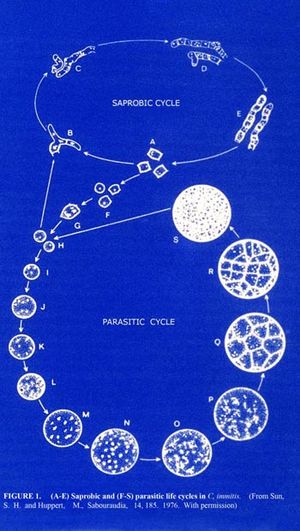
Coccidioides immitis Coccidioides immitis possess branching septate hyphae that are 2 to 4m in diameter (16) and allow them to develop infectious arthroconidia along its length. Such structure is the most major virulent factor for Coccidioides immitis. As the arthroconidia mature, they become spores that are easily airborne and can be carried long distances by the wind. It takes only a few inhaled arthroconidia to produce infection in the host. When inhaled, the arthroconidia is small enough (< 10m) (16) to escape upper respiratory defenses to lodge in the lungs where it transforms into a multinucleated spherule rather than a yeast. Using internal division, the spherule produces hundreds of uninucleated endospores. The spherule ruptures on maturity releasing the endospores, which in turn matures to from new spherules. (16)
The initial host immune response is a transient polymorphonuclear leukocyte response directed at arthroconidia. As spherules develop, the inflammatory response becomes mononuclear and persists throughout the course of the infection. The primary lung infection is asymptomatic in 60% of individuals. The other 40% develop a mild to moderate influenza-like syndrome 1 to 3 weeks (17) after exposure that includes cough, fever, night sweats, pleuritic chest pains, arthralgias, myalgias, fatigue, and anorexia. Chest radiographs often show lung infiltrates and perihilar adenopathy with occasional pleural effusions. Most pulmonary infections are cleared by the immune system with symptoms resolving in 2 to 3 weeks (17) or occasionally longer if the individual is immunosuppressed.
Application to Biotechnology
Antigen 2 is a glycosylated protein present in the cell walls of Coccidioides immitis.The gene encoding Ag2 was cloned from genomic DNA derived from the mycelial phase of C. immitis by PCR (18). As a result, it was predicted that a prepro-Ag2 undergoes a post-translational modification to yield the mature glycosylated Ag2 protein which is anchored on the extracellular plasma membrane of mycelial and spherule-phase cells. The production of a recombinant peptide that expresses C. immitis-specific Ag2 epitopes provides a useful reagent for examining the role of anti-Ag2 antibodies in coccidioidomycosis. (18)
Current Research
Coccidioides posadasii Δchs5 is a strain that is excluded from the select agent list. Sixteen assays using test reagents from three different ACCUPROBE Coccidioides immitis culture identification test lots had an average of 132,998 relative light units (RLU), which is well beyond the 50,000-RLU positive cutoff value for the test. Coccidioides posadasii Δchs5 is a satisfactory quality control isolates in the ACCUPROBE culture identification test for Coccidioides immitis. (19)
Coccidioidal meningitis (CM) is a devastating disease that requires long-term therapy and for which there is little hope of a cure. A model was used to compare the efficacies of itraconazole and fluconazole. CD-1 mice were infected intrathecally with 30 to 36 viable arthroconidia of Coccidioides immitis. Cyclodextrin (control) or itraconazole or fluconazole at were compared. Treatment with both antifungal at all doses prolonged survival compared with that of the control treatment. Itraconazole and fluconazole were equivalent. Itraconazole prolonged survival compared to that achieved with fluconazole at certain dosages. Thus, both itraconazole and fluconazole were effective at controlling CM, but neither eliminated Coccidioides from tissues. Overall, itraconazole was more efficacious on an mg/kg basis; at high doses they were similarly effective. (20)
Currently, direct smear and culture are the most common means of identifying Coccidioides spp. These methods offer relatively sensitive and specific means of detecting Coccidioides spp., but may take up to 3 weeks, potentially delaying the diagnosis and initiation of appropriate antifungal therapy. The need for a rapid and safe means of diagnosing coccidioidomycosis prompted a development of real-time PCR assay to detect Coccidioides spp. directly from clinical specimens. Primers and fluorescent resonance energy transfer (FRET) probes were designed to target the internal transcribed spacer 2 region of Coccidioides. The sensitivity of the assay testing 148 paraffin-embedded tissue samples is 73.4%. A rapid method for the detection of Coccidioides spp. directly from clinical material will greatly assist in the timely diagnosis and treatment of patients, while at the same time decreasing the risk of accidental exposure to laboratory personnel. (21)
References
1. http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?id=5501
2. Drutz, D. J., and M. Huppert. Coccidioidomycosis: factors affecting the host-parasite interaction. J. Infect. Dis. 147: 372-390 (1983)
3. Rixford E, Gilchrist TC.Two cases of protozoan (coccidioidal) infection of the skin and other organs Johns Hopkins Hosp Rep 1:209-268. (1896)
4. Ophuls, W. and H. Moffitt.new pathogenic mould. Philadelphia Med. J. 5: 1471-1472.(1900)
5. Ampel, N. M., K. J. Ryan, P. J. Carry, M. A. Wieden, and R. B. Schifman Fungemia due to Coccidioides immitis. Medicine. 65:312-321 (1986)
6. Mandel MA, Barker BM, Kroken S, Rounsley SD, Orbach MJ, Genomic and population analyses of the mating type Loci in Coccidioides species reveal evidence for sexual reproduction and gene acquisition. Epub 1189-99. (2007)
7. Coccidioides immitis Sequencing Project. Broad Institute of Harvard and MIT (http://www.broad.mit.edu)
8. Hung, C., Yu J.,Seshan K., Reichards U., and Cole G.T. A parasitic phase-specific adhesin of Coccidioides immitis contributes to the virulence of this respiratory fungal pathogen. Infect. Immun. 70:3443-3456.(2002)
9. Cox, R. A., Brummer, E., and Lecara, G. In vitro lymphocyte transformation responses of coccidioidin skin test-positive and -negative persons to coccidioidin, spherulin, and a Coccidioides cell wall antigen. Infect. Immun. 15:751-755.(1976)
10. Current, W L.,Tang, J.,Boylan, C.,Watson, P.,Zeckner, D.,Turner, W.,Rodriguez, M.,Dixon, C.,Ma, D., Radding, J A. Biosynthesis as a target for antifungals: the echinocandin class of antifungal agents. In: Dixon G K, Copping L G, Hollomon D W. , editors. Antifungal agents. 1st ed. Oxford, United Kingdom: Bios Scientific Publishers; (1995)
11. Breslau, A. M., Hensley, T. J. & Erickson, J. O.,Electron microscopy of cultured spherules of Coccidioides immitis. J. Biophys. Biochem. Cytol. 9: 627–637.(1961)
12. Lones, G., Peacock, C. Studies of growth and metabolism of Coccidioides immitis. Annals of the New York Academy of Sciences 89 (1), 102–108.(1960)
13. Sippel, J., Levine H. B. Sugars and Amino Acids as Carbon, Nitrogen, or Energy Sources for Coccidioides immitis Spherules and Endospores. Appl Microbiol. 18(3): 522–524 (1969)
14. Davis, B. L., Jr., Smith, R. T. &Smith, C. E. An epidemic of coccidioidal infection (coccidioidomycosis). J. Am. Med. Assoc. 118:1182–1186. (1942)
15. Egeberg, R. O. &Ely, A. F. Am. Coccidioides immitis in the soil of the Southern San Joaquin Valley.J. Med. Sci. 231:151–154. (1956)
16. Zaki SR, Greer PW, Coffield LM, Goldsmith CS, Nolte KB, Foucar K, Feddersen RM, Zumwalt RE, Miller GL, Khan AS: Hantavirus pulmonary syndrome. Pathogenesis of an emerging infectious disease. Am J Pathol 146:552-579 (1959)
17. Shubitz, L., T. Peng, R. Perrill, J. Simons, K. Orsborn, and J. N. Galgiani. Protection of mice against Coccidioides immitis intranasal infection by vaccination with recombinant antigen 2/PRA. Infect. Immun. 70:3287-3289.(2002)
18. Zhu, Y., V. Tryon, D. M. Magee, and R. A. Cox. Identification of a Coccidioides immitis antigen 2 domain that expresses B-cell-reactive epitopes. Infect. Immun. 65:3376-3380.(1997)
19. Zimmermann CR, Johnson SM, Martens GW, White AG, Pappagianis D. Cloning and expression of the complement fixation antigen-chitinase of Coccidioides immitis. Infect Immun.64(12):4967–4975.(1996)
20. Kamberi, P., Sobel, R., Clemons, K., Waldvogel, A., Striebel, J., Williams, P., Stevens, D. Comparison of Itraconazole and Fluconazole Treatments in a Murine Model of Coccidioidal Meningitis. Antimicrob Agents Chemother: 51(3): 998–1003. (2007)
21. Binnicker M.J., Buckwalter S.P, Eisberner J.J., Stewart R., McCullough A., Wohlfiel S.L., Wengenack N.L. Detection of Coccidioides Species in Clinical Specimens by Real-Time PCR (2007) J Clin Microbiol. 45(1): 173–178.
Edited by Kevin Phung, student of Rachel Larsen
