Colwellia psychrerythraea neu2011
Classification
Higher order taxa
Domain: Bacteria
Phylum: Proteobacteria
Class: Gammaproteobacteria
Order: Alteromonadales
Family: Colwelliaceae
Genus: Colwellia
Species: Colwellia psychrerythraea
Strain: 34H / ATCC BAA-68
Description and Significance
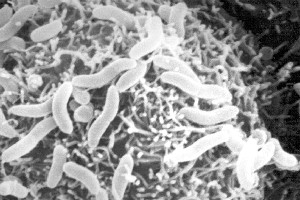
Colwellia psychrerythraea is a gamma proteobacteria that is strictly psychrophilic. It is a gram-negative facultative anaerobic that appears rod-shaped and red in pigment. [2] Figure 1 of C. psychrerythraea depicts this rod-shape. This bacterium possesses flagella for motility in its natural habitat of the deep sea Arctic and Antarctic Ice, the coldest marine habitat on Earth. [see Ecology] The strain 34H/ATCC BAA-681 (Vibrio psychroerythus) that was isolated from this marine habitat shows flagella present. Its psychrophillic nature allows it to grow from -1°C to 10°C; while optimal growth appears at 8°C. Strain 34H is able to grow under deep sea pressure and maintain swimming capabilities at temperatures of -10°C. Colwellia also produces extracellular polysaccharides and enzymes with low temperature optima for activity and marked heat instability. [3]
The extreme habitat that Colwellia is able to survive in makes this species unique and valuable. Since completion of its genome, C. psychrerythraea strain 34H has become a model for the study of life in permanently cold environments, revealing capabilities important to carbon and nutrient cycling, bioremediation, production of secondary metabolites, and coldadapted enzymes. There are suggestions that coldadaptation throughout the entire cell, especially proteome composition, may enhance enzyme effectiveness at low temperatures [3]. These enzymes are capable of degrading high molecular weight compounds, obtaining energy and nutrients for the cell, confirming this organism's evolutionary success and importance in environmental cycles. Adaptation, rather than a unique set of genes, allows Colwellia to survive in such harsh conditions [3]. These features make Colwellia species important in carbon and nutrient cycling in the cold marine environments in which they inhabit. Colwellia can possibly give insight into earlier Earth environments as well as those on other planets and moons. Adaptations to protein architecture essential to cold-active enzymes has made Colwellia attractive for exploitation in a number of biochemical, bioremediation, and industrial processes.
Genome Structure
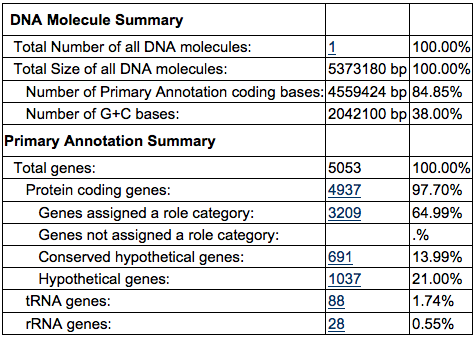
C. psychrerythraea consists of a single, circular chromosome. The chromosome is made up of 5,373,180 base pairs. About 38% of the chromosome consists of G-C pairs and 62% contains A-T pairs, as seen in Table 1. C. psychrerythrae has 5,053 genes. Of those genes, 4,397 are protein coding genes. [4] Protein structure of cold-active enzymes is still not well understood. However, this makes C. psychrerythraea a model organism for cold adaptation. [3] Further studies confirmed that C. psychrerythraea has an outer membrane and peptidoglycan layer. The peptidoglycan layer contains proteins like FtsZ and FtsA, which aid in cell division. [5]
C. psychrerythraea is one of the few organisms that have overcome environmental hardships. Most organisms cannot tolerate low temperatures or high levels of pressure. However, C. psychrerythraea has been found to have polyunsaturated and methyl-branched fatty acids and cis-unsaturated double bonds. These qualities help with membrane fluidity and the ability to withstand pressure. C. psychrerythraea also contains cold-shock proteins, which help the organism tolerate such cold temperatures. [6]
Cell Structure and Metabolism
Cell membrane
The cell membrane with flagella for mobility in C. psychrerythraea is maintained in a liquid-crystalline state with enhanced fluidity and reduced viscosity. Coding sequences (CDSs) reveal the production of polyunsaturated fatty acids, including polyketide-like polyunsaturated fatty acid synthases (CPS3104, CPS3103, CPS3102, and CPS3099). In addition, a fatty acid cis/trans isomerase that can alter the ratio of cis- to trans-esterified fatty acids in phospholipids has also been identified. Such polyunsaturated fatty acids and increased cis-isomerization increase membrane fluidity at low temperature. [3]
Several genes have been identified in the synthesis of fatty acid and phospholipid biosynthesis. CPS2297, or 3-oxoacyl-(acyl-carrier-protein) reductase, catalyzes the first reduction step in fatty acid biosynthesis. CPS1608, one of the putative genes, is thought to be located in an operon with other fatty acid metabolism genes in an approximate 15-kilobase region of the genome involved in fatty acid metabolism and branched-chain amino acid catabolism. Such short distance suggests that the presence of branched-chain amino acids results in the sysnthesis of branched-chain fatty acids. The branched-lipids in the cell membrane reduces membrane viscosity at cold temperature. Other proteins, β-keto-acyl carrier proteins (KAS-II and KAS-III), are key to the synthesis and regulation of the straight and branched-chain lipid ratios in the cell membrane. [3]
Motility
C. psychrerythraea has found to be motile at low temperatures. The C. psychrerythraea strain 34H shows motility at as low as -10°C. [7]
Extracellular compounds
C. psychrerythraea synthesizes extracellular polysaccharides and degradative enzymes. The extracellular polysaccharides can act as cryoprotectants; the extracellular degradative enzymes are located external to the cytoplasm, and can dissolve organic carbon in the cold environments to meet threshold requirements. [3]
Compatible solutes
Transporter families in C. psychrerythraea are involved in the uptake of compatible solutes that may play a role in osmoprotection as well as cryoprotection. C. psychrerythraea may benefit from these compounds also as carbon, nitrogen, and energy source. [3]
Carbon and nitrogen reserves
C. psychrerythraea produces and degrades polyhydroxyalkanoate (PHA) compounds, a family of polyesters that serves as intracellular carbon and energy reserves. Some of the PHA compounds have been found to be associated with pressure adaptation. There are three copies of genes devoted to PHA synthesis, two of which have no homologs in other bacterial lineages. C. psychrerythraea also produces and degrades polyamides similar to cyanophycin, protein-like polymers that function as nitrogen reserves. [3]
Synthesis of both PHA and cyanophycin-like compounds is beneficial to the psychrophilic habitat to ensure the intracellular reserves of carbon and nitrogen to elude the limitation in carbon and nitrogen uptake due to cold temperature. Additionally, PHA and cynanophycin-like compounds are of industrial interest as good sources of chemical synthesis as well as substitutes. [3]
Energy metabolism
The genome of C. psychrerythraea encodes enzymes for glycolysis, the tricarboxylic acid cycle, pentose phosphate pathway, beta-oxidation of fatty acids, and fermentative reactions (propionate catabolism). The carbon sources range from simple sugars to high-molecular-weight organic compounds. Under aerobic conditions, oxidation of ethanol occurs. ATP synthesis is completed with proton and sodium ion translocation. [3]
As a facultative anaerobe, under aerobic conditions, even with low concentrations of oxygens, C. psychrerythraea can use oxygen as a terminal electron acceptor. Under anaerobic conditions, C. psychrerythraea produces energy via an alternative electron transport chains, using molecules like trimethylamine-N-oxides (TMAO) and dimethylsulfoxides as alternative electron acceptors. [3]
C. psychrerythraea may also be involved in sulfur metabolism. Oxidation of intracellular sulfur, and hydrolysis of sulfur esters and aryl sulfates by sulfatases result in sulfur for growth and energy production. [3]
Lastly, sarcosine metabolism exists in C. psychrerythraea as well. Sarcosine oxidase reaction produces serine from sarcosine, which can be used in biosynthetic pathways and catabolism of glycine to carbon dioxide and ammonia. [3]
Unique capacities
Cold-shock domain proteins in the cytoplasm in C. psychrerythraea enhance cold adaptation. Such proteins are active and of optimal activity at cold temperatures and during heat instability period. Synthesis and catabolism of complex, high-molecular-weight organic compounds and possible C1 metabolism that facilitate a wide range of reactions to its environment also emphasizes the fact that C. psychrerythraea can play a role in carbon and nutrient cycles of cold environments. These compounds have practical indications in detoxification and bioremediation of cold environments and other biotechnogical applications. [3]
The biosynthesis and utilization of coenzyme F420 are also discovered in C. psychrerythraea. F420 was first found in methanogens in methanogenesis. F420 may be critical in C1 or aromatic compound metabolism. [3]
To protect the polyunsaturated fatty acids in the cell membrane from oxidative damage, C. psychrerythraea possesses the ability to respond to oxygen species. There are genes in C. psychrerythraea encoding antioxidants, and superoxide dismutase that neutralizes oxygen species. [3]
Ecology

C. psychrerythraea inhabits the deep Arctic sea and Antarctic Ice, the coldest marine habitat on Earth, seen in figure 2. By volume, most of Earth's biosphere is cold and marine, with 90% of the ocean's waters at 5°C or colder. Twenty percent of Earth's surface environment is frozen, including permanently frozen soil, glacial ice, polar sea ice, and snow cover. [8]
The psychrophillic Colwellia has adapted to these cold habitats. This genus produces enzymes capable of degrading high-molecular-weight organic compounds, making Colwellia an important component of carbon and nutrient cycling in these cold marine environments. Genome analysis suggests that cold adaptation of C. psyhchrerythraea consists of a collection of synergistic changes in overall genome configuration in terms of gene content and amino acid composition rather than the presence of a unique set of genes indicative of a psychrophilic lifestyle [3]. Enzymes favoring cold-adaptation show increased polar residues (particularly serine), the substitution of the amino acid aspartate for glutamate, and a general decrease in charged residues [3]. These changes are consistent with current theories that increased flexibility and reduced thermostability contribute to enzyme cold adaptation. Other adaptations noted are synthesis of cryotolerant cell membranes, PHAs (polyhydroxyalkanoates) to aid in pressure adaptation, cyanophycin-like compounds, and glycine betaine, as well as production of copious quantities of extracellular enzymes and polysaccharides [3]. Colwellia are also able to overcome temperature-dependent barriers in the uptake of carbon and nitrogen. These adaptive strategies appear to increase fitness by effectively overcoming multiple obstacles at low temperatures and help to maintain nutrient cycling in these cold environments.
Pathology
C. psychrerythraea is not known to be pathogenic to any organisms.
Current Research
In June 2005, The Institute of Genomic Research conducted an experiment to sequence the genome of C. psychrerythraea strain 34H. Due to C. psychrerythraea’s psychrophilic lifestyle, the purpose of this experiment was to further understand the genomic structures that aid in cold adaption. Since there is little known about cold adaption, C. psychrerythraea was chosen as the best organism to study. Extensive genomic analysis revealed the organisms’ genome structure, including its singular chromosome with 5,373,180 base pairs. The cell membrane was also found to contain polyunsaturated fatty acids and cis-isomerases, which aid in the fluidity of the cell membrane. C. psychrerythraea was also found to contain several metabolic properties in carbon and nitrogen cycling, and cold-shock proteins. These features, among others found, further explained why the organism can survive in such cold-weather conditions. [3]
In September 2009, The Department of Bioscience and Biotechnology at Konkuk University conducted an experiment involving recombinant expression of cold shock proteins (Csp’s). C. psychrerythraea was found to contain cold-shock proteins, which are proteins that can have increased activity under cold conditions. However, they have not been fully studied. To further understand these proteins in psychrophilic organisms, plasmids of Csp’s from C. psychrerythraea were added to E.coli, because the Csp’s of C. psychrerythraea are similar to the Csp’s of E.coli. After purification, the Csp’s were found to have an optimal temperature of 37 degrees which is the same range for mesophilic organisms. This concluded that Csp’s used for this experiment can be subject to purification and expression in various psychrophilic organisms. [10]
In January 2009, The Canadian Journal of Microbiology published an article on the extracellular polysaccharide substances (EPS) by C. psychrerythraea. EPS production was tested under different temperature, pressure and salinity variables. The experiment showed that the EPS increased drastically under lower temperature conditions, higher pressure conditions, and lower salinity levels. This experiment concluded that the EPS of C. psychrerythraea shows more activity under extreme environmental conditions. [11]
Cool Factor
C. psychrerythraea is a promising bacterium for the development of temperature-sensitive strains of vaccines. Researchers at the University of Victoria in Canada have successfully isolated several essential genes from C. psychrerythraea and implanted them into the human pathogen Francisella tularensis.[11] Since the enzymes of C. psychrerythraea can only function at temperatures up to 20°C, this modified pathogen could then be implanted into cooler parts of the body to allow for an immune response to occur. Indeed, when Francisella tularensis was implanted into the tail of labs rats, it was able to thrive locally and elicit an immune response within the rat, but was unable to spread to warmer body parts due to the temperature-sensitivity of C. psychrerythraea enzymes. Upon dosing these same rats with normal Francisella tularensis several weeks later, the rats remained healthy, providing evidence that temperature-sensitive vaccines could provide a useful therapy for combating a vast variety of future illnesses. [12]
References
[1] Finkelstein, Richard. NCBI: Colwellia psychrerythraea 34H Genome Project at TIGR.
[4] J. Craig Venter Institute (2005). “General Information for Colwellia psychrerythraea 34H.”
[5] J. Craig Venter Institute (2005). “Genome Properties Report.”
[6] D’Amico, et al. (2006). "Psychrophilic microorganisms: challenges for life." EMBO Reports 12.
[7] Junge, ‘’et al.’’ (2002). “Motility of ‘’Colwellia psychrerythraea’’ Strain 34H at Subzero Temperatures.” ‘’Applied Environmental Microbiology’’ 69(7): 4282–4284.
[8] Deming, J.W., and H. Eicken (2007). "Life in ice." in Planets and Life: The Emerging Science of Astrobiology W.T. Sullivan and J.A. Baross, eds. Cambridge University Press, pp. 292-312
[9] Redsky (2010). "Arctic Ice in Methane Death Spiral." RedSky News.
[10] Chang-Hun Moon, et al. (2009). "Recombinant Expression, Isotope Labeling, and Purification of Cold Shock Protein from Colwelliapsychrerythraeafor NMR Study." Bull. Korean Chem. Soc. 30(11).
[12] Duplantis, et al. (2010). "Essential genes from Arctic bacteria used to construct stable, temperature-sensitive bacterial vaccines." Proceedings of the National Academy of Sciences.
Edited by Samantha Bogle, Kylie Huang, Cassie Madeiros, and Stacy Rivera; students of Iris Keren
