Cytomegalovirus in post transplant patients: Difference between revisions
No edit summary |
|||
| Line 1: | Line 1: | ||
{{Curated}} | |||
==Introduction== | ==Introduction== | ||
By: Rachel Rhee | By: Rachel Rhee | ||
Latest revision as of 18:22, 1 October 2015
Introduction
By: Rachel Rhee
The cytomegalovirus (CMV) is a double stranded DNA virus part of the Herpesviridae viral family also known as herpesviruses. Herpesviruses share common characteristics including the ability to remain metabolically inert within the human body for extended periods of time. The human cytomegalovirus (HCMV) also known as Human herpesvirus 5 (HHV-5) is a species that causes infections typically associated with the salivary gland. HCMV is a common virus where about 50%-80% of the general adult population is infected (Slonczewski, 2013). However, most people who are infected are unaware since the virus is typically dormant in healthy individuals. The high prevalence of the virus among the general population is due to the many avenues of viral transmission. It is easily transmitted by person-to-person contact through saliva, urine, semen, blood, and cervical secretions during birth. Those with compromised immune systems manifest symptoms similar to mononucleosis (Epstein-Barr virus), and are affected in just about every organ in the body. Immunocompromised individuals such as organ transplant recipients and AIDS patients are especially at risk for more severe primary infections including latent infection, asymptomatic viral shedding (when the virus is active but doesn’t show visible signs or symptoms), and life-threatening multisystem disease (Farrugia, 1992). In such patients, HCMV not only causes problems in the acute phase but also increases the risk of long-term complications (Soderberg-Naucler, 2008).
CMV virology
Among hundreds of herpes virus, there are only eight known to target humans. The human herpes viruses (HHV) are classified as α, β, and γ-herpesvirus. All the HHV have similar morphology— a unique 4-layered structure that is distinguished according to the type of glycosylated proteins on the cell envelope (Figure 2). The herpes virus is known for having a relatively large genome. Its double-stranded DNA is encapsulated in an icosahedral shaped capsid surrounded by a layer of tegument, accessory proteins between the capsid and envelope. Viral entry into host cells occurs via phagocytosis or by fusing the virus envelope with the host cell membrane. Upon entering the cell, virus particles are synthesized and put together in the nucleus. The lipid bilayer envelope is formed by budding off the nuclear inner membrane where it then travels to the trans-Golgi complex. Mature virions can kill the host cell through the lytic cycle by filling the host cell and bursting. Or they can enter the lysogenic cycle and remain inert in a latent state for long periods of time.

The CMV virion is part of the β-herpesvirus family and about 150-200 nm in diameter thus considered to be one of the largest animal viruses (Brennan 2001). The virus genome consists of 220 kb double-stranded DNA and shares a similar genome to the herpes simplex virus, but is about 50% larger (Evans and Kaslow, 1997). The human CMV (HCMV) is typically found in white blood cells and CD13-positive cells (Brennan 2001). HCMV is distinguished from other CMV strains according to the types of glycoprotein found on its cell envelope. The predominant glycoprotein on HCMV is glycoprotein B (gB) which is responsible for its pathogenicity. gB dominates most of the viral envelope and aids in attachment, penetration, and spread.
Even when the virus is dispersed in an infected patient, it can remain latent in multiple organs. It is detected in many cell types and easily transmissible through urine, saliva, and breast milk (Sinclair and Sissons 2006). The virus is harbored in a large population of the U.S. (about 60%) yet primary infection does not cause serious problems until the virus is reactivated (Cook 2007). The exact mechanism of HCMV reactivation is poorly understood but has been determined to cause severe illness in those that are immunocompromised such as organ transplant recipients and patients with AIDS. HCMV is also easily passed from an infected mother to her fetus resulting in congenital birth defects.
CMV infection in post-transplant patients
HCMV infection is the most common viral infection among transplant patients- about 20%-60% of all transplant recipients are infected (Brennan). Viral spread can occur easily prior to transplant if patients are already infected with HCMV. The potential for infection in organ transplant patients must be carefully assessed as it could result in not only rejection of the donor organ but also the death of the immunosuppressed organ recipient.
Primary (latent) infection
Part of its lifelong infection in post-transplant human hosts is due to its ability to remain dormant in myeloid lineage cells (Traylen et al., 2011). In latent infection, HCMV-seropositive patients have already established humoral and cellular immunity to the permanent residence of the latent virus. Latent infection is characterized by low-grade, persistent infection but the carriers of the virus do not manifest signs of active viral replication (Traylen et al., 2011). When the latent virus switches to a lytic phase, the virus is reactivated and the seropositive individual is characterized with secondary infection, the more common type of HCMV infection.
Secondary (reactivated) infection
Individuals that have never experienced a primary viral infection are HCMV-seronegative and become HCMV-seropositive upon receiving an infected donor organ and/or white blood cells after the transplant and transfusion. Those at highest risk for developing a primary infection after surgery are HCMV-seropositive donors and HCMV-seronegative recipients who become infected through HCMV-seropositive donors (Stitt, 2003). HCMV infections are severe including tissue-invasive HCMV and recurrence of the virus. Recurrence of the virus occurs when the virus undergoes replication resulting in more severe and chronic infections in immunocompromised individuals (Traylen et al., 2011). It is currently thought that myeloid cells that differentiate to dendritic cells are responsible for reactivating lytic gene expression which results in the production of infectious virus particles (Traylen et al., 2011).
The virus is commonly transmitted to HCMV-seronegative transplant patients through hemodialysis and blood transfusion (Stitt 2003). In transplant HCMV-seropositive recipients, the main stimulus for reactivating the virus is through immunosuppressive therapy which is deliberately induced for organ transplants or for graft-versus-host disease after a bone marrow transplant (Evans and Kaslow, 1997). Immunosuppressive drugs are necessary to prevent rejection of the donor organ however interacts with the virus in a complex manner which can result in increased risks associated with the virus.
Method of infection
HCMV is an immunomodulatory virus thus evades and is able to evade the host antiviral immune response by synthesizing proteins that protects it the virus from host immune system attacks (Lucas and McFadden, 2004). The four immunomodulatory proteins include are variations of glycoproteins that decrease expression of major histocompatibility complex (MHC) class I proteins on the surface of infected cells (Mocarski 2002). The virus acquires self-protective strategies by encoding and expressing immunomodulatory proteins which are prevalent with large-genome viruses such as the herpesvirus. HCMVs have a large, complex DNA with hundreds of open reading frames that result in two-to threefold greater number of gene products than other herpesviruses (Mocarski 2002). A large genome enables the virus to produce a variety of gene products that share related functions to host proteins associated with the immune system. Large genomes are an evolutionary advantage of the virus’s ability to respond to the host immune control and establish widespread infection.
The exact method of HCMV infection has yet to be elucidated but HCMV is known to be an opportunistic pathogen. Thus the intense activation of the immune system after organ transplantation provides an ideal opportunity for the HCMV virus to reactivate and replicate in seropositive recipients. From what is known about HCMV replication, reactivation is under control of an early promoter that controls expression of immediate early genes (Hunninghake et al., 1999). Two major transcription factors involved in replication include nuclear factor (NF)KB and cyclic adenosine monophosphate (cAMP)-dependent transcription factors which are expressed at high levels in infected cells (Hunninghake et al., 1999). Thus HCMV replication is largely dependent on the expression of transcription factors in infected host cells.
Implications of infection
There are severe implications for HCMV-infected patients, especially following renal transplantation. Whether the virus was endogenous in the patient or introduced through transfused blood or the donor kidney, the infection can be clinically manifested or asymptomatic. Nonetheless the success of the kidney transplant largely depends on whether the virus is active in the recipient or recurrent. The kidney recipient develops primary infection when an allograft donor is HCMV-seropositive but the receiver is seronegative (Evans and Kaslow, 1997). Even if both donor and receiver are seronegative, the administration of seropositive blood can lead to primary infection. The intensity of HCMV shedding in reccurent infection is contingent on the intensity of the immunosuppressant therapy post transplantation. In the acute phase, HCMV infection is clinically manifested in fever, chills, splenomegaly, and lymphopenia (Evans and Kaslow, 1997) and can cause long-term complications including transplant vascular sclerosis (TVS), autoimmune issues such as chronic graft, and host disease in bone marrow transplant recipients (Soderberg-Naucler, 2008).
Following renal transplantation, the success of the transplant is largely dependent on the virus. Whether the virus infection leads to allograft failure or the new kidney activates the virus, the exact mechanism is unknown due to the complexity of the issue. Factors that determine the severity of the infection depends on whether the HCMV infection is primary and also depends on the administration of anti-thymocyte globulin (ATG), an infusion of derived antibodies used in the prevention of organ rejection (Evans and Kaslow, 1997). ATG affects the cell-mediated immune response by lowering the T-cell count which exacerbates HCMV-infection symptoms (Plotkin et al., 1990). Clinical studies have shown that following the transplant of CHMV positive donor kidneys, patients who were vaccinated showed increased transplant success in comparison to unvaccinated patients (Plotkin et al., 1990). The influence of HCMV vaccination indicates the significant role of HCMV infection in graft-versus-host disease pathogenicity.
Disease Management
There are two common methods for managing symptomatic HCMV disease in infected transplant recipients: prophylactic and preemptive therapy. Prophylactic treatment is initiated at the time of cell engraftment or organ transplant. Typically, using a less potent antiviral agent can successfully suppress HCMV reactivation. Prophylaxis with ganciclovir, an antiviral agent, has resulted in a significant decline in HCMV-related morbidity (Singh, 2001). A major drawback to administrating ganciclovir is that patients are not likely to benefit from prophylaxis (Singh, 2001). The other approach, preemptive therapy, only involves prophylaxis for high-risk organ recipients that show asymptomatic symptoms. In these situations, HCMV replication occurs early thus preemptive therapy is an attempt to prevent disease progression. It is also known as an early treatment. The preemptive approach involves intermittent therapy in order to achieve a greater selection of resistant virus (Singh, 2001).
The role of HCMV in cancer
Research has shown that HCMV may be an oncomodulator, responsible for gene mutations that result in malignant transformation of healthy cells. However, there is no conclusive evidence proving that HCMV infection leads to transformation of normal cells nor the mechanism by which HCMV may contribute to oncogenesis. Nonetheless, there have been numerous recordings of viral DNA, mRNA, and/or antigens in tumor tissues implicating HCMV in the etiology of cancer cells. Studies on the molecular mechanisms of HCMV has been geared towards the viral modulation of apoptosis, angiogenesis, and cell migration (Figure 4).

Ongoing studies show that HCMV infection can protect tumor cells from apoptosis which is induced by the adenovirus E1A protein (Cinatl et al., 2004). The virus binds cell proteins Bcl-2, ∆Np73α, and phosphatidylinositol 3- kinase (PI3K)/AKT which lowers the rate of apoptosis, increases tumor progression, and increases resistance to chemotherapeutic agents (Cinatl et al., 2004). Angiogenesis is also an important feature of invasive tumor growth and regulated in HCMV infected cells. HCMV can modulate angiogenesis, the formation of new blood vessels, by down regulating negative regulators such as thrombospondin (TSP)-1 and TSP-2 in glioma cells (Cinatl et al., 2004).
Clinical research has shown that treating HCMV-infected neuroblastoma (NB) cells by inhibiting virus production restored the patient’s sensitivity to chemotherapy and lowered Bcl-2 protein expression to induce apoptosis as seen in normal cells (Cinatl et al., 1998). Figure 5a shows an ultrasound image of cells undergoing apoptosis which is characterized by masses of condensed chromatin in a fragmented nucleus (Figure 5, Cinatl et al., 1998). Upon treatment with a chemotherapy drug (CDDP), cells infected by HCMV and adjacent noninfected cells showed no sign of apoptosis (Figure 5b). A close up of the nucleus and cytoplasm from Figure 5b shows the dense viral capsids (Figure 5c and d). These findings demonstrate the implications of HCMV infection in cancer patients and its interference with chemotherapy drugs.
While it is unsure whether HCMV establishes persistent virus replication in tumor cells, data from experiments with HCMV-infected cells support the potential of HCMV to be an oncogenic virus. Oncomodulation of HCMV may not involve direct transformation of malignant cells but rather through virus regulatory proteins and noncoding RNA which can result in cell proliferation, survival, production of angiogenic factors, and immunogenicity (Michaelis et al., 2009).
Protection Against the Virus
Natural Immunity
While the HCMV virus does not cause much harm in the human body when latent, upon reactivation the virus can cause serious harm for immunocompromised individuals, blood transfusion patients, and for immature fetuses that acquire the infection from a seropositive mother. The most important use of HCMV vaccination would be to immunize women before pregnancy. The virus is readily transmitted from an infected mother to the fetus, therefore preconceptual HCMV immunity could protect newborns by establishing maternal immunity (Figure 5). Clinical data shows that mothers with naturally acquired immunity prior to conception are 69% less likely to pass the virus than women who are HCMV-seronegative (Heineman, 2007). Even if the virus is transmitted to the infant, infants born to mothers with HCMV immunity are less likely to show symptomatic disease at birth. Natural immunity to HCMV through vaccination confers substantial protection (between 70 - 90%) against HCMV infection and disease. These clinical observations strongly indicate that vaccination of seronegative women can be a strong preventer of congenital HCMV disease. In addition, vaccination can also result in reduced asymptomatic infection and viral shedding in HCMV -seropositive infants which would generally reduce the reservoir of HCMV viruses (Heineman, 2007).
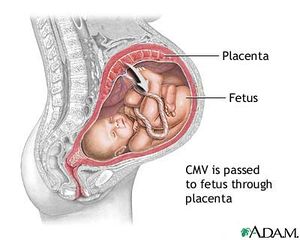
Likewise, HCMV disease from primary infection occurs more frequently in organ transplant recipients. Seronegative patients prior to the transplant showed symptomatic HCMV-infection 61% of the time upon receiving kidneys from seropositive donors. In contrast, only 24% of seropositive recipients who received kidneys from seropositive donors developed HCMV disease (Plotkin et al., 1990). Considering most HCMV infection was caused by the donor virus, this supports that prior immunity may lessen the severity of HCMV infection for transplant recipients. The problem with prior infection is the possible reactivation of latent HCMV at any moment, thus an attenuated virus vaccine called the Town vaccine was developed.
Immunology of CMV protection in the body
HCMV disease is controlled primarily through antibodies and cell-mediated immunity. Past studies have found that newborn blood transfusion recipients showed less severe HCMV disease when born to seropositive mothers. This suggests that antibodies, in the form of HCMV immune globulin, shared from the mother to child protected premature infants. Thus administration of HCMV-specific hyperimmune globulin to seronegative pregnant women can lead to effective prevention of congenital HCMV infection and disease (Heineman, 2007). Nonetheless, antibodies alone does not offer enough protection in mitigating CMV disease. The HLA-restricted CD8+ cytotoxic T lymphocyte (CTL) response plays an important role in cell-mediated immunity to HCMV. Thus an effective HCMV vaccine requires the induction of CTL responses possibly following inoculation with live attenuated vaccines.
One such vaccination, is the Towne strain which was isolated from the urine of a congenitally infected infant (Heineman, 2007). The Towne strain successfully induced antibodies similar to naturally induced antibodies of infected individuals, and also effectively induced cell-mediated immune responses. In a past clinical study, HCMV-seronegative kidney recipients were administered the live, attenuated Towne strain. Among those infected by the disease, symptoms were less severe and the incidence of primary infection among recipients did not change (Heineman, 2007).
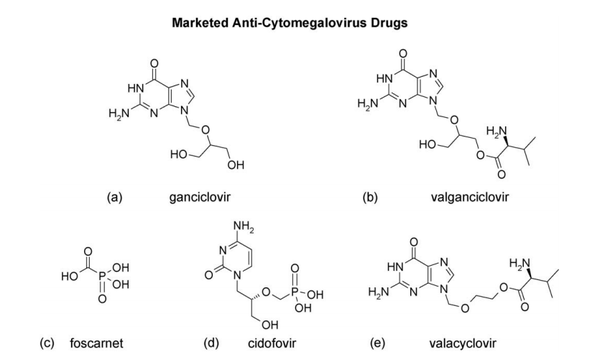
Antiviral Therapy
Ganciclovir is considered to be the gold standard for CMV disease management and is the first treatment regimen for infection in transplant recipients (Biron 2006). Like most antiviral drugs, it is a nucleoside analog which is effective in targeting viral DNA polymerase in order to inhibit DNA synthesis (Biron, 2006). Ganciclovir is first converted to ganciclovir triphosphate which replaces dGTP in the elongating viral DNA chain thus slowing transcription elongation (Biron, 2006). However, low bioavailability of oral ganciclovir results in lowered ganciclovir exposure. Thus concern over inadequate viral suppression led to the development of prodrug, valganciclovir, the L-valyl ester of the original ganciclovir (Biron, 2006). Other anti- HCMV drugs such as Foscavir and Cidofovir (Figure 4) have also been approved for treatment or prophylaxis of HCMV disease. Widespread use of prophylactic therapy has led to greater onset of late HCMV disease. Additionally, patients at risk for primary HCMV infection undergo longer regimens of antiviral therapy due to the threat of immunosuppression. This has led to the emergence of resistant viruses. Nonetheless, therapy treatments with antiviral agents have come a long way in significantly reducing the risk of other herpesviruses as well as bacterial and fungal infections (Biron, 2006).
Despite the availability of antiviral drugs, HCMV infection still remains a threat for patients in immunosuppressive therapy. Individuals with malignancies, organ recipients, and AIDS infection have severely impaired immune systems upon which HCMV causes symptomatic illness and often death. Additionally, there is current no antiviral drug for congenital HCMV disease in neonates. However, there has been considerable progress in developing several new drug candidates with potentially greater efficacy and safety.
References
1. Biron, K. K. (2006). Antiviral drugs for cytomegalovirus diseases. Antiviral research, 71(2), 154-163.
2. Cinatl, J., Scholz, M., Kotchetkov, R., Vogel, J. U., & Doerr, H. W. (2004). Molecular mechanisms of the modulatory effects of HCMV infection in tumor cell biology. Trends in molecular medicine, 10(1), 19-23.
3. Cook, C. H. (2007). Cytomegalovirus Reactivation in" Immunocompetent" Patients: A Call for Scientific Prophylaxis. Journal of Infectious Diseases, 196(9), 1273-1275.
4. Evans, A. S., & Kaslow, R. A. (Eds.). (1997). Viral infections of humans: epidemiology and control. Springer Science & Business Media.
5. Farrugia, E., & Schwab, T. R. (1992, September). Management and prevention of cytomegalovirus infection after renal transplantation. In Mayo Clinic Proceedings (Vol. 67, No. 9, pp. 879-890). Elsevier.
6. Heineman TC. Human cytomegalovirus vaccines. In: Arvin A, Campadelli-Fiume G, Mocarski E, et al., editors. Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis. Cambridge: Cambridge University Press; 2007. Chapter 71.
7. Gary W. Hunninghake, Martha M. Monick, and Lois J. Geist "Cytomegalovirus Infection", American Journal of Respiratory Cell and Molecular Biology, Vol. 21, No. 2 (1999), pp. 150-152. 8. doi: 10.1165/ajrcmb.21.2.f157
9. Lucas, A., & McFadden, G. (2004). Secreted immunomodulatory viral proteins as novel biotherapeutics. The Journal of Immunology, 173(8), 4765-4774.
10. Michaelis, M., Doerr, H. W., & Cinatl, J. (2009). The story of human cytomegalovirus and cancer: increasing evidence and open questions. Neoplasia, 11(1), 1-9.
11. Mocarski, E. S. (2002). Immunomodulation by cytomegaloviruses: manipulative strategies beyond evasion. Trends in microbiology, 10(7), 332-339.
12. Plotkin, S. A., Starr, S. E., Friedman, H. M., Gonczol, E., & Brayman, K. (1990). Vaccines for the prevention of human cytomegalovirus infection. Review of Infectious Diseases, 12(Supplement 7), S827-S838.
13. Singh, N. (2001). Preemptive therapy versus universal prophylaxis with ganciclovir for cytomegalovirus in solid organ transplant recipients. Clinical infectious diseases, 32(5), 742-751.
14. Slonczewski, J., Foster, W. Microbiology: An Evolving Science, 3rd Edition. W. W. Norton & Company, 10/2013. VitalBook file.
15. Traylen, C. M., Patel, H. R., Fondaw, W., Mahatme, S., Williams, J. F., Walker, L. R., … Akula, S. M. (2011). Virus reactivation: a panoramic view in human infections. Future Virology, 6(4), 451–463. doi:10.2217/fvl.11.21
Edited by Rachel Rhee, student of BIOL 238 Microbiology, taught by Joan Slonczewski, 2015, Kenyon College.
