Ecosystem and Microbial Populations in Pangong Lake, Himalaya
Introduction
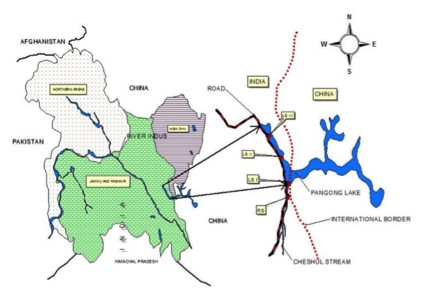
Pangong Lake, also known as Pangong Tso, is a high altitude brackish water lake expanding from India to Tibet[1]. It is 4350 m above sea level, with a length of 134 km and a catchment area of about 2000 km2[1]. Lying in the valley of the upper drainage basin of Indus River, Pangong Lake is the largest and most brackish wetland in the Trans-Himalayan cold desert ecosystem[1]. Due to its physical isolation and short summer months, Pangong Lake is less well understood than lowland lakes, and it is characterized by extreme environmental conditions and a relatively simple ecosystem in its lake body[1].
Physical Environment
Although melt water from catchment and rainwater keep feeding into Pangong Lake, the lack of an outlet and continuous evaporation of surface water raise the salinity to an average of 2-4% [1]. In addition, the lake water has a basic pH ranging from 8.74 to 9.11. Water temperature of Pangong Lake also varies greatly throughout the year due to its geographic location in the Ladakh cold desert. The surface water temperature reaches up to 19°C while the bottom water temperature only reaches 8°C in summer months. During winter, Pangong Lake remains frozen from late December to early April[1]. Therefore, Pangong Lake is categorized as a cold monomictic lake[1].
Ecosystem Members in Pangong Lake Body
Phytoplankton
Due to its high salinity and harsh environmental conditions, Pangong Lake body has relatively low biodiversity. No macrophyte has been found, However 23 taxa of phytoplankton have been recorded and together they are grouped into five classes: Bacillariophyceae, Chlorophyceae, Cyanophyceae, Xanthophyceae, and Rodophyceae[1]. Phytoplankton growth in Pangong Lake is controlled by several physio-chemical parameters including light intensity, temperature, nutrient concentration, lake currents, as well as predation by zooplankton [2][3]. Populations of phytoplankton only dominate during warm summer months[1]. In addition, the diversity and density of phytoplankton are inversely proportional to lake water salinity[4].
Zooplankton
The zooplankton population consists of adult Diaptomus species and their Nauplius larva. Besides high salinity and low temperature, zooplankton organisms in Pangong Lake have to protect themselves against high UV radiation. Both adult Diaptomus and their Nauplius larva synthesize special photo-protective compounds to capture harmful UV-induced radicals[5]. Carotenoids and melanin also form pigmentations on the body surfaces of zooplankton organisms. Together, these mechanisms act to protect the organisms from UV radiation.
Fish
No fish has been recorded in Pangong Lake body[1]. However, in its inlet Cheshul stream, three fish species have been identified: Schizopygopsis stoliczkae, Triplophysa stoliczkae and Triplophysa gracilis[1]. These species are also identified from Kashmir Valley[6], equatorial high mountain lake Lago San Pablo[7] and Antarctica saline lakes[8]. These findings suggest that these fish species are eurytopic and can tolerate wide ranges of environmental fluctuations to survive in extreme conditions.
Recently Discovered Microbial Populations
Microbial species in Pangong Lake are generally psychrophilic and halotolerant. These characteristics give the organisms competitive advantages to survive in this harsh isolated habitat. Many recently discovered microorganisms from Pangong Lake are of particular interest as they produce distinctive cold-adapted secondary metabolites, and their key enzymes remains active in low temperature environments. The metabolite compounds and enzymes are suggested to be economically significant and can be potentially applied to industrial, agricultural and medical processes in the future.
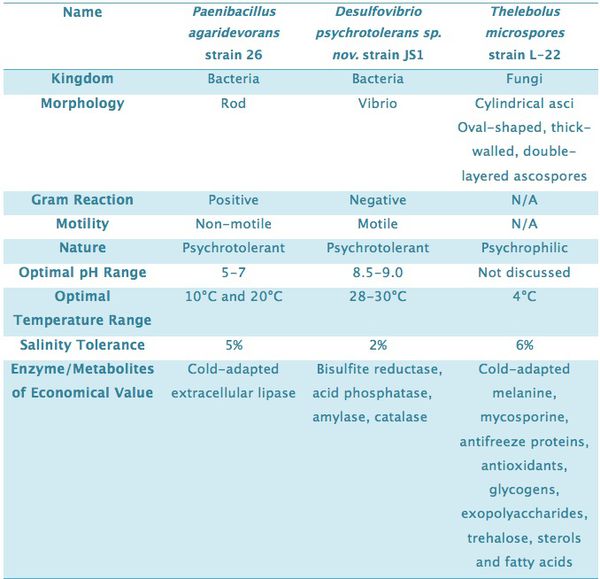
Paenibacillus agaridevorans strain 26
The Paenibacillus genus are rod-shaped bacteria which are capable of aerobic or facultative anaerobic respirations[9]. The Paenibacillus agaridevorans strain 26 was isolated from water and sediment samples from Pangong Lake[10]. It is a non-motile gram-positive psychrotolerant bacterium that grows optimally at 10°C and 20°C. P. agaridevorans strain 26 produces an extracellular cold-adapted lipase to hydrolyze substrate to obtain carbon source for cell metabolism and growth[10]. This enzyme is of particular interest as it is used widely in fat and oil processing, pharmaceutical production, paper manufacture and cosmetics[11].
Thelebolus microspores strain L-22
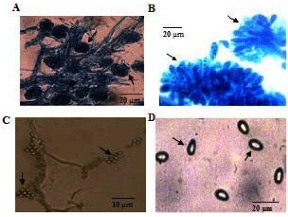
Thelebolus microspores L-22 is a strain of psychrophilic fungi isolated from water samples of Pangong Lake. This strain grows optimally at 4°C and can tolerate up to 6% salinity[12]. Genetically close strains were also isolated from the Ross Sea and Dry Valleys regions in Antarctica[13][14]. Similar to Pangong Lake, these areas are subjected to long periods of subzero temperature. T. microspores L-22 synthesizes cold-adapted melanine, mycosporine, antifreeze proteins, antioxidants, glycogens, exopolyaccharides, trehalose, sterols and fatty acids to protect itself from the extreme environment[15]. These proteins and metabolites are also important for biotechnological and pharmaceutical industries as they can withstand low or moderate temperatures[16].
Desulfovibrio psychrotolerans sp. nov. strain JS1
Desulfovibrio psychrotolerans sp. nov. strain JS1 was isolated from mud and water samples from Pangong Lake. It is a gram-negative bacterium with a single polar flagellum that mediates motion[17]. D. psychrotolerans JS1 is psychrotolerant (10°C - 50°C), and can tolerate up to 2% salinity[17]. The bacterium is also alkaliphilic and grows optimally at pH 8.9 – 9.0. The growth occurs in an anaerobic environment but the bacterium itself is aerotolerant. D. psychrotolerans JS1 is a chemo-organoheterotroph that utilizes various carbon sources. However it uses sulfate as its only electron acceptor[17]. Several key enzymes have been identified from D. psychrotolerans JS1, including amylase, catalase, bisulfite reductase, and acid phosphatase[17]. The psychrotolerant feature of these enzymes makes them valuable and useful for various biotechnological processes in the future.
References
1. Bhat F.A., Yousuf A.R., Aftab A., Arshid J., Mahdi M.D., and Balkhi M.H. “Ecology and Biodiversity in Pangong Tso (lake) and Its Inlet Stream in Ladakh, India”. International Journal of Biodiversity and Conservation, 2011. 3: 501-511.
2. Khan T.A. “Limnology of Four Saline Lakes in Western Victoria, Australia: I. Physico-chemical Parameters”. Limnologica, 2003. 33: 316-326.
3. Bhat F.A. “Limnological Characterization of Lake Tso Moriri, Ladakh”. University of Kashmir, 2009. Ph.D. Thesis.
4. Kipriyanova L.M., Yermolaeva N.I., Bezmaternykh M.M., Dvurechenskaya S.Y., and Mitrofanova E.Y. “Changes in the Biota of Chany Lake Along a Salinity Gradient”. Hydrobiologia, 2007. DOI: 10.1007/s10750-006-0295-9
5. Hansson L., Hylander S., and Sommaruga R. “Escape from UV Threats in Zooplankton: A Cocktail of Behavior and Protective Pigmentation”. Ecology, 2007. 88:1932-1939.
6. Wanganeo A. and Wanganeo R. “Algal population of Kashmir”. Himalaya. Arch. Hydrobiol. 1991. 121: 219-233.
7. Gunkel G. and Casallas J. “Limnology of an Equatorial High Mountain Lake – Lago San Pablo, Ecuador: the Significance of Deep Diurnal Mixing for Lake Productivity”. Limnologica, 2002. DOI: 10.1016/S0075-9511(02)80015-9
8. Hodgson D.A., Vyverman W., and Sabbe K. “Limnology and Biology of Saline Lakes in the Rauer Islands, Eastern Antartica”. Antarctic Science, 2001. DOI: 10.1017/S0954102001000372
9. Ash C., Priest F.G., and Collins M.D. “Molecular Identification of rRNA Group 3 bacilli (Ash, Farrow, Wallbanks and Collins) Using a PCR Probe Test”. Antonie van Leeuwenhoek, 1993. DOI: 10.1007/BF00873085
10. Vardhan S., Yadav A.K., Kashyap S., Panday A.K., Srivastava A.K., and Arora D.K. “Isolation and Characterization of Psychrotolerant Paenibacillus Agaridevorans Strain S26 from Subglacial Himalayan Lake”. International Journal of Pharma and Bio Sciences, 2012. 3: 819-827.
11. Chung Y.R., Kim C.H., Hwang I., and Chun J. “Paenibacillus koreensis sp. nov., a New Species that Produces an Iturin-like Antifungal Compound”. Int. J. Syst. Evol. Microbiol., 2000. 50:1495-1500.
12. Anupama P.D., Praveen K.D., Singh R.K., Kumar S., Srivastava A.K., and Arora D.K. “A psychrophilic and halotolerant strain of Thelebolus microsporus from Pangong Lake, Himalaya.” Mycosphere, 2011. DOI: 10.5943/mycosphere/2/5/10
13. Arenz B.E., Held B.W., Jurgens J.A., Farrell R.L., and Blanchette R.A. “Fungal Diversity in Soils and Historic Wood from the Ross Sea Region of Antarctica”. Soil Biology and Biochemistry, 2006. DOI: 10.1016/j.soilbio.2006.01.016
14. Connell L.B., Redman R., Craig S.D., and Rodriguez R. “Distribution and Abundance of Fungi in the Soils of Taylor Valley, Antarctica”. Soil Biology and Biochemistry, 2006. DOI:10.1016/j.soilbio.2006.02.016
15. Ruisi S, Barreca D, Selbmann L, Zucconi L, Onofri S. “Fungi in Antarctica”. Environmental Science and Biotechnology, 2007. 6:127–141.
16. Rosa M., Franz S., Jean-Claude M., and Charles G. “Psychrophiles from Biodiveristy to Biotechnology: Cold-adpated Fungi as a Source for Valuable Metabolites”. Springer, 2008. DOI: 10.1007/978-3-540-74335-4_22
17. Sasi Jyothsna T.S., Sasikala Ch., and Ramana Ch.V. “Desulfovibrio psychrotolerans sp. nov., a psychrotolerant and moderately alkaliphilic sulfate- reducing deltaproteobacterium from the Himalayas”. International Journal of Systematic and Evolutionary Microbiology, 2008. DOI: 10.1099/ijs.0.65402-0