Geobacter metallireducens
Template:Geobacter metallireducens
Classification
Higher order taxa
Bacteria; Proteobacteria; Delta Proteobacteria; Desulferomonadales; Geobacteraceae
Species
Geobacter metallireducens
Description and significance
Geobacter metallireducens is a rod shaped, gram negative, anaerobic bacteria and can be seen to have flagella and pili. The first G. metallireducens (initially known as strain GS-15) was first isolated from freshwater sediment, and was able to gain energy through dissimilatory reduction of iron, manganese, uranium and other metals. This organism was the first organism found to oxidize organic compounds to carbon dioxide with iron oxides as the electron acceptor (Lovely and Phillips 1988). G. metallireducens can also oxidize short chain fatty acids, alcohols, and monoaromatic compounds such as toluene and phenol using iron as its electron acceptor (Lovely et al 1993).
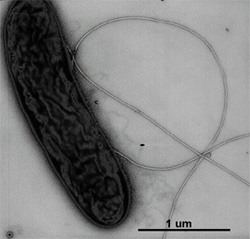
Genome structure
G. metallireducens contains genes for flagella synthesis. G. metallireducens was orginally thought to be immotile because they were grown in labs under ideal conditions where the bacteria had plenty of soluble metals. Synthesis for flagella do not initiate unless nutrient conditions are poor. Under conditions where soluble metals were replaced with iron oxide, G. metallireducens was seen to grow flagella and swim (Genome News Network 2002). G. metallireducens also contains genes that allow the bacteria the ability of chemotaxis.
Cell structure and metabolism
G. metallireducens is chemotactic towards Fe(II) and Mn(II)and expresses flagella and pili only when grown on insoluble Fe(III) or Mn(IV) oxide (Childers et al 2002). These results suggest that G. metallireducens senses when soluble electron acceptors are depleted which then triggers the synthesis of the appropriate appendages allowing it to search for, and establish contact with, insoluble Fe(III) or Mn(IV) oxide.
Ecology
G. metallireducens has been known to take part in bioremediation. In addition to using iron, the G. metallireducens uses metals such as plutonium and uranium to metabolize food. G. metallireducens consumes these radioactive elements and eats away at the contaminants. In the case of uranium, it changes the metal from a soluble to an insoluble form. The insoluble uranium drops out of the groundwater, thus decontaminating streams and drinking water. It remains in the soil and could then be extracted (Childer 2002). The use of an insoluble electron acceptor may explain why Geobacter species predominate over other Fe(III) oxide-reducing microorganisms in a wide variety of sedimentary environments.
Describe any interactions with other organisms (included eukaryotes), contributions to the environment, effect on environment, etc.
Pathology
How does this organism cause disease? Human, animal, plant hosts? Virulence factors, as well as patient symptoms.
Application to Biotechnology
Does this organism produce any useful compounds or enzymes? What are they and how are they used?
Current Research
Enter summaries of the most recent research here--at least three required
References
Edited by Christine Tang student of Rachel Larsen and Kit Pogliano
