Gunnera Cyanobacteria symbiosis
Gunnera
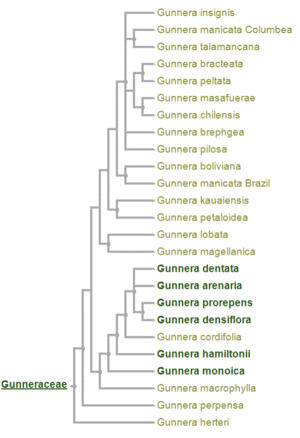
Gunnera is the only genus in the family Gunneraceae [1]. Gunnera, consisting of about 30-40 species, is a herbaceous flowering plant found primarily in the southern hemisphere [2] in places like Central and South America, Africa, Madagascar, Tasmania, New Zealand and Hawaii [1].
Some species of Gunnera have well documented uses. Gunnera tinctoria, for example, is often used in dishes and liquors in Southern Chile and Argentina [3]. It is also known to be used as a medical herb for respiratory diseases, gastrointestinal problems, and as an anti-inflammatory [4].
Gunnera manicata is one of the largest species of Gunnera native to the Serra do Mar mountains of southeastern Brazil. Leaves range from 1.5 to 2.0 meters long [3] with an average diameter of 2.5m. Some leaves have been measured to be up to 3.3m across when cultivated [5]. In contrast, Gunnera magellanica is a species of Gunnera native to Chile, Argentina, the Falkland Islands, Peru, and Ecuador. The leaves of G. magellanica only grow 3-4 cm in diameter and leaf stalks reach 5-6 cm tall [6].
Cyanobacteria
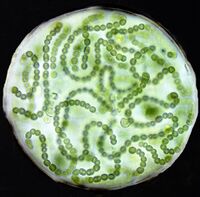
Cyanobacteria are the largest phylum of gram positive prokaryotes on earth dating back to 3.5 billion years ago [9]. They evolved the ability to use photosynthesis to obtain their energy, making them different from other single celled organisms at the time [10]. By photosynthesizing, the Cyanobacteria produce oxygen as a byproduct. This is believed to have both helped the bacteria successfully prosper because the environment in which they were developing was otherwise occupied by anaerobic bacteria and caused the Great Oxidation Event [11]. The Great Oxidation Event is where the earth first experiences a rise in the amount of oxygen in the atmosphere and oceans [12].
Many species of Cyanobacteria live in large colonies of cells ranging from hundreds to thousands of cells in a single colony [13]. These colonies are able to form filaments, sheets, or hollow spheres [14]. When too large, they can form algal blooms that can cause great harm to the aquatic ecosystem and the surrounding area in which it lives [15]. Cyanobacteria can be found in almost any terrestrial or aquatic environment ranging from oceans and lakes to extremes like wet rocks in a desert or in the Antarctic [16].
Cyanobacteria have been shown to make an important contribution to many biological cycles around the globe. Planktonic Cyanobacteria, specifically, are key sources of food in the aquatic food web. Cyanobacteria also are essential in contributing to major global biogeochemical cycles [14]. They have been shown to be responsible for a large portion of the earth’s N2 and CO2 fixation and fluctuations. N2 fixation is a process where the Cyanobacteria can convert the nitrogen gas into ammonia, nitrites, and nitrates which can then be used by plants to make proteins and nucleic acids [17]. In CO2 fixation, Cyanobacteria have large protein-shell bacterial microcompartments called carboxysomes which have been seen to take up inorganic carbon and efficiently “fix” it into organic compounds that can then be used by living organisms [18].
Gunnera and Cyanobacteria symbiosis

Unique Relationship
The filamentous Cyanobacteria genus Nostoc has been observed to form colonies in both terrestrial and aquatic habitats. Nostoc has also been seen to screen damaging ultraviolet light and possesses the ability to fix atmospheric N2 [20]. For this reason, scientists believe several Gunnera species have developed a complex symbiotic relationship with Nostoc. This relationship is the only known relationship between a cyanobacterium and an angiosperm [21]. Even more compelling is that Nostoc is the only genus of Cyanobacteria that have been observed to be in a symbiotic relationship with Gunnera based on 16S rRNA analyses [22].
Stages of Symbiosis
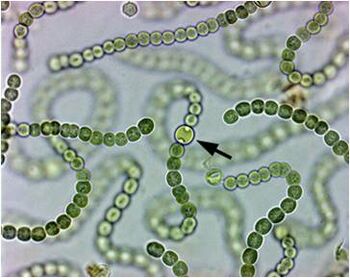
The symbiosis takes place when the Nostoc bacteria enter into the Gunnera by way of specialized gland organs on the stems of the plants [21]. According to research conducted by Johansson and Bergman (1992), establishing the symbiotic relationship between Nostoc and Gunnera takes place in a series of 6 stages: 1) Gland development and mucilage secretion, 2) Collection of Cyanobacteria on the gland, 3) Hormogonia, motile filaments of Cyanobacteria, enter Gunnera, 4) Nostoc can be detected between the cell walls and folds of the Gunnera, 5) Nostoc enters the host cells, and 6) Nostoc cells differentiated into heterocysts [24]. A small red gland develops on the Gunnera right below the base of the leaf when the plant is experiencing nitrogen deprivation, possibly using the C/N-sensing mechanism as a way of regulation. The growth of the gland is further sped up if the plant is also experiencing surroundings with high levels of carbon from sucrose. The gland can be found as early as the cotyledon stage of the plant’s development [25]. The glands then secrete a solution that attracts the Nostoc [26]. The solution is mucilage that has low levels of soluble sugars that attracts the Nostoc to the Gunnera [27]. Although other bacteria are attracted to the mucilage as well, only the Nostoc are able to enter the glands. Within three days of the first detection of the hormogonia on the mature gland, the colony of Nostoc is established. Because of the nature of Nostoc to follow previous paths of other hormogonia, more Nostoc are possibly also enticed to invade the plant as well [25]. The glands of the Gunnera consist of nine papillae surrounding one central papillae [25]. Between the papillae, the Nostoc are able to enter the cell by way of channels from which the mucilage is released. While moving down into the Gunnera, the Nostoc moves in between the cell walls of the plant cells. The cell walls of the Gunnera are then dissolved where Nostoc is in close proximity. This allows the Nostoc to “invade” the cells [24]. Nostoc then enters the cells of the Gunnera where it produces N2-fixing cells [21]. These cells are known as heterocysts which are thick walled, specialized N-fixing cells [28]. The nitrogen fixation allows the Gunnera to grow and thrive in its environment [29].
Benefits
Previous studies have revealed that Gunnera is unable to use nitrate because it lacks the enzyme nitrate reductase. Nostoc is able to fix nitrogen to fill the needs of the Gunnera [30]. In return, the Nostoc gets reduced carbon from the plant that it uses for nutrients since it is unable to photosynthesize due to lack of sunlight. The plant also provides the bacteria with a habitat where it can thrive [29]. With the cells of the Gunnera now filled with the Nostoc, the plant must control the numbers of bacteria to avoid overgrowth through monitoring the exchange of nutrients to the bacteria [31].
Genetic Changes in Nostoc
Once the Nostoc has successfully “invaded” the Gunnera, gene transcription changes happen within the Nostoc where genes associated with heterocyst differentiation and nitrogen fixation are amplified. The hetR gene frequency, which is a gene involved in regulating heterocyst differentiation, is increased. hetR is positively correlated with heterocyst frequency increases and ntcA expression which codes for nitrogen-responsive transcription factors. nifH, which codes for dinitrogen fixation, continues to be high as observed in bacteria that are not in an endosymbiotic relationship with Gunnera, however, glnB (PII) decreases when the bacteria is in the symbiotic relationship [32].
Conclusion
A Cyanobacterium is a photosynthesizing bacterium that is thought to have caused the Great Oxidation Event where the earth's atmosphere and oceans had it first rise in oxygen levels making the life present today possible. Gunnera is a genus of angiosperm which grows in the southern hemisphere that lacks the ability to produce its own nitrogen it need to survive. To compensate for that lack in ability, Gunnera has evolved a symbiotic relationship with Nostoc, a genus of Cyanobacteria that has nitrogen fixation ability, to obtain nitrogen. Though the symbiosis, the Nostoc acquire a protected area to live and obtain carbon from the Gunnera since it is no longer able to photosynthesize while living in the plant.
References
- ↑ 1.0 1.1 1.2 Gunneraceae: gunnera. Phylogeny of New Zealand Plants. (n.d.). Retrieved November 24, 2021, from https://plantphylogeny.landcareresearch.co.nz/webforms/viewtree.aspx?ObjectID=e6155e0d-5659-4813-86f7-70a5f1c250ca.
- ↑ Wanntorp, L., Wanntorp, H. E., Oxelman, B., & Källersjö, M. (2001). Phylogeny of Gunnera. Plant systematics and evolution, 226(1), 85-107.
- ↑ 3.0 3.1 Gunnera Explained. (n.d.). Retrieved November 24, 2021, from http://everything.explained.today/Gunnera/.
- ↑ Molares, S., & Ladio, A. (2014). Medicinal plants in the cultural landscape of a Mapuche-Tehuelche community in arid Argentine Patagonia: an eco-sensorial approach. Journal of ethnobiology and ethnomedicine, 10(1), 1-14.
- ↑ 5.0 5.1 BBC. (2011, October 14). Abbotsbury Gardens celebrates plant's 'monster' leaves. BBC News. Retrieved November 24, 2021, from https://www.bbc.com/news/uk-england-berkshire-15308919
- ↑ Söderbäck, E., Lindblad, P., & Bergman, B. (1990). Developmental patterns related to nitrogen fixation in the Nostoc-Gunnera magellanica Lam. symbiosis. Planta, 182(3), 355-362.
- ↑ https://upload.wikimedia.org/wikipedia/commons/0/07/Devils_Strawberry_%283278834731%29.jpg
- ↑ Nostoc Vaucher ex Bornet et Flahault. Cyanobacteria. (n.d.). Retrieved December 8, 2021.
- ↑ Schopf, J. W., & Packer, B. M. (1987). Early Archean (3.3-billion to 3.5-billion-year-old) microfossils from Warrawoona Group, Australia. Science, 237(4810), 70-73.
- ↑ Sinha, R. P., & Häder, D. P. (2008). UV-protectants in cyanobacteria. Plant Science, 174(3), 278-289.
- ↑ Whitton, B. A. (Ed.). (2012). Ecology of cyanobacteria II: their diversity in space and time. Springer Science & Business Media.
- ↑ Lyons, T. W., Reinhard, C. T., & Planavsky, N. J. (2014). The rise of oxygen in Earth’s early ocean and atmosphere. Nature, 506(7488), 307-315.
- ↑ Tamulonis, C., Postma, M., & Kaandorp, J. (2011). Modeling filamentous cyanobacteria reveals the advantages of long and fast trichomes for optimizing light exposure. PLoS One, 6(7), e22084.
- ↑ 14.0 14.1 Aguilera, A., Klemenčič, M., Sueldo, D. J., Rzymski, P., Giannuzzi, L., & Martin, M. V. (2021). Cell death in Cyanobacteria: current understanding and recommendations for a consensus on its nomenclature. Frontiers in Microbiology, 12, 416.
- ↑ Paerl, H. W., & Otten, T. G. (2013). Harmful cyanobacterial blooms: causes, consequences, and controls. Microbial ecology, 65(4), 995-1010.(Pearl and Otten)
- ↑ De Los Ríos, A., Grube, M., Sancho, L. G., & Ascaso, C. (2007). Ultrastructural and genetic characteristics of endolithic cyanobacterial biofilms colonizing Antarctic granite rocks. FEMS microbiology ecology, 59(2), 386-395.
- ↑ Adams, D. G., & Duggan, P. S. (2008). Cyanobacteria–bryophyte symbioses. Journal of Experimental Botany, 59(5), 1047-1058.
- ↑ Hill, N. C., Tay, J. W., Altus, S., Bortz, D. M., & Cameron, J. C. (2020). Life cycle of a cyanobacterial carboxysome. Science advances, 6(19), eaba1269.
- ↑ Adams D.G., Bergman B., Nierzwicki-Bauer S.A., Duggan P.S., Rai A.N., Schüßler A. (2013) Cyanobacterial-Plant Symbioses. In: Rosenberg E., DeLong E.F., Lory S., Stackebrandt E., Thompson F. (eds) The Prokaryotes. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-30194-0_17.
- ↑ Dodds, W. K., Gudder, D. A., & Mollenhauer, D. (1995). The ecology of Nostoc. Journal of Phycology, 31(1), 2-18.
- ↑ 21.0 21.1 21.2 Bergman, B., Johansson, C., & Söderbäck, E. (1992). The Nostoc–Gunnera symbiosis. New Phytologist, 122(3), 379-400.
- ↑ Rasmussen, U., & Svenning, M. M. (2001). Characterization by genotypic methods of symbiotic Nostoc strains isolated from five species of Gunnera. Archives of microbiology, 176(3), 204-210.
- ↑ Haneef, J. (2018, October 14). Heterocyst of Nostoc structure and function. Plant Science 4 U. Retrieved December 8, 2021, from https://www.plantscience4u.com/2018/10/heterocyst-of-nostoc-structure-and-function.html.
- ↑ 24.0 24.1 Johansson, C., & Bergman, B. (1992). Early events during the establishment of the Gunnera/Nostoc symbiosis. Planta, 188(3), 403-413.
- ↑ 25.0 25.1 25.2 Chiu, W. L., Peters, G. A., Levieille, G., Still, P. C., Cousins, S., Osborne, B., & Elhai, J. (2005). Nitrogen deprivation stimulates symbiotic gland development in Gunnera manicata. Plant Physiology, 139(1), 224-230.
- ↑ Pawlowski, K., & Bergman, B. (2007). Plant symbioses with Frankia and cyanobacteria. In Biology of the Nitrogen Cycle (pp. 165-178). Elsevier.
- ↑ Khamar, H. J., Breathwaite, E. K., Prasse, C. E., Fraley, E. R., Secor, C. R., Chibane, F. L., ... & Chiu, W. L. (2010). Multiple roles of soluble sugars in the establishment of Gunnera-Nostoc endosymbiosis. Plant physiology, 154(3), 1381-1389.
- ↑ Borowitzka, M. A. (2018). Biology of microalgae. In Microalgae in health and disease prevention (pp. 23-72). Academic Press.
- ↑ 29.0 29.1 Gautam, K., Rajvanshi, M., Chugh, N., Dixit, R. B., Kumar, G. R. K., Kumar, C., ... & Dasgupta, S. (2021). Microalgal applications toward agricultural sustainability: Recent trends and future prospects. Microalgae, 339-379.
- ↑ Sprent, J.I. (2005). NITROGEN IN SOILS | Symbiotic Fixation. In Encyclopedia of Soils in the Environment(pp. 46-56). Elsevier.
- ↑ Rai, A. N., Söderbäck, E., & Bergman, B. (2000). Cyanobacterium-plant symbioses. New Phytologist, 147(3), 449-481.
- ↑ Wang, C. M., Ekman, M., & Bergman, B. (2004). Expression of Cyanobacterial genes involved in heterocyst differentiation and dinitrogen fixation along a plant symbiosis development profile. Molecular plant-microbe interactions, 17(4), 436-443.
Edited by Rachael Tomasko, student of Joan Slonczewski for BIOL 116 Information in Living Systems, 2021, Kenyon College.

