Hamiltonella defensa
Classification
Domain: Bacteria; Phylum: Proteobacteria; Class: Gammaproteobacteria; Order: Enterobacterales; family: Enterbacteriaceae; Genus: Hamiltonella
Species
Hamiltonella defensa
Description and Significance
Hamiltonella defensa is a host-restricted mutualist symbiont. It is conditionally beneficial, which means that a couple of factors have to be present in order for H. defensa to serve its purpose. Its main purpose is protecting aphids from braconid wasp parasitoids. It is an endosymbiont of aphids, so it lives inside these sap-sucking insects. This bacterium is important because it has the unique ability to defend its aphid host from these invasive parasitoids.
Genome Structure
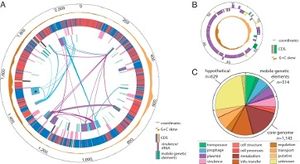
Hamiltonella defensa has an extremely dynamic genome. It is relatively small, only 2.1 Mb circular chromosome. It encodes 2,100 protein-coding genes, has a relatively large number of pseudogenes, and is littered with mobile DNA, insertion sequences, and phage remnants. Horizontal gene transfer plays a role in its dynamic genome.
Additionally, approximately half of H. defensa's DNA comes from toxin-encoding bacteriophages called APSEs. APSEs are similar to lamda-like phages, which are bacterial viruses that infect E. coli. It has other similarities to the E. coli bacterium, including a similar number of pseudogenes. Depending on which strain of APSE H. defensa obtains its DNA from, the development stage at which the parasitoid wasps are killed varies.
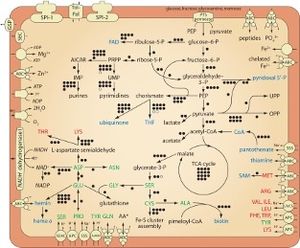
H. defensa is auxotrophic for 8 of its 10 essential amino acids. It relies on Buchnera, a primary endosymbiont of aphids, to synthesize these amino acids. However, despite H. defensa's limited biosynthetic capabilities, it has considerably more cell structural, DNA replication, recombination, and repair genes than do obligate endosymbionts. This is most likely due to its extremely varied sources of DNA. Additionally, its diverse genome allows for a wide variety of regulatory genes which help H. defensa to cope with changes to its environment, such as attack of hosts by parasitoids or an invasion of a new host species.
Cell Structure, Metabolism and Life Cycle
Hamiltonella defensa can be maternally transmitted or passed through horizontal gene transfer. It is found in approximately 34% of aphids. The reason it is not found in a higher percentage of aphids is because its presence can be costly to its host if there is no threat present. Specific costs to a host vary among aphid species, but generally, survivorship and life span were lower in aphids infected with H. defensa when no threat was present.
Additionally, H. defensa can conduct glycolysis on its own, which is a very unique ability for endosymbionts. This is important because it allows for energy production, even if H. defensa is living in oxygen-limited environments.
Protective Mechanism
As previously mentioned, Hamiltonella defensa has the ability to kill braconid wasp parasitoids. The exact mechanism by which it does this is unknown. Some proposed theories are that the phages could be directly poisoning the wasps or that the phages could be splitting the Hamiltonella apart causing their toxins to spill over the wasps, or a combination of the two. In order for this bacterium to be beneficial to its aphid host, a parasite threat has to be present and the bacteriophage APSE DNA has to be a part of H. defensa's genome. This is why it is conditionally beneficial. Without these two conditions present, H. defensa is useless to its host.
References
6. Yong, Ed. I Contain Multitudes: the Microbes Within Us and a Grander View of Life. Ecco, an Imprint of HarperCollinsPublishers, 2016.
Author
Page authored by Isabella Valli, student of Prof. Jay Lennon at IndianaUniversity.