Hepatitis E Virus: Difference between revisions
No edit summary |
No edit summary |
||
| Line 4: | Line 4: | ||
[[Image:OULOGOBIANCO.JPEG|thumb|200px|left|University of Oklahoma Study Abroad Microbiology in | [[Image:OULOGOBIANCO.JPEG|thumb|200px|left|University of Oklahoma Study Abroad Microbiology in | ||
Arezzo, Italy[http://cas.ou.edu/studyabroad/]]] | Arezzo, Italy[http://cas.ou.edu/studyabroad/]]] | ||
[[Image:Disease 0721.jpg|thumb|250px|right|Structure of HEV Decoded Through X-ray Crystallography[http://www.medgadget.com/2009/07/structure_of_hep_e_decoded_through_xray_crystallography.html]]] | |||
==Etiology/Bacteriology== | ==Etiology/Bacteriology== | ||
===Taxonomy=== | ===Taxonomy=== | ||
Revision as of 19:48, 29 July 2015
[[Category:Pages edited by students of Tyrrell Conway at the University of Oklahoma]]

Etiology/Bacteriology
Taxonomy
genus: Hepevirus
Description
HEV is the leading cause of acute viral hepatitis in the world, especially in developing countries.Hepatitis E virus (HEV) was first discovered and accurately documented as a large acute outbreak in New Delhi around 1955. At the time they just called it enteric non-A, non-B hepatitis.[8] it wasn't until the 90s when its genome was cloned, studied and identified that it was named Hepatitis E.[9] is a non-enveloped, positive-sense, single-stranded ribonucleic acid (RNA) virus.[1] It is also the only virus within the genus Hepevirus and the family Hepeviridae.[2] HEV is one of the five identified hepatitis viruses (A,B,C,D, E) . Hepatitis E is most similar to hepatitis A, in that they are both transmitted through contaminated food or water. The other types of hepatitis(B,C,D) are transmitted through infected blood, sexual contact and from mother to child. HEV is the causative agent of the infectious disease Hepatitis E. HEV is unique in that it displays different clinical and epidemiologic characteristics depending on where the infection is acquired which is mainly due to the four viral genotypes of HEV that have been identified.[3] Every year there are 20 million hepatitis E infections, with over 3 million symptomatic cases of hepatitis E, and 56 600 hepatitis E-related deaths.[4]
Epidemiology
HEV has been found in all regions but has been more prevalently identified in certain parts of the world. The main factor identified in the transmission of HEV is through contaminated water and food. Hence, HEV outbreaks are common in developing countries that lack water supply and environmental sanitation. Although uncommon, cases of hepatitis E caused by HEV have been found in developed countries such as the United States ,usually by individuals who have traveled to areas where HEV is commonly an issue.[3] HEV can rarely happen in single cases or also in large epidemics. The epidemics occurs in areas where lots of people are using a common source of fecally contaminated water.
Pathogenesis
Molecular virology of HEV
HEV consists of a positive-sense, single-stranded RNA molecule that contains three distinct open reading frame. The HEV genome is arranged in three overlapping open reading frames (ORF) (ORF1, ORF2, ORF3). The three coding frames are used to express different proteins which help to regulate the host cell environment. ORF1 encodes a polyprotein that undergoes post-translational cleavage into multiple nonstructural proteins required for virus replication, including a methyltransferase, a putative papain-like cystein protease, an RNA helicase and an RNA-dependent RNA polymerase. ORF2 is located at the 3’-end of the genome and encodes the principal structural protein, the capsid protein. ORF3 begins with the last nucleotide of ORF1 and encodes for a small immunogenic 123 amino acid phosphoprotein which associates with the cytoskeleton, suggesting its possible role in the assembly of virus particle.[12 Its important to note that ORF3 is also very important in HEV pathogenesis due to its three broad functions which are.One is promotion of cell survival. It does this by binding and therefore activating MAP kinase and inactivating its cognate phosphatase. Another function of ORF3 is modulation of the acute phase response by localizing to early and recycling endosomes, and inhibiting the movement of activated growth factor receptors to late endosomes. Lats ORF 3 causes an immunosuppression reaction in the host cell. Its able to do this by promoting the secretion of α1-microglobulin, an immunosuppressive protein that could act in the immediate vicinity of the infected cell. [13]
transmission
Although uncommon and not usually associated with the characteristics of transmission of HEV studies have found the rare but possible zoonotic transmission of HEV. For example one study reported direct transmission from deer to human by the consumption of raw deer meat.[14] Also genotyping data indicate clustering of swine and human HEV strains from patients in the USA, Japan and other countries The zoonotic transmission of HEV is a serious issue in developed countries. Though a majority of infections are asymptomatic, sustained transmission can lead to the evolution of virulent strains in future. [13]
Infectious dose, incubation, and colonization
The Incubation period of HEV is 15 to 60 days with a mean of 40 days.[11] The infections may be asymptomatic
virulence factor
No specific virulence factor has been associated with the human or animal strains of HEV.[5]
Genotypes
HEV has four distinct types of genotypes that different epidemiological and clinical characteristics.
Genotype one: is found in Africa and Asian. It's transmitted through water-borne fecal- oral and also person-to-person. Although person-to-person transmission of HEV is is extremely uncommon there is a few studies that show its possibility in genotype one. The occurrence of outbreak of the genotype one is common.
Genotype two: genotype is found in Mexico and and West Africa. It too is transmitted through water-borne fecal- oral. The occurrence of genotype two is smaller scale outbreaks.
Genotype three geographic location is in developed countries. The route of transmission is food-borne. The occurrence of genotype three is very uncommon. Genotype three: is the only one of the four that can cause chronic infection.
Genotype four: occurs in China, Taiwan, and Japan. Its transmission route is also food-borne and its occurrence is rare (like genotype three). There is studies also suggesting that genotype three and four can be passed between humans and animals. So these two genotypes could have the ability of zoonotic spread of the virus. This was concluded from a study in which HEV RNA (genotypes 3 and 4) had been extracted from pork, boar, and deer meat. Infection could occur in the consumption of infected animals.[3]
Clinical features
The clinical symptoms are typically of acute viral hepatitis including jaundice, malaise, weight-loss and loss of appetite, nausea, abdominal pain, fever and hepatomegaly(enlarged liver).
Diagnosis
symptoms are indistinguishable from any acute hepatic illness so to diagnose suspected in outbreaks of waterborne hepatitis occurring in developing country, in pregnant women (The mortality rate of HEV infection is higher than HAV infection in pregnant women (20%)), if hepatitis A has been excluded. The commonly used tests for HEV infection include detection of IgM and IgG anti-HEV antibodies and detection of HEV RNA.
Treatment
Hepatitis E is self-limiting and doesn't result in chronic hepatitis.[6] This means it will resolve on its own and have no long term effect. Therefore treatment should be supportive to provide management of the symptoms.
Prevention
Several vaccines for HEV are under development. A vaccine for HEV that uses recombinant baculoviruses was developed and clinically tested on human with a 95% success rate.[7] There is no information on when this vaccine will be in the market. Until a vaccine is approved for use, prevention includes not consuming contaminated food and water, avoiding travel to areas where HEV is prevalent and sustaining a general sanitary environment. When water contamination seems to be the cause of an epidemic measures to filter the water of HEV include chlorination of water supplies. Also Iodinated disinfectants or autoclaving destroys the virus.[6
Host immune response
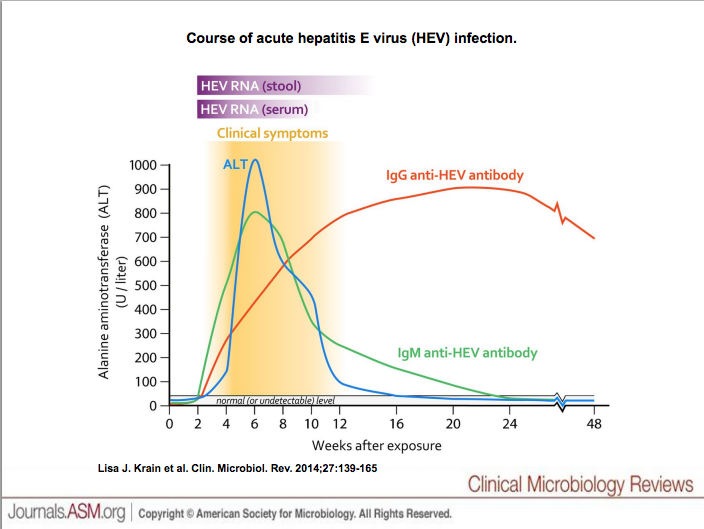
The host immune response can be broken down into two parts, the humoral and cellular immune responses to HEV, and the antibodies produced in response infection.
Incubation period of HEV usually lasts roughly 4 to 6 weeks from infection to onset of symptoms. Figure 1 helps to better understand the host immune response to HEV. ALT(depicted in blue on the figure) is serum of alaine transaminase that is found in the body. If the liver is damaged or not functioning properly, ALT is released into the blood causing ALT levels to increase. So blood could be drawn and tested for ALT levels. This test is used in clinical settings to evaluate liver function.We know that the onset of clinical symptoms of HEV greatly affect the liver, causing inflammation (). So this would result in a sharp rise of serum alanine transaminase (ALT) levels. Symptoms may persist for a few weeks to a month or more. ALT levels return to normal during recovery time.
The host produces two isotypes, IgG and IgM, antibodies in response to HEV infection.IgG has many functions while IgM antibodies function to activate the classical compliment cascade mechanism of the inmate immune response. IgG can be found throughout the body, while IgM can be found in the blood stream. As we see in figure one Anti-HEV IgM increases rapidly and then slowly decreases over the weeks after infection occurs. On the other hand, anti-HEV IgG antibody continue to rise, even during the recovery period, when IgM is decreasing. We can see that detectable anti-HEV IgG may persist for months to years.[15
References
1."Hepatitis E." WHO. Web. 25 July 2015. < http://www.who.int/mediacentre/factsheets/fs280/en/>
2. Purcell, R.H. and Emerson, S.U. Hepatitis E: an emerging awareness of an old disease. J Hepatol. 2008; 48: 494–503
3."Hepatitis E FAQs for Health Professionals." Centers for Disease Control and Prevention. Centers for Disease Control and Prevention, 31 May 2015. Web. 25 July 2015. <http://www.cdc.gov/hepatitis/hev/hevfaq.htm>.
4..Rein DB, Stevens GA, Theaker J, Wittenborn JS, Wiersma ST. The Global Burden of Hepatitis E Virus Genotypes 1 and 2 in 2005. Hepatology, Vol. 55, No. 4, 2012: 988-997
5.Fayer R, Orlandi P, Perdue ML. 2009. Virulence factor activity relationships for hepatitis E and Cryptosporidium. J. Water Health 7(Suppl 1):S55–S63 doi:10.2166/wh.2009.044 [PubMed]
6. Smith J L 2001 A review of hepatitis E virus; J. Food. Prot. 64 572–586
7. Shrestha M P, Scott R M, Joshi D M, Mammen M P, Thapa G B, Thapa N, Myint K S, Fourneau M, et al 2007 Safety and efficacy of a recombinant hepatitis E vaccine. N. Engl. J. Med. 356 895–903 <http://www.ncbi.nlm.nih.gov/pubmed/17329696>
8. Vishwanathan R 1957 Infectious hepatitis in Delhi (1955–56). A critical study: epidemiology; Indian J. Med. Res. 45 49–58
9.ReyesGR,PurdyMA,KimJP,LukKC,YoungLM,FryK E and Bradley D W 1990 Isolation of a cDNA from the virus responsible for enterically transmitted non-A, non-B hepatitis; Science 247 1335–1339 <http://www.ncbi.nlm.nih.gov/pubmed/2107574>
10. Ticehurst JR. Hepatitis E Virus. In: Murray PR, Baron EJ, Pfaller MA, Tenover FC, and Yolken RH, eds. Manual of Clinical Microbiology, 7th ed. Washington DC, American Society for Microbiology Press, 1999:1053-1069.
11.62. Chauhan A, Dilawari J, Chawla Y, Jameel S, Kaur U, Ganguly N. Hepatitis E virus transmission to a volunteer. Lancet. 1993;341:149–50.
12. Bradley DWJ Hepatol.Hepatitis E virus: a brief review of the biology, molecular virology, and immunology of a novel virus. 1995; 22(1 Suppl):140-5. <http://www.ncbi.nlm.nih.gov/pubmed/7602068>
13. Chandra, Vivek, Shikha Taneja, Manjula Kalia, and Shahid Jameel. "Molecular Biology and Pathogenesis of Hepatitis E Virus." J Biosci Journal of Biosciences<http://www.ncbi.nlm.nih.gov/pubmed/19208971>
14. Tei S, Kitajima N, Ohara S, Inoue Y, Miki M, Yamatani T, Yamabe H, Mishiro S et al 2004 Consumption of uncooked deer meat as a risk factor for hepatitis E virus infection: an age- and sex- matched case-control study; J. Med. Virol. 74 67–70 <http://www.ncbi.nlm.nih.gov/pubmed/15258970>
15. American Society for Microbiology. Clin. Microbiol. Rev. January 2014 vol. 27 no. 1 139-165.1 January 2014 <http://cmr.asm.org/content/27/1/139.full>
