Human Papillomavirus and Cervical Cancer
Introduction
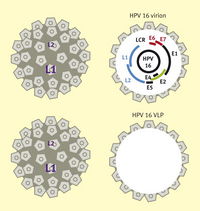
Human papillomavirus (HPV) is a non-enveloped protein capsid containing eight genes in a circular, double-stranded DNA genome (Figure 1). The genes are characterized as either early (E) or late (L) genes. The names are related to when the genes are expressed; e.g. early genes are produced early in the virus life cycle. The early genes are associated with DNA replication, regulatory functions, and activation of the lytic life cycle, while the late genes are involved with production of the viral capsid parts.
There are over 100 types of HPV, which cause almost all cases of cervical cancer and can also cause cancer of the head and neck, vulva, vagina, penis, and anus. Additionally, it can cause several types of warts, including genital warts. Anogential neoplasia--unusual growth of cells--is caused by high-risk (HR) types, which include types 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68, 73, and 82. These types can integrate their DNA into the host genome, which in some cases, can lead to unregulated cell proliferation. The two most common types are 16 and 18, which cause 70% of cervical cancer cases.4 Low-risk (LR) types are more associated with anogential warts.
Recent research of HPV pertains to developing effective testing, vaccines, and treatment of HPV.
Life Cycle
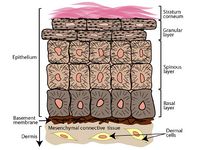
The progression of the life cycle of HPV depends on several different factors, such as: type of host cell, type of HPV, or host immune system. HPV can only infect parabasal or basal cells--undifferentiated stem cells of the epidermis--most commonly in the transformation zone of the cervix, which is adjacent to the border of the endocervix and ectocervix.16 This area becomes more accessible during puberty, pregnancy, and when using oral contraceptives.19 Infection may be facilitated by micro abrasions on the surface; this allows better access to the basal cells, which are usually below several layers of epithelial cells (Figure 2).
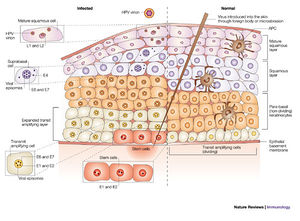
As the host cell undergoes the normal differentiation, the virus undergoes different parts of its life cycle (Figure 3). When the virus initially enters host basal cells, it cannot replicate until the cell matures into a keratinocyte.13 The virus has to wait until mitosis occurs in order to use the host's replication machinery. The virus expresses the early proteins--E1, E2, E5, E6, and E7--in the lower spinous layers (earlier in the infection).16 Normally, as epithelial cells mature the cell cycle is halted as part of forming a protective barrier; however, terminal differentiation is hindered by E7 and E6. This has most likely evolved to allow the host cell to continue to reproduce viruses. As the host cell life progresses to the upper spinous layer, gene expression of HPV changes. The late proteins--L1 and L2--and E4 are upregulated.16 At this point virus assembly occurs and exfoliating cells release complete virions. These cells are resilient in dry environments and virions shed from cornified squames--cells that have more keratin, a protective agent that hardens the cell--have a higher chance of survival.3,10
Each HPV gene contributes to the pathogenesis of the virus. E1 is essential for viral replication and is linked to genome maintenance. E2 is involved with regulation of viral replication and transcription. This regulation is both positive and negative, which is achieved through interactions with promoters located in the upstream regulatory region.16 E5 interacts with cell membrane growth factors and is linked to the down regulation of major histocompatibility complex (MHC) class I molecules. E6 and E7 are oncogenes that are involved in host cell proliferation. Although E4 is part of the early region, it is expressed later in the virus life cycle.16 The exact role E4 plays is not clear, but it interacts with the cytoskeleton proteins, more specifically the keratin intermediate filaments in epithelial cells.16 L1 encodes the major and L2 encodes the minor portions of the capsid. L2 is expressed before L1 to allow for proper construction of the capsid. L1 is known for self-assembling into virus-like particle, but it is thought that L2 may improve infectivity or packaging.11,15 These genes facilitate the entry into the host cell and can differ from type to type.6 The genes and corresponding functions are summarized in Table 1.
The exact mechanism the virus uses to enter the host cell is still unclear, but it is known that the virus enters through endocytosis. Data has suggested that this pathway may be facilitated through clathrin- or caveola-mediated endocytosis (Bousarghin et al. 2003). The virus finishes entering the cell by membrane fusion then uncoating and breakdown of the capsid. The genome then enters the nucleus so that it can use the host’s DNA and RNA polymerases.13 Once inside the cell the DNA is present at a low level as an episome, but once the host cell matures the viral genome can replicate. After the genes are transcribed, they exit the nucleus and undergo processing. In the case of the capsid proteins later in the life cycle, they re-enter the nucleus for assembly after which they exit the cell completely.
There are data that detect HPV in cervical lesion without any apparent disease; this provides evidence that HPV has a latent stage.6 A few infected basal cells undergoing normal epithelial differentiation could explain detection without any visible cause. Once the immune system declines the HPV infection can resume normal activity and reproduce.6 It is suggested that the virus achieves this state by highly expressing E1 and E2, to regulate viral gene expression especially, that of E6 and E7.20
Cancer Mechanism
If an individual were to become infected with HPV, it does not necessarily mean that individual would contract cervical cancer even with infection of high-risk types, which are associated with cancer only because they are more prone to cause cell proliferation. Integration of HPV’s genome into the host is connected with neoplasia--unusual growth of cells. This could be caused by proliferation of cells and subsequent loss of DNA repair mechanisms, which leads to further accumulation of chromosomal mutations.16
The two genes highly associated with carcinogenic activity are E6 and E7. It is commonly thought that these work by interaction with two different target genes. The mechanism of E7 is well established. It is hypothesized that E7 binds to the retinoblastoma protein (pRb), which inhibits its normal function.6,16,17 During normal cell function, pRb binds to E2F, which is a transcription factor that can activate oncogenes that contain sequences for cyclin E and c-Myc proteins. Cyclin E is needed to advance the cell to mitosis and c-Myc is needed for the mitosis process.13 If production of these proteins is uninterrupted by pRb, then the cell continues to divide unregulated. The binding of E7 to pRb also promotes degradation of pRb by the cell’s proteasomes, which are large protein complexes that degrade proteins.13 Both of these outcomes result in cell proliferation. The mechanism for E6 is simpler. The protein E6 binds to p53 and inactivates it.16 The gene p53 has several roles that regulate the cell cycle, induce apoptosis, promote DNA repair, and prevent tumor growth. Inhibition of p53 would stop these functions and could cause unregulated cell proliferation and cancer.
Although E6 and E7 genes are associated with carcinoma of HPV, they are not the sole cause. There are detectable levels of E6 and E7 in non-cancerous infections. The integration of HPV genome into host genome is connected with cancer. This could be due to the loss of E2 during integration into the host genome.17,19 E2 encodes proteins that regulate viral gene expression, if E2 does not regulate the expression of E6 and E7, then the cell goes can proliferate and cause cancer. It should be noted that a nonsense or missense mutation of E2 could also cause the same problem.
HPV and the Immune System
It has been found that there is a higher rate of neoplasia associated with HPV infections in immuno-compromised individuals, such as HIV-positive individuals.9 This discovery opens the door to the role of the immune system in differences between benign HPV infections and development of anogenital neoplasia.
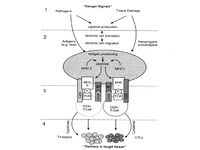
Normally, when a foreign agent infects a cell, the cell invokes a complex system to activate immune responses in order to protect the organism (Figure 4). It starts with the construction of danger signals shortly after a cell is infected. These danger signals produce anigens, which are processed and create peptides. These peptides cause MHC class I or II to react with CD4+ or CD8+ T-cells, respectively. CD4+ and CD8+ T-cells generate interleukins (ILs), which activate larger immune responses.16
Evidence has suggested that HPV progresses successfully, because it does not induce immune system responses for most of its life cycle. This is due to several different factors. First, keratinocytes, the target cell for HPV, are less likely to incite a novel T-cell response.19 Second, HPV produces genes that suppress part of the human immune system. The protein encoded by E5 down regulates MHC class I molecules.19 MHC class I molecules present fragments of invading elements in order for a T-cell to detect and illicit an immune response. Third, the immune system has better detection in the lower layers of the epithelium, but in those cells, HPV produces low levels of detectable elements, such as capsid proteins.19 Capsid proteins are produced in the upper layers of the epithelium, which is far away from immune system response elements. Fourth, a HPV infection does not involve the blood steam, which means the mechanisms for the immune system to detect an infection are limited.19 Finally, a HPV infection does not cause host cells to lyse or die, which would induce normal danger signals.19
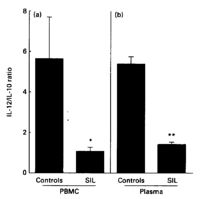
Data presented by Jacobs et al. (1997) suggests that an augmented immune response is correlated with HPV infection in squamous intraepithelial lesions (SILs). They found a change in IL-12/IL-10 ratios from blood samples of HPV uninfected and infected women (Figure 5). IL-10 suppresses immune responses by inhibiting IL-12 production. IL-12 is associated with the progression of immune responses against tumor growth.8 This means that an infection by HPV is connected with a decline in immune responses against cancerous cells. Unfortunately, the data does not demonstrate causality of this observation.
Testing
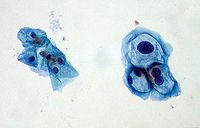
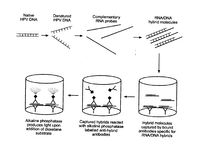
HPV infections can be indicated by koilocytes (Figure 6), which are indicated by nuclear enlargement, irregularities of the nuclear membrane, and a halo effect around the nucleus.19 They are not always present and could be caused by another source. The key method for verifying a HPV infection is by indentifying the presence of HPV DNA in host cells. There is only one FDA-approved commercially available product for testing for cancerous HPV, the Hybrid Capture II by Qiagen.5 The cells collected from a pap smear are put through a “hybrid capture” test. These tests detect DNA from 18 most prevalent types of HPV that affect the cervix (13 high-risk types and 5 low-risk types). The test includes RNA probes with complementary sequences to HPV DNA sequences. The test is positive when a certain level of hybridized RNA-DNA is achieved. The test does not specify what type is present and the high and low-risk testing are done separately.19
Hybrid Capture II starts by lysing the collected cells (Figure 7).5 Any DNA from the cells is denatured into single stranded DNA. The RNA probes are then added and hybridize with any complementary sequences. This is done under conditions that promote hybridization between RNA and DNA. Antibodies that recognize RNA-DNA strands trap any hybridized strands. This solution is then treated with alkaline phosphatase, which conjugates with the antibodies. The mixture is then washed to remove any excess reagents. Next, dioxetane is added to the mixture, which produces light when it reacts with any present alkaline phosphatase enzyme.5 The light is measured to quantify any HPV DNA from the sample cells.
There are alternative ways of testing, such as polymerase chain reaction-based (PCR-based) systems. These have not been standardized nor are they approved by the FDA. These tests tend to be more sensitive, can identify the specific HPV type, can be used on several different HPV types, and need less sample; however they are time consuming and are easily contaminated because of the sensitivity.19
The PCR process includes cycles of denaturing and annealing by heating and cooling, respectively. These cycles allow the DNA to replicate several times to an appreciable level for testing. Oligonucleotides, short DNA sequences, are added to act as primers for the sample DNA. These sequences are specific to HPV types, which determines the specificity of the test. Contamination can happen if any amplicons are present in reagents or the sample. Amplicons are short DNA sequences that can act as primers for non-target genes, which gives false data. The amplified DNA could either be used for a microplate assay, which does not specify a HPV type, or a line-blot assay, which does give information about the specific type present.5
Triginelli et al. (2006) suggests a promising method of testing for cancerous infections before they become visible lesions. The authors found the telomerase activity was higher in cells from squamous cell carcinomas (SCCs)--the characterization of most cervical cancers--than uterine myomas (Figure 8), which were the control group, because they are benign tumors. Heightened telomerase activity is correlated with immortalized cells and cancerous growths.18 Somatic cells under normal cell division have undetectable or low levels of telomerase activity.18 Consequently, telomerase activity could therefore be used as a marker of cancerous cells, because it is expressed during unregulated cell proliferation but not normal cell division. This method would be preferential, because it is relatively sensitive, uses a small sample size, and most importantly specific to cancerous cells.18 Not all HPV infections are cancerous, but when this method is paired with usual HPV screening, it would be effective in determining if the infection is malignant.
Prophylactic Vaccines
There are two FDA-approved prophylactic vaccines on the market, Gardasil and Cervarix.4 Gardasil protects against HPV types 6, 11, 18, and 16. Types 6 and 8 cause the majority of genital warts. Cervarix protects against HPV types 16 and 18. Both vaccines are given in a three shot regiment over six months.4
Both vaccines were created when it was discovered that L1 capsid proteins were capable of self-assembling into virus-like particles (VLPs) (Figure 9).19 VLPs are structurally the same as HPV, but do not contain DNA, which means the genes involved in pathogenesis are not present. These VLPs cause an immune response that produces anti-virion antibodies. The L1 capsid proteins are unique to different HPV types, which means the vaccines are type specific.
Complications
Prophylactic vaccines can only prevent not treat HPV, which means that it should be administered before sexual activity. Therefore, it is recommended that the vaccine be given to prepubescent girls. Gardasil is approved for use in males to prevent genital warts and anal cancer caused by HPV.4 It, however, has not been studied whether or not there is a link between vaccinating males and a decrease in cervical cancer caused by HPV.19 This option may be ineffective if vaccination in females becomes widespread. Since the development of these vaccines is relatively new, the length of effectiveness has not yet been determined.4 Another risk to the vaccines would be the occurrence of one HPV type would increase with the decrease of the currently more prevalent HPV types.19
Treatment
There is no standard treatment for HPV or cervical cancer. The CDC has stated that 90% HPV infections can clear up in two years. Although it is possible that it may not be gone, but just at low levels that can not be detected.17 In the case of neoplasia, there are some surgical options, which include ablation or excision. These options are not always the most efficient method, because they can leave diseased tissue or remove healthy along with diseased tissue.19 Often the success of these treatments depends on the follow-up. There can also be some corresponding obstetric complications.14 There are some other alternative options such as therapeutic vaccines and anti-viral medications.
Therapeutic Vaccines
Therapeutic vaccines can have many different targets, but most aim to cause cytotoxic T-lymphocytes (CTLs) to respond in order to clear the established infection.16 These CTLs are usually designed to target viral oncogenes antigens of the most frequent high-risk HPVs.
One method is using a prime-boost strategy. This entails priming with plasmid or viral vector DNA followed by a different viral vector encoding the immunogen.16 This method has an improved response in animal models but has problems with generation of CTL activity because of inhibition by T-helper 2-driven humoral immunity.16 Features used by this method to improve immune response include: high-affinty T-cell receptors, stimulation of IL-I2 production, use of multiple antigen exposure to produce affinity maturation of antibodies, specificity, use of non-replicating viral vectors for boosting step to concentrate the specificity of the target antigen, or use of low levels of immunogen to produce better secondary responses.16
Another option for a therapeutic vaccine is the use of recombinant viral vectors. This method can be a problem for people with poor or suppressed immune systems, because generally, it can cause an infection or other problems for the patient, such as central nervous system or skin complications.16 This method should produce CTLs specific to HPV type with no issues with specific characteristics of the patient.16
One method is introducing HPV peptides to produce an immunogenic response. This method is well tolerated, but it has some issues. One issue is that the peptide is restricted by the human leukocyte antigen (HLA) genotype of an individual. It is possible to predict the peptide that would be compatible with the HLA genotype, but not all individuals carry the correct HLA genotype.16
Another option is the use of recombinant proteins. This can be given without considering HLA genotype of an individual and it is relatively well tolerated. It is difficult to produce and purify the proteins. This method uses an adjuvant, which is an additive agent that either stabilizes or increases the immune response. Although, it can be difficult to choose an appropriate adjuvant for the recombinant protein.
Anitvirals
Three antivirals that can be used to treat HPV are cidofovir, imiquimod, and interferon. Cidofovir, an acyclic nucleoside phosphonate, is a broad spectrum antiviral. It works by inducing apoptosis in infected cells. This may happen by lowering the levels of E6 and E7 and increase the levels of p53 and pRb, which suppress tumor growth.19 It is commonly used intravenously and can be used for immunosuppressed individuals.14 Topical use on cervical intraepithelial neoplasia (CIN) has been investigated and shows promising results after three applications of 1% cidofovir gel (Snoeck et al 2000). Imiqimod is an immune response modulator normally used to treat genital warts, but there is some research on using it to treat vulvar intraepithelial neoplasia (VIN).19 Interfereon has shown best results when used intramuscularly as well as intralesionally (Rotola et al. 1995). Interferon is an antiviral immune response that either induces dsRNA endonuclease or acts as eIF2 kinase.13 Interferon is a group of proteins artificially made and isolated by drug companies to serve as an antiviral medication.
References
1. Alonso, L., & Fuchs, E. (2003). Stem cells of the skin epithelium. Proceedings of the National Academy of Sciences of the United States of America, 100(Suppl 1), 11830-11835.
2. Bousarghin, Latifa, Antoine Touze, Pierre-Yves Sizaret, and Pierre Coursaget. (2003) Human Pappilomavirus Types 16, 31, 58 Use Different Endocytosis Pathways to Enter Cells. Journal of Virology, 77(6), 3846-3850.
3. CDC http://www.cdc.gov/
4. Bryan, J.T. and Brown, D.R. (2001). Transmission of human papilloma type 11 infection by desquamated cornified cells. Virology, 281, 35-42.
5. Davies, P., Kornegay, J., & Iftner, T. (2001). Current methods of testing for human papillomavirus. Best Practice & Research in Clinical Obstetrics and Gynaecology, 15(5), 677-700.
6. Doorbar, J. (2005). The papillomavirus life cycle. Journal of Clinical Virology, 32, 7-15.
7. Frazer, Ian H. (2004) Prevention of cervical cancer through papilomavirus vaccination. Nat Rev Immunol, 4(1), 46-55.
8. Jacobs, Giannini, Doyen, Baptista, Moutschen, Boniver, et al. (1998). Inverse modulation of IL-10 and IL-12 in the blood of women with preneoplastic lesions of the uterine cervix. Clinical & Experimental Immunology, 111(1), 219-224.
9. Man, S., & Fiander, A. (2001). Immunology of human papillomavirus infection in lower genital tract neoplasia. Best Practice & Research in Clinical Obstetrics and Gynaecology, 15(5), 701-714.
10. Roden, R.B., Lowy, D.R., Schiller, J.T. (1997) Papillomavirus is resistant to desiccation. J Infect Dis, 176, 1076-1079.
11. Roden, R.B., Day, P.M., Bronzo, B.K., Yutzy, W.H., Yang, Y., Lowy, D.R., et al. (2001) Positively charged termini of the L2 minor capsid protein are necessary for papillomavirus infection. J Virol, 75, 10493-10497.
12. Rotola A, Costa S, Di Luca D, Stefanon B, Villani C, Micheletti L, Montemagno U, Bolis PF, Cassai E. (1995). b-Interferon treatment of cervical intraepithelial neoplasia: a multicenter clinical trial. Intervirology 38:325–331.
13. Slonczewski, Joan L., & Foster, John W. (2010). Microbiology: an evolving science. New York, NY: W.W. Northon & Co. Inc.
14. Snoeck, R., Noel, J., Muller, C., De Clercq, E., & Bossens, M. (2000). Cidofovir, a new approach for the treatment of cervix intraepithelial neoplasia grade III (CIN III). Journal of Medical Virology, 60(2), 205-209.
15. Stauffer
16. Stern, P. L., Faulkner, R., Veranes, E. C., & Davidson, E. J. (2001). The role of human papillomavirus vaccines in cervical neoplasia. Best Practice & Research in Clinical Obstetrics and Gynaecology, 15(5), 783-799.
17. Stoler, M. H. (1996). A brief synopsis of the role of human papillomaviruses in cervical carcinogenesis. American Journal of Obstetrics and Gynecology, 175(4), 1091-1098.
18. Triginelli
19. Tristram, A., & Fiander, A. (2007). Human papillomavirus (including vaccines). Obstetrics, Gynaecology & Reproductive Medicine, 17(11), 324-329.
20. Zhang, P., Nouri, M., Brandsma, J. L., Iftner, T., & Steinberg, B. M. (1999). Induction of E6/E7 expression in cottontail rabbit papillomavirus latency following UV activation. Virology, 263(2), 388-394.
Edited by Erin Armentrout, student of Joan Slonczewski for BIOL 238 Microbiology, 2011, Kenyon College.