Leuconostoc: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{Biorealm Genus}} | {{Biorealm Genus}} | ||
[[Image:Leuconostoc-mesenteroides.jpg|thumb|580px|right|Leuconostoc mesenteroides]] | |||
==Classification== | ==Classification== | ||
| Line 18: | Line 18: | ||
==Description and Significance== | ==Description and Significance== | ||
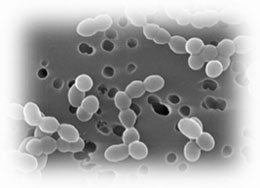
Leuconostocs are traditionally found in association with plant matter, fermenting vegetables, milk, dairy products, and wines and meats. Leuconostocs were first isolated in 1878 by Cienkowski. ''Leuconostoc'' usually nonpathogenic acid-tolerant organisms with optimal temperature 18 and 25°C, but the group is quite diverse. For example, ''L. carnosum'' is an anaerobic bacterium found in spoiled, packaged meat. Optimal temperature for the organism is 2°C. | |||
''Leuconostoc'' is a gram-positive coccus with spherical cell, often lenticular on agar and usually occurs in pairs or chains. ''Leuconostoc'' is nonmotile, not spore forming chemoorganotrophic facultative anaerobe. ''Leuconostoc'' requires rich, complex media. It is catalase-negative nonproteolytic organism without cytochromes. ''Leuconostoc'' is nonhemolytic, vancomycin resistant organism. ''Leuconostoc'' requires nicotinic acid, thiamin, biotin, and pantothenic acid or one of its derivatives. It is heterofermentative (uses a combination of the pentose phosphate and phosphoketolase pathways). Milk is usually not acidified and clotted without added yeast | |||
extract. (Thunnel 1995) | |||
==Genome Structure== | |||
[[Image:Leuconostoc-mesenteroides_genome.png|thumb|300px|left|Leuconostoc mesenteroides genome structure]] | |||
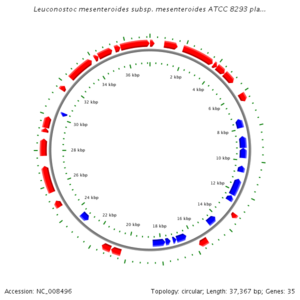
The genome of Leuconostoc is comprised of 2,038,396 bp with 2,075,763 bp nucleotides arranged in a circular design, and encodes for 2005 different proteins, with 54% of them being assigned putative biological functions. As Luconostoc spp. is a lactic acid bacteria, the genome encoded all enzymes present in the 6-phosphogluconate phosphoketolase pathway. Genes were also found that encoded pyruvate dissipating enzymes that are predicted to catalyze the production of many metabolites leading to various end products of fermentation. (F. Breidt Jr.) | |||
==Cell Structure and Metabolism== | |||
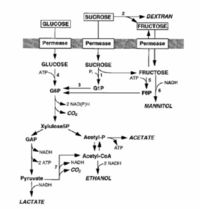
[[Image:Carbon and energy flow.jpg|thumb|200px|right|Schematic representation of carbon and energy flow through the central metabolic pathways of L. mesenteroides during metabolism of various sugars. G1P, glucose-1-phosphate; G6P, glucose-6-phosphate; F6P, fructose-6-phosphate; GAP, glyceraldehyde-3-phosphate; acetyl-P, acetylphosphate; acetyl-CoA, acetyl coenzyme A; 1, sucrose phosphorylase; 2, dextransucrase; 3,phosphoglucomutase (PGM); 4, glucokinase; 5, fructokinase; 6, mannitol dehydrogenase; 7, pyruvate dehydrogenase.]] | |||
''Leuconostoc'' is a gram-positive coccus with spherical cell, often lenticular on agar and usually occurs in pairs or chains. ''Leuconostoc'' is nonmotile, not spore forming chemoorganotrophic facultative anaerobe. ''Leuconostoc'' requires rich, complex media. It is catalase-negative nonproteolytic organism without cytochromes. ''Leuconostoc'' is nonhemolytic, vancomycin resistant organism. ''Leuconostoc'' usually nonpathogenic with optimal temperature 20 to 30’C. ''Leuconostoc'' requires nicotinic acid, thiamin, biotin, and pantothenic acid or one of its derivatives. It is heterofermentative (uses a combination of the pentose phosphate and phosphoketolase pathways). Milk is usually not acidified and clotted without added yeast | ''Leuconostoc'' is a gram-positive coccus with spherical cell, often lenticular on agar and usually occurs in pairs or chains. ''Leuconostoc'' is nonmotile, not spore forming chemoorganotrophic facultative anaerobe. ''Leuconostoc'' requires rich, complex media. It is catalase-negative nonproteolytic organism without cytochromes. ''Leuconostoc'' is nonhemolytic, vancomycin resistant organism. ''Leuconostoc'' usually nonpathogenic with optimal temperature 20 to 30’C. ''Leuconostoc'' requires nicotinic acid, thiamin, biotin, and pantothenic acid or one of its derivatives. It is heterofermentative (uses a combination of the pentose phosphate and phosphoketolase pathways). Milk is usually not acidified and clotted without added yeast | ||
extract. | extract. Size of the cell is usually about 1 micrometer. | ||
==Ecology== | |||
''Caulobacter'' generally live in a dilute aquatic environment where the most common limiting nutrient is phosphorus, an essential element for healthy growth. Lack of this nutrient induce ''Caulobacter'' to dramatically elongate its stalk up to 30 times longer than those in phosphate-rich medium (Brun et al. 2000). | |||
==Cell Division== | |||
''Caulobacter'' asymmetrically divides to produce a motile swarmer cell and a stalk cell. The swarmer cell, which has a flagellum, swims for about 30-45 minutes before shedding the flagellum and differentiating into a stalk cell. The flagellum is ejected from the swamer by the destruction of the structures (MS ring) at the base of the flagellum. Within the swarmer cell, chromosomes do not replicate; however, chromosome replication begins immediately in the daughter cell with the stalk and when the swarmer loses its tail (Stanford). The stalk adheres to surfaces through an adhesive organelle called the holdfast. | |||
Several two-component signal transduction proteins are involved in the cell cycle progression by accumulating at one or both poles "in a spatial and temporal pattern that is reproduced during each cycle" (Jacobs-Wagner 2003). In addition to this, DNA methylation is main component of signaling differentiation. Throughout the cell cycle, the chromosome progressively goes from being fully methylated to hemimethylated during DNA replication - this results in differential binding of regulatory proteins to activate or repress transcription. This was studied by using the CtrA gene, which encodes for an important cell cycle regulatory protein. The CtrA gene has two promoters, one of which "fires early" in the S phase and is recognized by the CcrM DNA methyltransferase. Analysis showed that this P1 promoter is actually repressed by DNA methylation (Reinsenauer and Shapiro 2002). | |||
==References== | |||
[http://jds.fass.org/cgi/reprint/78/11/2514.pdf R. K. Thunnel. 1995. "Taxonomy of the ''Leuconostocs''" ''Journal of Dairy Science'', vol. 78, no. 11. 2514-2522] | |||
[http://www.blackwell-synergy.com/doi/pdf/10.1111/j.1365-2621.2004.tb17874.x F. Breidt Jr. 2004. "A Genomic Study of ''Leuconostoc mesenteroides'' and the Molecular Ecology of Sauerkraut Fermentations" ''Journal of Food Science'', Vol. 69 Issue 1 Page FMS30 January] | |||
Revision as of 18:58, 6 April 2008
A Microbial Biorealm page on the genus Leuconostoc
Classification
Higher order taxa:
Bacteria; Firmicutes; Bacilli; Lactobacillales; Leuconostocaceae
Species:
L. carnosum, L. citreum, L. durionis, L. fallax, L. ficulneum, L. fructosum, L. garlicum, L. gasicomitatum,
L. gelidum, L. inhae, L. kimchii, L. lactis, L. mesenteroides, L. pseudoficulneum, L. pseudomesenteroides
|
NCBI: Taxonomy |
Description and Significance
Leuconostocs are traditionally found in association with plant matter, fermenting vegetables, milk, dairy products, and wines and meats. Leuconostocs were first isolated in 1878 by Cienkowski. Leuconostoc usually nonpathogenic acid-tolerant organisms with optimal temperature 18 and 25°C, but the group is quite diverse. For example, L. carnosum is an anaerobic bacterium found in spoiled, packaged meat. Optimal temperature for the organism is 2°C.
Leuconostoc is a gram-positive coccus with spherical cell, often lenticular on agar and usually occurs in pairs or chains. Leuconostoc is nonmotile, not spore forming chemoorganotrophic facultative anaerobe. Leuconostoc requires rich, complex media. It is catalase-negative nonproteolytic organism without cytochromes. Leuconostoc is nonhemolytic, vancomycin resistant organism. Leuconostoc requires nicotinic acid, thiamin, biotin, and pantothenic acid or one of its derivatives. It is heterofermentative (uses a combination of the pentose phosphate and phosphoketolase pathways). Milk is usually not acidified and clotted without added yeast extract. (Thunnel 1995)
Genome Structure
The genome of Leuconostoc is comprised of 2,038,396 bp with 2,075,763 bp nucleotides arranged in a circular design, and encodes for 2005 different proteins, with 54% of them being assigned putative biological functions. As Luconostoc spp. is a lactic acid bacteria, the genome encoded all enzymes present in the 6-phosphogluconate phosphoketolase pathway. Genes were also found that encoded pyruvate dissipating enzymes that are predicted to catalyze the production of many metabolites leading to various end products of fermentation. (F. Breidt Jr.)
Cell Structure and Metabolism
Leuconostoc is a gram-positive coccus with spherical cell, often lenticular on agar and usually occurs in pairs or chains. Leuconostoc is nonmotile, not spore forming chemoorganotrophic facultative anaerobe. Leuconostoc requires rich, complex media. It is catalase-negative nonproteolytic organism without cytochromes. Leuconostoc is nonhemolytic, vancomycin resistant organism. Leuconostoc usually nonpathogenic with optimal temperature 20 to 30’C. Leuconostoc requires nicotinic acid, thiamin, biotin, and pantothenic acid or one of its derivatives. It is heterofermentative (uses a combination of the pentose phosphate and phosphoketolase pathways). Milk is usually not acidified and clotted without added yeast extract. Size of the cell is usually about 1 micrometer.
Ecology
Caulobacter generally live in a dilute aquatic environment where the most common limiting nutrient is phosphorus, an essential element for healthy growth. Lack of this nutrient induce Caulobacter to dramatically elongate its stalk up to 30 times longer than those in phosphate-rich medium (Brun et al. 2000).
Cell Division
Caulobacter asymmetrically divides to produce a motile swarmer cell and a stalk cell. The swarmer cell, which has a flagellum, swims for about 30-45 minutes before shedding the flagellum and differentiating into a stalk cell. The flagellum is ejected from the swamer by the destruction of the structures (MS ring) at the base of the flagellum. Within the swarmer cell, chromosomes do not replicate; however, chromosome replication begins immediately in the daughter cell with the stalk and when the swarmer loses its tail (Stanford). The stalk adheres to surfaces through an adhesive organelle called the holdfast.
Several two-component signal transduction proteins are involved in the cell cycle progression by accumulating at one or both poles "in a spatial and temporal pattern that is reproduced during each cycle" (Jacobs-Wagner 2003). In addition to this, DNA methylation is main component of signaling differentiation. Throughout the cell cycle, the chromosome progressively goes from being fully methylated to hemimethylated during DNA replication - this results in differential binding of regulatory proteins to activate or repress transcription. This was studied by using the CtrA gene, which encodes for an important cell cycle regulatory protein. The CtrA gene has two promoters, one of which "fires early" in the S phase and is recognized by the CcrM DNA methyltransferase. Analysis showed that this P1 promoter is actually repressed by DNA methylation (Reinsenauer and Shapiro 2002).