Mitochondrial DNA and Type II Diabetes Mellitus
Introduction
1.1 Evolution of Mitochondrial DNA

In 1966, Lynn Margulis wrote a paper entitled “On the Origin of Mitosing Cells”. In her paper, Margulis came up with an influential theory that has been shaping modern science ever since. Although it was hard for her to publish it in magazines, she was determined that the similarity between mitochondria and bacteria was far more meaningful than sheer coincidence.That theory is now famously known as the Endosymbiosis theory. Endosymbiosis explains the origin of eukaryotes. It is suggested that the first step in the origin of eukaryotes from prokaryotes was related to survival in the new oxygen-containing atmosphere: an aerobic prokaryotic microbe (i.e. the protomitochondrion) was ingested into the cytoplasm of a heterotrophic anaerobe. This endosymbiosis became obligate and resulted in the evolution of the first aerobic amitotic amoeboid organisms. By hypothesis, some of these amoeboids ingested certain motile prokaryotes. Eventually these, too, became symbiotic in their hosts. The association of the motile prokaryote with the amoeboid formed primitive amoeboflagellates. In these heterotrophic amoeboflagellates classical mitosis evolved. The evolution of mitosis, insuring an even distribution of large amounts of nucleic acid (i.e. host chromosomes containing host genes) at each cell division, must have taken millions of years. It most likely occurred after the transition to the oxidizing atmosphere, since all eukaryotic organisms contain mitochondria and are fundamentally aerobic. [1]
When the amoeboids ingested the prokaryotes, they also took in the genetic material of the prokaryotes. This genetic material is termed as mitochondrial DNA. A few years after the publication of the paper, scientists were able to study and sequence genomes of different species. In fact, the year 2014 saw more than a thousand new mitochondrial genome sequences deposited in GenBank—an almost 15% increase from the previous year. Hundreds of peer-reviewed articles accompanied these genomes, making mitochondrial DNAs (mtDNAs) the most sequenced and reported type of eukaryotic chromosome. [2]
A cell contains numerous mitochondria, and each mitochondrion contains dozens of copies of the mitochondrial genome. Moreover, the mitochondrial genome has a higher mutation rate (about 100-fold higher) than the nuclear genome. This leads to a heterogeneous population of mitochondrial DNA within the same cell, and even within the same mitochondrion; as a result, mitochondria are considered heteroplasmic. When a cell divides, its mitochondria are partitioned between the two daughter cells. However, the process of mitochondrial segregation occurs in a random manner and is much less organized than the highly accurate process of nuclear chromosome segregation during mitosis. As a result, daughter cells receive similar, but not identical, copies of their mitochondrial DNA.[3]
1.2 The Human Mitochondrial DNA
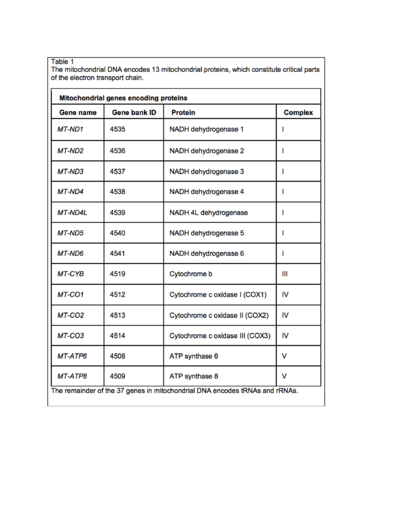
The human mitochondrial DNA was sequenced and the different genes and their products-RNAs and tRNAs- are under constant study. The human mitochondrial DNA (mtDNA) is a double-stranded, circular molecule of 16 569 bp and contains 37 genes coding for two rRNAs, 22 tRNAs and 13 polypeptides. The mtDNA-encoded polypeptides are all subunits of enzyme complexes of the oxidative phosphorylation system. [4]
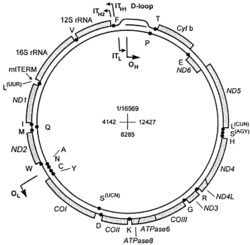
The sequence shows extreme economy in that the genes have none or only a few noncoding bases between them, and in many cases the termination codons are not coded in the DNA but are created post-transcriptionally by polyadenylation of the mRNAs.[6]
Insulin secretion by pancreatic β‐cells is critically dependent on ATP synthesis by mitochondrial oxidative phosphorylation (OXPHOS).
1.3 Type II Diabetes Mellitus
Diabetes, type 2: One of the two major types of diabetes, the type in which the beta cells of the pancreas produce insulin but the body is unable to use it effectively because the cells of the body are resistant to the action of insulin. Although this type of diabetes may not carry the same risk of death from ketoacidosis, it otherwise involves many of the same risks of complications as does type 1 diabetes (in which there is a lack of insulin).[7] Type 2 diabetes used to be known as adult-onset diabetes, but today more children are being diagnosed with the disorder, probably due to the rise in childhood obesity. There's no cure for type 2 diabetes, but losing weight, eating well and exercising can help manage the disease. If diet and exercise aren't enough to manage your blood sugar well, you may also need diabetes medications or insulin therapy.[8]
The Human Mitochondrial DNA and Type II Diabetes Mellitus
Approximately 0.5–1.5% of all diabetic patients exhibit pathogenic mtDNA defects such as duplications, point mutations and large-scale deletions. Most of the known mtDNA mutations cause diabetes by affecting insulin secretion from pancreatic beta cells. Gerbitz et al. have well illustrated the mitochondrial metabolisms in the process of glucose-induced insulin secretion in beta cells. Glucose enters the cells through a specific transporter and stimulates binding of glucokinase to the mitochondrial pore protein, porin. This is followed by phosphorylation of glucose, activation of glycolysis and stimulation of mitochondrial oxidative phosphorylation, resulting in an increase of intracellular ATP. This leads to the closure of the ATP-sensitive K+-channel, the opening of the Ca2+-channel and the increased intracellular Ca2+, which eventually triggers insulin secretion. It is thus likely that pancreatic beta cells with abnormal mitochondria would show a poor insulin secretory response to glucose stimulation. [9]
Thirteen essential OXPHOS proteins are synthesised within mitochondria from the maternally inherited mitochondrial DNA (mtDNA). This raises the possibility that a more subtle genetic variation of the mtDNA might contribute to the risk of developing the disorder by interacting with other genetic and environmental factors.[10]
Multiple studies show that mutations on the different sections of the mitochondrial DNA could be reasons for the development of type 2 diabetes. The following are a few of those many studies.
1. A gradual development of pancreatic β-cell dysfunction upon aging, rather than insulin resistance, is the main mechanism in developing glucose intolerance. Carriers of the A3243G mutation show during a hyperglycemic clamp at 10 mmol/l glucose a marked reduction in first- and second-phase insulin secretion compared with noncarriers. The molecular mechanism by which the A3243G mutation affects insulin secretion may involve an attenuation of cytosolic ADP/ATP levels leading to a resetting of the glucose sensor in the pancreatic β-cell, such as in maturity-onset diabetes of the young (MODY)-2 patients with mutations in glucokinase. Hepatic glucose production may be another factor that becomes deregulated by the A3243G mutation. A mitochondrial dysfunction in muscle is expected to lead to a higher lactate flux to the liver, fueling gluconeogenesis. At this time, no data are available on hepatic glucose production and its suppression by insulin in carriers of the A3243G mutation.[11]
2. Variants in mitochondrial DNA (mtDNA) could be associated with type 2 diabetes because ATP plays a critical role in the production and release of insulin. Diabetes can be precipitated both by mtDNA mutations and by exposure to mitochondrial poisons. The risk of inheriting diabetes from an affected mother is greater than that from an affected father, but this is not explained by maternally inherited diabetes and/or deafness (MIDD) caused by the 3243G : C mtDNA point mutation, which accounts for less than 0.5% of cases of diabetes. A common mtDNA variant (the 16189 variant) is positively correlated with blood fasting insulin, but there are no definitive studies demonstrating that it is associated with diabetes. [12]
3. Mitochondrial DNA polymorphisms that are specifically associated to a particular group have been identified, and recent research is geared towards analyzing metadata and identifying polymorphs that contribute to genetic predisposition of type II diabetes. The T3394C and A12026G polymorphs are factors for mitochondrial diabetes in the Chinese Han population.[13]
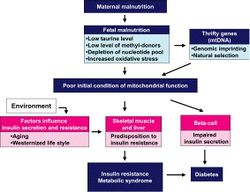
4. Malnutrition during the fetal stage and early development has been known to cause diabetes II, but the link between the two phenomena has not been established. New research is stating genomic imprinting and epigenetic changes in the mtDNA as possible links. If a fetus or an infant goes through a harsh period of malnutrition, new changes will occur in the DNA of the mitochondria. These new changes are beneficial in low food conditions, but they dispose a person to type II diabetes when food is in abundance. [15]
Type II Diabetes and Pathogens
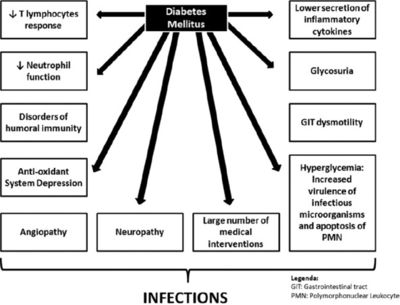
Diabetes is a case where blood sugar levels are high. Most of the diseases associated with diabetes are non-contagious and multifactorial. It is common to mention heart problems, kidney diseases, stroke and hypertension along with diabetes. However, high blood sugar level also interferes with the body's immune system and entails lower responsiveness to infections. When glucose breaks down, dicarbonyls are formed. Dicarbonyls like methylglyoxal (MGO) and glyoxal (GO) alter the structure of human beta-defensin-2 (hBD-2) peptides, hobbling their ability to fight inflammation and infection. Thus, diabetic patients have decreased immune responses to bacteria.[17]The hyperglycemic environment also favors immune dysfunction (e.g., damage to the neutrophil function, depression of the antioxidant system, and humoral immunity), micro- and macro-angiopathies, neuropathy, decrease in the antibacterial activity of urine, gastrointestinal and urinary dysmotility, and greater number of medical interventions in these patients.[18]
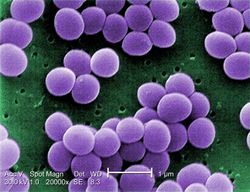
The Staphylococcus species are the major infection causing bacteria seen in Diabetic patients. The species cause a wide variety of diseases starting from foot infection to pneumonia. Sometimes it is hard to attribute a certain disease to a single bacteria; usually, there is a combination of Staphylococcus species and other types of bacteria. The species generally have a symbiotic relationship to the human body; they are commensals who occupy the skin and nostrils with other bacteria. However, they act opportunistic and get pathogenic when our immune system is compromised. Moreover, the species have adaptively evolved so that the immune system can't respond effectively in case of reinfection. Staphylococcus aureus can O-acetylate its peptidoglycan so that the cell wall doesn't breakdown. That in turn limits the induction of pro-inflammatory signals required for optimal Th17 polarization. Th17-T helper 17 cells- are types of T helper cells known for their production of interleukin 17 (IL-17). [19] IL-17 is a pro-inflammatory cytokine that induces the chemokines that signal phagocytes to come and engulf the pathogens.[20]This way the species makes sure that the immune system can't establish memory of infection. [21] In addition to not being killed by the immune system, the species is known to produce toxins that have the potential to kill the innate immune cells.[22]
Bacterial pneumonia- a disease caused by the bacteria Streptococcus pneumoniae- is a very common infection that affects diabetic patients.The bacteria is normally found in air and the upper respiratory tract, but healthy bodies with strong immune systems can effectively prevent infection. However, people who have had other infections and surgery have trouble managing the pathogen. Diabetic patients join the latter group. On average, diabetic patients are hospitalized 0.8 days more than non diabetic patients for community-acquired pneumonia. The mortality rate from pneumonia is also significantly higher for diabetic patients.[23] The effects are even more severe when the pneumonia is acquired from a healthcare center. HCAP(Health Care Acquired Pneumonia) is usually caused by a <Staphylococcus species called Staphylococcus pneumoniae . Since the bacteria live around the hospital, they have evolved to resist the actions of antibiotics.
Conclusion
Overall text length should be at least 1,000 words (before counting references), with at least 2 images. Include at least 5 references under Reference section.
Bold
Italic
Subscript: H2O
Superscript: Fe3+
References
- ↑ [Lynn Sagan, On the origin of mitosing cells, Journal of Theoretical Biology, Volume 14, Issue 3,1967,Pages 225-IN6,ISSN 0022-5193,https://doi.org/10.1016/0022-5193(67)90079-3.(http://www.sciencedirect.com/science/article/pii/0022519367900793)
- ↑ Smith DR. The past, present and future of mitochondrial genomics: have we sequenced enough mtDNAs?. Brief Funct Genomics. 2016;15(1):47–54. doi:10.1093/bfgp/elv027
- ↑ Smith DR. The past, present and future of mitochondrial genomics: have we sequenced enough mtDNAs?. Brief Funct Genomics. 2016;15(1):47–54. doi:10.1093/bfgp/elv027
- ↑ Jan-Willem Taanman, The mitochondrial genome: structure, transcription, translation and replication, Biochimica et Biophysica Acta (BBA) - Bioenergetics, Volume 1410, Issue 2,1999, Pages 103-123, ISSN 0005-2728, https://doi.org/10.1016/S0005-2728(98)00161-3.
- ↑ Fex, M., Nicholas, L., Vishnu, N., Medina, A., Sharoyko, V., Nicholls, D., Spégel, P., & Mulder, H. (2018). The pathogenetic role of β-cell mitochondria in type 2 diabetes, Journal of Endocrinology, 236(3), R145-R159. Retrieved Dec 4, 2019, from https://joe.bioscientifica.com/view/journals/joe/236/3/JOE-17-0367.xml
- ↑ Anderson, S., Bankier, A., Barrell, B. et al. Sequence and organization of the human mitochondrial genome. Nature 290, 457–465 (1981) doi:10.1038/290457a0
- ↑ Jr. WCS. Definition of Diabetes, type 2. MedicineNet. https://www.medicinenet.com/script/main/art.asp?articlekey=2976. Published January 25, 2017.
- ↑ Type 2 diabetes. Mayo Clinic. https://www.mayoclinic.org/diseases-conditions/type-2-diabetes/symptoms-causes/syc-20351193. Published January 9, 2019.
- ↑ Yun Yong Lee, Kyong Soo Park, Youngmi Kim Pak, Hong Kyu Lee, The role of mitochondrial DNA in the development of type 2 diabetes caused by fetal malnutrition, The Journal of Nutritional Biochemistry, Volume 16, Issue 4, 2005, Pages 195-204, ISSN 0955-2863, https://doi.org/10.1016/j.jnutbio.2004.11.002. (http://www.sciencedirect.com/science/article/pii/S0955286304002165)
- ↑ [Chinnery PF, Mowbray C, Patel SK, et al. Mitochondrial DNA haplogroups and type 2 diabetes: a study of 897 cases and 1010 controls. J Med Genet. 2007;44(6):e80. doi:10.1136/jmg.2007.048876]
- ↑ Maassen, J. Antonie, et al. “Mitochondrial Diabetes.” Diabetes, American Diabetes Association, 1 Feb. 2004, https://diabetes.diabetesjournals.org/content/53/suppl_1/S103.short.
- ↑ Poulton, Joanna, Luan, et al. Type 2 diabetes is associated with a common mitochondrial variant: evidence from a population-based case–control study. OUP Academic. https://academic.oup.com/hmg/article/11/13/1581/744945. Published June 15, 2002.
- ↑ Liu S-M, Zhou X, Zheng F, et al. Novel mutations found in mitochondrial diabetes in Chinese Han population. Diabetes Research and Clinical Practice. https://www.sciencedirect.com/science/article/pii/S0168822706004347?via=ihub. Published November 27, 2006.
- ↑ Lee YY, Park KS, Pak YK, Lee HK. The role of mitochondrial DNA in the development of type 2 diabetes caused by fetal malnutrition. The Journal of Nutritional Biochemistry. https://www.sciencedirect.com/science/article/pii/S0955286304002165. Published April 1, 2005.
- ↑ Lee YY, Park KS, Pak YK, Lee HK. The role of mitochondrial DNA in the development of type 2 diabetes caused by fetal malnutrition. The Journal of Nutritional Biochemistry. https://www.sciencedirect.com/science/article/pii/S0955286304002165. Published April 1, 2005.
- ↑ Casqueiro J, Casqueiro J, Alves C. Infections in patients with diabetes mellitus: A review of pathogenesis. Indian J Endocrinol Metab. 2012;16 Suppl 1(Suppl1):S27–S36. doi:10.4103/2230-8210.94253
- ↑ High blood sugar of diabetes can cause immune system malfunction, triggering infection. ScienceDaily. https://www.sciencedaily.com/releases/2015/08/150806151354.htm. Published August 6, 2015.
- ↑ Casqueiro J, Casqueiro J, Alves C. Infections in patients with diabetes mellitus: A review of pathogenesis. Indian J Endocrinol Metab. 2012;16 Suppl 1(Suppl1):S27–S36. doi:10.4103/2230-8210.94253
- ↑ Wikipedia contributors. T helper 17 cell. Wikipedia, The Free Encyclopedia. November 13, 2019, 16:00 UTC. Available at: https://en.wikipedia.org/w/index.php?title=T_helper_17_cell&oldid=925989292. Accessed December 6, 2019.
- ↑ Wikipedia contributors. Interleukin 17. Wikipedia, The Free Encyclopedia. November 18, 2019, 13:21 UTC. Available at: https://en.wikipedia.org/w/index.php?title=Interleukin_17&oldid=926748387. Accessed December 6, 2019.
- ↑ Marisel Sanchez, Stacey L. Kolar, Sabrina Müller, Christopher N. Reyes, Andrea J. Wolf, Chihiro Ogawa, Rajat Singhania, Daniel D. De Carvalho, Moshe Arditi, David M. Underhill, Gislâine A. Martins, George Y. Liu, O-Acetylation of Peptidoglycan Limits Helper T Cell Priming and Permits Staphylococcus aureus Reinfection, Cell Host & Microbe, Volume 22, Issue 4, 2017, Pages 543-551.e4, ISSN 1931-3128, https://doi.org/10.1016/j.chom.2017.08.008. (http://www.sciencedirect.com/science/article/pii/S1931312817303414)
- ↑ Thammavongsa V, Kim HK, Missiakas D, Schneewind O. Staphylococcal manipulation of host immune responses. Nat Rev Microbiol. 2015;13(9):529–543. doi:10.1038/nrmicro3521
- ↑ Martins, Boavida JM, Raposo, Nunes, Ribeiro, Macedo. Diabetes hinders community-acquired pneumonia outcomes in hospitalized patients. BMJ Open Diabetes Research & Care. https://drc.bmj.com/content/4/1/e000181. Published May 1, 2016.
Edited by [Beimnet Beyene Kassaye], student of Joan Slonczewski for BIOL 116 Information in Living Systems, 2019, Kenyon College.