Murine Leukemia Virus (MuLV)
by Rodney S. Tucker II
Introduction
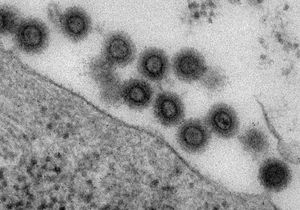
The murine leukemia virus (MuLV)(Figure 1) is one of the simplest retroviruses. The virus uses the mouse as a host and is able to cause cancer in the host. The retrovirus MuLV has a positive, single-stranded sense RNA that replicates via reverse transcription. The virus has been of interest since its discovery the beginning of 1950’s. The virus was considered significant since it was shown to induce leukemia in inoculate mice [1]. While it hasn’t been shown to be effective at infecting humans, it has still proven to be relevant in the study of viral infection in humans through the observation of its behavior in mice models. As a retrovirus, it has also been targeted for possible use as a viral vector.
MuLV Structure and Genome
The murine leukemia virus has a spherical shape that is usually has a diameter of 80 to 100 nm. The envelope of the virus is covered with glycoprotein spikes similar to that of the Human Immunodeficiency Virus (HIV). MuLV belongs to a subgroup of the retrovirus called the gammaretrovirus [1]. Gammaretroviruses have only four ORF (open reading frame) and a genome of more than 8 kb (kilo-base pairs) [3].
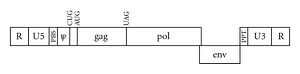
The MuLV genome (Figure 2) can be catagorized into three sections with each section coding for a certain gene. Three major genes produced by the murine leukemia virus are gag, pol, and env. The gag gene codes for the group-specific antigen which is responsible for the production of the viral matrix capsid and nucleoproteins, which are categorized into three domains. The MA domain of Gag protein targets the plasma membrane of the host mouse blood cell. The Gag protein’s plasma membrane association is aided by myristic acid. The CA domain of gag is controls the interaction of gag molecules in immature MuLV virions before they exits the host cell. The NC monitors the interaction of gag molecules and RNA. The pol gene encodes RT (reverse transcriptase), PR (protease), and IN (integrase). The reverse transcriptase, which is unique to retroviruses, is used to make complementary DNA by reverse transcribing its own RNA into DNA. Unlike reverse transcriptase found in other retroviruses, the reverse transcriptase in MuLV is able to act as a monomer as opposed to a dimer. The integrase takes over from this stage and intergrates the synthesized viral DNA into the host cell’s DNA. The integrase removes two nucleotides from the 3’ end of both strands of the viral DNA and fastens the processed 3’ ends to the host cell’s DNA. The integrated DNA can now be replicated by the mouse cell when it performs its DNA replication and synthesis. Although it isn’t confirmed, it is believed that the protein comes in the form of a tetramer. The protease acts as an enzyme for the cleavage of the mature virus from the mouse cell as it exits. This protease acts in the form of a dimer. The env gene codes for the protein that composes the viral envelope. This aids the virus in selecting and attacking certain cell types in the host, or blood cells in the host mouse via the RBD (receptor binding domain). The env protein works by fusing to the selected cell type. Once this fusion occurs, the viral capsid and RNA is released into the mouse cell to begin the process of replication through reverse-transcription [1]. The specific protein produced in the MuLV virus’s viral envelope is gp (glycoprotein) 71.
Process of Infection
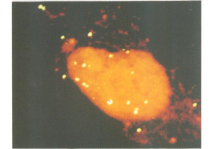
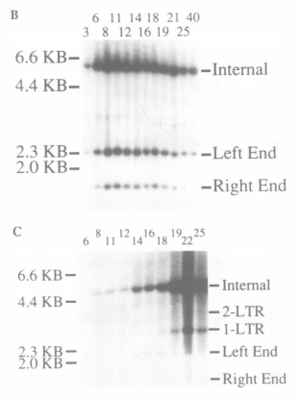
As discussed in the previous section, integrase is the protein responsible for the integration of the viral DNA into the host cell’s chromosomal DNA. However, the process of the RNA entering the nucleus and the mechanism used to integrate the viral DNA was poorly understood. While at first it was thought that the S (DNA replication) phase of the mouse cell was responsible for the integration of MuLV viral DNA, it was proven that the viral DNA is only integrated after the mouse cell had progressed through mitosis and not during the S phase [2]. This was proven using fluorescent in situ hybridization (Figure 3) and Hirt procedure (Figure 4).
Rat-1 cells synchronized at the G1/S border by aphidicolin treatment were infected with the MuLV virus. The washing of the aphidicolin removed the arrestment of the cell allowing it to progress through mitosis. These cells are monitored and counted using a DNA histogram and visual aid to observe the transition from diploid to haploid cells. The Hirt procedure is used to quantify the synthesis and integration of the MuLV viral DNA. This process separates the unintegrated MuLV viral DNA from the genomic DNA. The DNA samples in both the free MuLV viral DNA found in the Hirt supernatant and the genomic DNA found in the Hirt pellet is cut using Xhol and Nsil. In the free MuLV viral DNA, this appeared as the distinct separation of three bands on a Southern blot. The viral DNA integrated into the genomic DNA reacted to the addition of the enzymes unpredictably, which is indicated by the lack of three distinct bands. This variation was analyzed to determine the amount/presence of MuLV viral DNA integrated at various steps of the cell’s mitosis. The further specification of the cell cycle phases essential for the integration of MuLV viral DNA was determined using FT210 mutant mice cells. The FT210 cells arrest clearly at the G2 phase when subjected to a restrictive temperature (39 ºC). Similar to the Rat-1 cells, the FT210 cells are treated with aphidicolin to pause the cellular mitosis at the G1/S phase border. Upon the removal of the aphidicolin, the cells are exposed at the restrictive temperature of 39 ºC to arrest the cells at the G2 phase. The integration was once again quantified using the Hirt procedure. The results of the test performed on the murine cells at 39 ºC revealed there was no integration of MuLV viral DNA at the G2 phase of mitosis. The resulting lack of integration of viral DNA was proven to be a sole result of mitosis phase as opposed to an effect of temperature using wild type FM3A cells.
Since it was proven that the cell must at least progress into the M phase of mitosis, the next logical step was to narrow down the specific stage of M phase, specifically before/after metaphase, required for integration. This was tested by treating the cells with nocodazole to arrest cells at the metaphase (lining up of chromosomes) of M phase mitosis.
The analysis by Hirt prodecure showed that integration doesn’t occur in the nocodazole treated cells. This would mean the viral DNA was integrated had integrated after the cell left metaphase or that integration occurs at the last moment in the last metaphase cells. However, the near impossibility of the second explanation resulted in the conclusion that integration occurs after the cell exits metaphase/mitosis. Therefore, MuLV viral DNA is only integrated after the host mouse cell has finished metaphase. It is hypothesized that the reason for the lateness in the integration of the viral DNA is due to the fact that the viral DNA is only able to enter the nucleus after the nucleus is dissolved for mitosis. This is supported by the fluorescence in situ hybridization that will be discussed in the following paragraph. Once the DNA is integrated, there is no need for the virus to wait for a second round of mitosis [2].
One conclusion that was considered was the fact that integration required the viral DNA to enter the host cell’s nucleus. The stage at which the virus invaded the host nucleus was determined using fluorescence in situ hybridization. The MuLV DNA present inside the individual nuclei was monitored by staining the cells with propidium iodide. After the cells are stained, analysis is carried out using confocal laser scanning microscopy to visualize sections of the nuclei. MuLV viral DNA levels in nuclei of cells arrested by aphidicolin were minute. However, shortly after the cell was cleansed of the aphidicolin, there was a sharp increase of MuLV viral DNA levels in the nuclei. This suggests that the MuLV DNA permeates through the nuclear membrane when the cell progresses through the G1 phase into the S phase [2]. This knowledge of how this retrovirus replicates can be applied and compared to other retroviruses to expose weak points to be selected for prevention of viral infection.
MuLV as a Model
In addition to analyzing the virus, MuLV has also been used in mice to model a viral infection in humans. One model the MuLV virus has formed is the study of severe respiratory disease as caused by the pneumonia virus. However, the use of the virus proves to be difficult as will be shown in the following paragraphs.
The mouse line RAW264.7 is used since it shows similarities to the macrophages from derived mouse lines. This cell line was taken from a tumor found in a BAB/14 mouse exposed to the Abelson murine leukemia virus. The mouse line is also susceptive to the propagation of the pneumonia virus which is the causative agent of the disease being studied. This in addition to its highly efficient DNA transfection (introduction of nucleic acids into cells), sensitivity to RNA interference, and possession of receptors for the virus makes it a key cell strain for the study. In fact, the virus was recognized by the Alliance of Cellular Signaling as major experimental model for large scale signaling pathways [4]. Recently, it has been discovered that the cell line still expresses the murine leukemia virus (Figure 5), which complicates the 1500 plus studies that have been conducted using the strain as a model. Determination of murine leukemia virus expression was conducted by using proteinase K to retrieve antigens stained by avidin-biotin immunohistochemistry (IHC), then fixing RAW264.7 cells with formalin and paraffin. This revealed the expression of MuLV p30, a viral core protein. The glycoprotein gp70 was found to be expressed on the cell surface with the aid of an immunofluorescence assay. The reason why this critical expression was not discovered earlier is unknown but lab conditions, cell health, and the culture condition have been attributed to the oversight of MuLV expression.
This also brings up the concern of whether the RAW264.7 has pathogenic abilities [4]. The testing for the cell strains pathogenic potential consisted of inoculating BALB/c mice with either a supernatant of RAW264.7 or Mo-MuLV for comparison of standard MuLV infection. The mice that weren’t inoculated for either solution served as a control group. The mice were then compared for various signs of infection recognized for MuLV. The RAW264.7 inoculated mice showed a spleen enlargement of up to 1800 mg compared to Mo-MuLV inoculated mice which showed spleen enlargement slightly over 600 mg. In addition, the RAW264.7 inoculated mice also showed MuLV p30 expression in the spleen and tissues showing signs of tumor development. The RAW264.7 inoculated mice also showed viral induction of B cell lymphomas which were not shown in the Mo-MuLV inoculated mice. A troubling discovery was the finding of a mast cell tumor that is rarely seen in the mouse neoplasm. This tumor had no confirmed correlation to a viral inoculation.
Since it was found that RAW264.7 mouse cell strains do have pathogenic capabilities, it has now proven problematic for previous studies conducted using the cell as a model. The ability to produce tumors complicates the testing of pathogenicity of other unrelated pathogenic agents. The cell strain can also cause B cell proliferation which would complicate the study of agents that can cause immunodeficiency. This one example has de-based over 1500 articles that have used this model tissue as the base of their study. This is an example of how unpredictable the MuLV virus can be in studying the effect it has on models [4]. It also raises the question of whether other cell strains similar to RAW264.7 are susceptible to pathogenic infection of the model organism.
Possible Use as a Viral Vector
Gene therapy has been considered as a method for curing various genetic and viral diseases. This could serve as an alternative to intrusive methods such as surgery or drugs. The concept behind gene therapy is either replacing a mutated gene with a normal or healthy gene, inactivating the mutant gene, or inserting a new gene to fight the disease [5]. It has been determined the best systematic treatment via gene therapy would be using a retrovirus, such as HIV or MuLV, capable of gene delivery to specific cells in vivo and efficiently expressing the desired gene in the cells. One such use is the binding of heregulin Env proteins to the Erb3/4 receptor. This allows the modified MuLV to infect breast cancer cells lines in humans [6]. The example this section will delve closely into is the possible use of modified MuLV viruses to treat AIDS.
Concerning the gene therapy approach towards HIV, it has been established that is safer for to attach HIV components on another retrovirus, such as the murine leukemia virus. This process of incorporating glycoprotein on a foreign virus’s envelope is known as pseudotyping. One problem previously faced was the inability to incorporate HIV glycoprotein onto the MuLV envelope due to the 150 amino acid long tail on gp 41. This issue was circumvented by truncating the cytoplasmic Env glycoprotein. The effectiveness of the truncated MuLV was tested by comparing the infection of T cells with HIV negative proviral DNA and MuLV GagPol encoded plasmids (truncated MuLV). It was shown that the MuLV virus was as, if not more, effective at infecting cells as its HIV origin. The further testing of the truncated MuLV virus’s specificity to CD4 cells was performed to determine if the virus’s host range was narrowed. This was tested by inoculating COS cells, HeLa, and HeLaCD4 with either HIV Env-negative proviral DNA, the HIV p17-matrix mutant Env-negative proviral DNA, or the truncated MuLV. The efficiency of genetic transference was tested by insering the lacZ gene plasmid into the viral vectors. If the virus proved to be effective, the CD4 cells would have the lacZ gene present in its DNA. In addition, the lacZ gene should be absent in the other two cell types that lack a CD4 receptor. The presence of the lacZ in each category of cells would be determined using a Southern blot. The resulting experiment revealed that the COS and HeLa cells didn’t express a significant amount of lacZ gene in the southern blot. More importantly, lacZ was successfully translocated into the HeLaCD4 cells. This allowed the MuLV virus to continue its biological activity in infecting HeLa CD4 cells. The virus was also able to successfully activate CD4+ peripheral blood lymphocytes (PBL) which indicates it can be used for gene therapy of cells expressing CD4. To further dissect the pseudotyped MuLV virus’s ability to activate PBL, CD4 and CD8 cell sorting was performed. The sorted cells were then lysed and analyzed using a PCR Southern blot to determine lacZ transduction. Although it was found that both CD4 and CD8 cells received the lacZ gene, only CD4 cells were susceptible to the truncated MuLV. This further proved that the truncated MuLV was capable of specifically targeting and infecting CD4 PBL populations [6].
One possibility in using the truncated MuLV viral vector is the genetic therapy of AIDS (auto immunodeficiency syndrome). The ironic use of a virus to prevent a viral infection is possible since the truncated MuLV selectively targets CD4 cells, the same target cells of HIV, it would be relatively easy to manufacture a plasmid to be inserted into the T cells which would either block the HIV genetic replication or prevent the binding of HIV to T cells. The issue of creating vectors capable of outcompeting HIV particles is still being faced to make sure the vector will completely block HIV infection. The viral vector can also be further tuned to make it more effective in transduction the desired plasmid into the selected cell type. The viral vector can also be used for further study of gene expression of CD4 cell populations [6]. Aside from its use in the treatment of AIDS, the MuLV virus can be further researched to be specific to other cell types to expand its use in the gene therapy of other disease. This will also serve as an example for modifying other viruses to become efficient at transducing genes for cell types not possible for the murine leukemia virus.
Conclusion
Since the murine leukemia virus’s discovery in the 1950’s, it has played a major role in the study of virology. Sometimes this role can be negative as seen in the effect MuLV expression had in the study of RAW264.7 mouse cell lines. Its role can also be positive as seen in its possible use as a viral vector in HIV and breast cancer treatment. The significance of the MuLV virus being a simple retrovirus with the capability of selective infection could lead to further use of it as a viral vector for other diseases. Even with the negative discovery of MuLV expression in a cell line that ended up interfering with the study of other infectious viruses, the discovery led to the corrected observation of viral expression in genetically engineered tissue. It also raises the suspicion of other engineered tissues that are consistently analyzed and studied as models for viral infection. Even the failure of the virus in model organism tissue lines doesn’t prevent its use as a model for retroviruses. The virus provides a perspective of how retroviruses act when infecting cells such as the mechanism of reverse transcriptase. In response to this discovered information, methods to block the viral infection in response to the conditions needed for the reproduction of the virus and different sections or stages the virus proceeds through in the process of reproduction can be found. More importantly, this information can be used and compared to other retroviruses, like HIV, to cure other viral infections.
Reference
1. Alan Rein. Murine Leukemia Virus: Objects and Organisms. Volume 2011 (2011), Article ID 403419, 14 pages doi:10.1155/2011/403419.
2. TaiYun Roe, Thomas C. Reynolds, George Yu, and Patrick O. Brown. Integration of murine leukemia virus DNA depends on mitosis. vol.12 no.5 pp.2099-2108, 1993.
3. DPV 2006. Available at http://www.dpvweb.net/notes/showgenus.php?genus=Gammaretrovirus
4. Janet W Hartley, Leonard H Evans, Kim Y Green, Zohreh Naghashfar, Alfonso R Macias, Patricia M Zerfas and Jerrold M Ward. Expression of infectious murine leukemia viruses by RAW264.7 cells, a potential complication for studies with a widely used mouse macrophage cell line. Available at http://www.retrovirology.com/content/5/1/1
5. Genetics Home Reference 2013. Available at http://ghr.nlm.nih.gov/handbook/therapy/genetherapy.
6. R Lodge, RA Subbramanian, J Forget, G Lemay and E.A Cohen. MuLV-based vectors pseudotyped with truncated HIV glycoproteins mediate specific gene transfer in CD4+ peripheral blood lymphocytes. Gene Therapy (1998) 5, 655–664.
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2013, Kenyon College.