Necrotizing fasciitis induced by Vibrio vulnificus
Necrotizing fasciitis (NF) is a soft-tissue infection that is most commonly caused by bacteria that infect open wounds and results in tissue damage and death. For this reason the bacteria that induce this infection are termed “flesh-eating bacteria”. Different variations of Necrotizing fasciitis exist and they are separated into three general groups based on the types of bacteria that cause the infection 1. The first and most common are the Streptococcus and Staphylococcus bacteria, which are of growing concern because of the emergence of MRSA’s. The second group involves polymicrobial-induced infection or infection by Vibrio vulnificus. Clostridium bacteria cause the third variation of Necrotizing fasciitis.
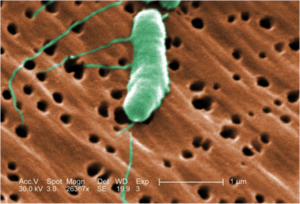
While Vibrio vulnificus is one of the more infrequent causes of NF, it has a relatively high mortality rate of 26% 2 because it spreads quickly and it is hard to diagnose. V. vulnificus thrives in warm, medium salinity water and causes infections either through the ingestion of contaminated seafood or in this case, through open wounds that come in contact with V. vulnificus contaminated water 3. There also appears to be increasing incidence of Vibrios infections, which could be the result of ocean warming due to global climate change 4.
Pathogenesis
Toxins Involved
The people most at risk for infection are those with immunodeficiencies, especially liver disease, hepatitis B or C or any disorder characterized by high iron levels 5. Iron is believed to promote infection by enhancing growth of the bacteria and reducing immune response 6. V. vulnificus uses cytotoxins i.e. compound that are toxic to cells, to cause the infection. A few mechanisms of V. vulnificus cytotoxicity resulting in tissue damage have been studied in mice, including the toxins VvTtxA, hemolysin (Vvha), and VvpE.
- VvpE is an extracellular metalloprotease and its purified form has been shown to cause tissue necrosis and bullous lesions, which are characteristic of NF caused by V. vulnificus. VvpE degrades type IV collagen, which is a structural component of basement membranes, and it also activates procaspase 3, which is a protein involved in cellular apoptosis 8.
Quorum Sensing
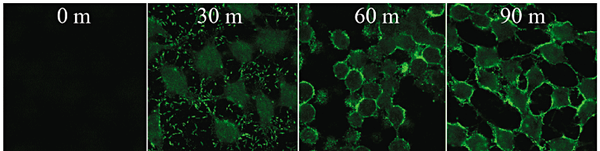
Cell-to-cell contact with the host is required for cytotoxicity (Figure 2) and V. vulnificus knows when to release its toxins using quorum sensing 9. LuxS synthesizes the autoinducer and LuxR is the transcriptional regulator. Mutation of either of the genes that codes these proteins resulted in decreased cytotoxicity. The gene luxS influences the transcription of vvhA and vvpE, resulting in increased or decreased levels of these toxins. The protein HlyU also regulates cytotoxicity 8. The gene hlyU regulates the expression of rtxA at the transcriptional level by binding upstream of the rtxA promoter region, initiating transcription.
Symptoms and Diagnosis
Symptoms tend to appear in the summer months, when the seawater temperature rises resulting in increased V. vulnificus presence 10. Early symptoms come across as the flu: fever, diarrhea, and nausea. Within 12-24 hours after infection, patients develop severe cellulitis with tissue swelling and hemorrhagic bullae and many become septic, which can increase the mortality rate from about 33% to a staggering 53-75% 5, 13. Some characteristic symptoms of this infection include vasodilation and edema (swelling). This can be explained by the presence of the toxin Hemolysin, which can form pores in the endothelial cells of blood vessels 8, 12. It induces histamine to be released from mast cells and it activates kinin pathways. Metalloproteases break down type IV collagen causing hemorrhagic skin damage. The later and more extreme symptom of necrotic cell death is mostly associated with the toxin RtxA, which causes depolymerization of actin in cells 12.
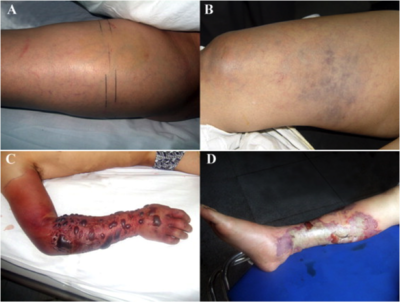
Many of the patients also experience a change in mental status, such as lethargy and disorientation 5, 11. Unfortunately because the infection acts so quickly, and can be misdiagnosed as the flu or a less dangerous skin infection, it is difficult to treat effectively. Knowing whether the patient has come into contact with the ocean recently is the most significant indicator of whether it is Vibrio induced NF, followed by whether the patient has any form of immunodifficiency.
Treatment Options
If treatment is delayed even by a few hours (4-8 hrs), the risk of death significantly increases 10. Unfortunately diagnosis of this infection is difficult, with defining symptoms, such as tissue necrosis, not arising until late into the infection. The bacteria propagate rapidly and by the time the infection is recognized, treatment is no longer as effective. A delay of 24 hours can increase the mortality rate from 33% to 53%, if the patient is septic. And if this patient is not treated within 72 hours, mortality rate can reach 100% 13. Treatment can vary from relatively noninvasive antibiotics to amputation of infected tissue and limbs.
Necrotizing tissue receives an insufficient amount of blood because of thrombosis of the blood vessels supplying that area. This means it is difficult to treat the site of infection with a therapeutic level of antibiotics. Thus it is essential to remove it. Early surgery results in decreased hospital and ICU stay time and a decreased mortality rate 10. V. vulnificus is vulnerable to a few classes of antibiotics, including third-generation cephalosporin’s, fluoroquinolones and tetracylcine. Multiple studies have shown that combined antibiotic therapy is much more effective against V. vulnificus NF infection than any single antibiotic treatment. One particularly effective combination is third generation cephalosporins with minocycline 14.
Further Reading
It appears that Vibrio caused infections are increasing in incidence and some researchers believe that this increase is related to global warming 4, 15. Vibrio diseases are increasing worldwide and increased surface seawater temperature has been linked to outbreaks of illness in Israel, Europe, Alaska and South America. Temperature effects the growth rates and pathogen city of Vibrio species. Increasing the water temperature in lab studies has resulted in increased growth rate and increased expression of virulence traits in the bacteria 4. More research still needs to be done to find a definitive link between these two factors, but understanding this connection can help us forecast future outbreaks and prepare for them.
References
[1] Davis C. 2012. Necrotizing Fasciitis (Flesh-Eating Disease) Symptoms, Treatment, Causes, Pictures, Diagnosis. MedicineNet [Internet]. Available from: http://www.medicinenet.com/necrotizing_fasciitis/article.htm
[2] Oliver J.D. 2005. Wound infections caused by Vibrio vulnificus and other marine bacteria. Epidemiol. Infect. 133: 383–391. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2870261/
[3] Vibrio vulnificus | Vibrio Illness (Vibriosis) | CDC. CDC [Internet]. 2013. Available from: http://www.cdc.gov/vibrio/vibriov.html
[4] Vezzulli, L. et al. 2013. Ocean warming and spread of pathogenic vibrios in the aquatic environment. Microb Ecol. 65: 817-825. http://link.springer.com/article/10.1007%2Fs00248-012-0163-2
[5] Horseman M, Surani S. 2011. A comprehensive review of Vibrio vulnificus: an important cause of severe sepsis and skin and soft-tissue infection. International Journal of Infectious Diseases 15:e157–e166. http://www.sciencedirect.com/science/article/pii/S1201971210025385
[6]Hor L., et al. 2000. Mechanism of high susceptibility of iron-overloaded mouse to Vibrio vulnificus infection. Microbiol Immunol 44:871–8. https://www.jstage.jst.go.jp/article/mandi1977/44/11/44_11_871/_article
[7]Lee J.H., et al. 2007. Identification and characterization of the Vibrio vulnificus rtxA essential for cytotoxicity in vitro and virulence in mice. J Microbiol 45:146–52. http://www.msk.or.kr/jsp/view_old_journalD.jsp?paperSeq=2520
[8] Jones MK, Oliver JD. 2009. Vibrio vulnificus: disease and pathogenesis. Infect Immun 77:1723–33. http://iai.asm.org/content/77/5/1723.full
[9] Kim, S. Y., et al. 2003. Regulation of Vibrio vulnificus virulence by the LuxS quorum-sensing system. Mol. Microbiol. 48:1647-1664. http://onlinelibrary.wiley.com/doi/10.1046/j.1365-2958.2003.03536.x/full
[10]Matsuoka Y, et al. 2013. Accurate diagnosis and treatment of Vibrio vulnificus infection: a retrospective study of 12 cases. Braz J Infect Dis 17:7–12. http://www.sciencedirect.com/science/article/pii/S141386701200219X
[11] Hong G-L, et al. 2012. Surgical treatment of 19 cases with Vibrio necrotising fasciitis. Burns 38:290–295. http://www.sciencedirect.com/science/article/pii/S0305417911001380
[12] Gulig P., Bourdage K., and Starks A. 2005. Molecular Pathogenesis of Vibrio vulnificus. Journal of Microbiology 5: 118-131. http://www.msk.or.kr/jsp/view_old_journalD.jsp?paperSeq=2131
[13] Bross M., et al. 2007. Vibrio vulnificus Infection: Diagnosis and Treatment. Am Fam Physician 76(4):539-544. http://www.aafp.org/afp/2007/0815/p539.html#afp20070815p539-b12
[14] Chen S-C, et al. 2012. Antibiotic therapy for necrotizing fasciitis caused by Vibrio vulnificus: retrospective analysis of an 8 year period. J Antimicrob Chemother 67: 488–493. http://jac.oxfordjournals.org/content/67/2/488.full.pdf+html
[15] Paz S. et al. 2007. Climate change and the emergence of Vibrio vulnificus disease in Israel. Environmental Research 103(3):390-396. http://www.sciencedirect.com/science/article/pii/S0013935106001526
Edited by (Elana Goldstein), a student of Nora Sullivan in BIOL168L (Microbiology) in The Keck Science Department of the Claremont Colleges Spring 2014.
