Nitrococcus mobilis
Classification
Domain: Bacteria
Phylum: Proteobacteria
Class: Gammaproteobacteria
Order: Chromatiales
Family: Ectothiorhodospiraceae
Genus: Nitrococcus
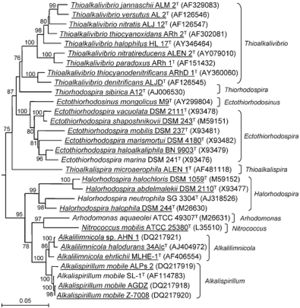
Originally all nitrifying bacteria were classified under the family Nitrobacteraceae. It wasn't until the ability to sequence the 16s rRNA gene that this family was broken up as the species were very diverse. N. mobilis is the only isolated nitrifier in the gammaproteobacteria class. (7)
Family Ectothiorhodospiraceae
The family Ectothiorhodospiraceae is composed mainly of halophilic and haloalkaliphilic bacteria. This family was originally created for purple sulfur bacteria with other bacteria being added later as the 16s rRNA genes were analyzed. Besides the anaerobic purple sulfur bacteria there are also aerobic chemolithotrophs such as Arhodomonas and Alkalispirillum. (10)
Species
|
NCBI: Taxonomy |
Nitrococcus mobilis
Description and Significance
Nitrococcus mobilis is found only in marine environments and is an obligate halophile. It is coccus in shape and has a flagellum to aid in movement. (12) This bacterium is a nitrifier meaning it oxidizes nitrite to nitrate (HNO2 + H2O to HNO3). This is important in nitrogen cycling in marine environments. N. mobilis has been found at lower concentrations than other nitrifying bacteria such as Nitrosomonas species. Since plate count methods were used for determining the concentrations, there is criticism that these numbers are not exact. (1) N. mobilis also plays a small role in the marine carbon cycle as it fixes inorganic carbon which can then be utilized by herterotrophs.
N. mobilis has been cultured in a laboratory which provided a greater insight into its growth requirements. The cultured sample was found in the Pacific Ocean off of the Galapagos Islands. In culture N. mobilis requires media compromised of at least 70% sea water to grow. Just adding salts does not produce a media capable of culturing. The optimal temperature for growth was found experimentally to be 25° to 30°C and a pH of 7.8-8.0. (12)
Biotechnological Uses
Methods for synthesizing polymers using enzymes found in microbes are being developed. Polymers can be synthesized using a thioester reaction with acetyl Co-A attached to different monomers. Therefore microorganisms with metabolic enzymes that use acetyl-co A as a substrate are being considered as possible mechanisms. N. mobilis was found to efficiently catalyze this reaction and is being considered along with various other bacteria.(9)
Fertilizer can also be synthesized in a more environmentally friendly manor by using ground fish, seaweed, and sea water innoculated by different microorganisms including N. mobilis, that participate in the nitrogen cycle. These organisms incorporate atmospheric nitrogen into a form that can be used by crops. (11)
Genome Structure
N. mobilis contains a single circular chromosome that is approximately 25.4 kilobase pairs large. The 16s rRNA gene, used in classification, is 1483 base pairs long. The guanine/cytosine concentration is 60%. (5)
N. mobilis has been found to have participated in horizontal gene transfer of a carboxysomal operon with Paulinella chromatophora which is an amoeba. The amoeba's DNA has evidence of several horizontal gene transfers. The DNA of Paulinella chromatophora shows evidence that it may have been one of the first eukaryotes as it is compromised of several bacterial operons. (2)
Cell Structure, Metabolism and Life Cycle
Nitrococcus mobilis is an obligate chemoautotroph. The energy required for metabolism comes from chemical reactions of oxidizing nitrogen from nitrite to nitrate. They required oxygen for the oxidation reaction and are therefor aerobic. For nitrification to take place, a dissolved oxygen concentration of at least 4.5 mg/ml is required. (4) Inorganic carbon in the form of carbon dioxide is fixed through the Calvin Cycle. (8) The energy from oxidizing nitrogen is very small. 100 moles of nitrite must be oxidized to fix one mole of carbon dioxide. The majority of the energy produced by the cell (80%) is therefore used for carbon fixation. This inhibits rapid growth rates of the cells. (8) If oxygen is not present, the cell can still survive but the growth rate decreases even further. It will use nitrite or nitrate as an electron acceptor instead of oxygen. (4)
The oxidation of nitrite occurs in a single reaction catalyzed by nitrite oxidoreductase. Two electrons from nitrite are transferred to water. This yields nitrate and two protons. Cytochrome oxidase then catalyzes a reaction between the protons released from the oxidation reaction and oxygen to produce water. Therefore, for two molecules of nitrite oxidized, one molecule of oxygen is required. (8) Nitrite oxidoreductase is located on the inner cytoplasmic and intracytoplasmic membranes. (1) The reactions carried out by N. mobilis have been found to be similar to other nitrifying bacteria by comparing nitrite oxidoreducatase used in the different species (1).
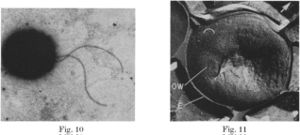
When free floating in the ocean, N. mobilis exists as a single or diploid cell. A biofilm forms when several hundred cells aggregate in a slime layer. (12)
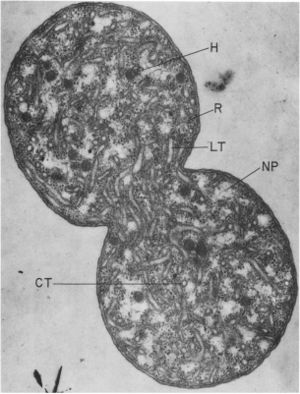
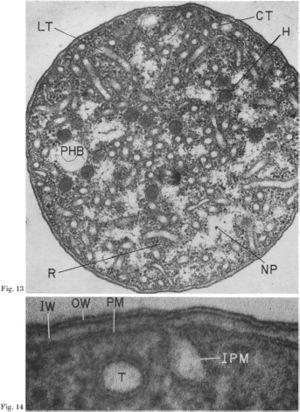
The cell is a cocci with a diameter after division of 1.5 to 1.7 micrometers (it elongates to 3.5 micrometers just before division. (6) N. mobilis is the only nitrifier that is coccus. A 3-4 micrometer long flagella aids in mobility when the cell is free floating. If part of a biofilm, the flagella is lost and the only mode of movement is by rolling, which often lacks a direction. Nitrococcus mobilis is gram negative as it has an outer cell membrane surrounding a multilayered cell wall. In the cytosol of N. mobilis is an unique cytomembrane system. This tubular system is continuous with the plasma membrane. The plasma membrane will invagenate to form tubules, at what appears to be randomly distributed intervals. The tubes themselves are 6-8 nm in diameter and are formed by a double tracked cytomembrane. Electron microscopy has shown that the tubes do have contents in them. Other distinct structures in the cytosol are inclusion bodies. These are aggregates of insoluble proteins. It is unclear if the insolubility is due to misfolding or if the proteins are in different states to be refolded at a later time. The particles are surrounded by a single layered membrane. The most prominent inclusion bodies are hexagonal in shape and have a diameter of approximately 100 nm. A single cell can contain as many as 200 of these particles. The cells also have glycogen storage areas and poly-beta-hydroxybutyrate granules. (12)
The fatty acid composition of the membranes is unique from other nitrifying bacteria. The majority of the membrane is composed of vaccenic acid (18:1 cis11) (42-52%). There is also a higher concentration of hexadcanoic acid (16:0) and cis-9-hexadecenoic acid (16:1 cis9) then other nitrifiers at 27% and 20% respectively. The fatty acid profiles can vary depending on the exact environmental conditions the bacterium is living in. (3)
Ecology and Pathogenesis
Nitrococcus mobilis has no known pathogenicity. There are also no known symbiotic relationships.
N. mobilis plays a role in the marine nitrogen cycle as it converts nitrite to nitrate. It, Nitrospina gracilis, and Nitrospira marina are the only halophiles capable of using nitrite as a substrate for energy. (7) Nitrite rarely collects in the oceans due to these nitrifiers who rapidly oxidize it further. Nitrite comes from the oxidation of ammonium by marine organisms such as Nitrosomonas species. (8)
As multiple nitrifiers are found in the ocean, N. mobilis must either use a resource more efficiently then the others or live at a slightly different depth in the water column. It most likely latter is true because N. mobilis has been found to be more susceptible to light than Nitrobacter species, another nitrifying bacteria found in the ocean. (3) If it is too close to the surface than it will experience more radiation which increases damage to the cell, especially to the DNA. The maximum depth the bacterium can reside is greatly affected by the oxygen profile which decreases with depth. To increase oxygen uptake, N. mobilis may have a specialized mechanism although this has yet to be determine. Its motile nature also allows it to move to areas with increased nutrients. Therefore, the location of N. mobilis is optimized between light intensity, oxygen concentration, and other nutrient profiles.
N. mobilis also plays a small role in the marine carbon cycle as it fixes carbon dioxide released by heterotrophic organisms. This carbon then once again becomes available to the heterotrophs when they consume the bacterium. Once the bacterium dies, it can sink out of the photic zone of the ocean and provide a carbon source to species at greater depths. The role of N. mobilis in the carbon cycle is much smaller than in the nitrogen cycle due to the amount of nitrogen that must be oxidized to provide enough energy to fix carbon. (8)
References
Author
Page authored by Hanna Miller and Toni Miskovich, students of Prof. Jay Lennon at Michigan State University.