Nitrogen Cycling in Tropical Rain Forest Soils
Introduction
Tropical rain forests are canopied biomes situated at and around the equator, specifically between the Tropic of Cancer and the Tropic of Capricorn. A key feature of tropical zones is a fluctuation between dry and wet seasons, although tropical rain forests arguably have no distinct changes in rainfall and therefore have no seasons [1]. A unique population of microbial species resides in tropical rainforests, many of which cannot be found in any other biome. Nitrogen is critical to the growth of all organisms. The genomes of nitrogen cycling microbes encode a diverse array of metabolic enzymes to make soil nitrogen available to all organisms, including themselves. In addition to biological growth, nitrogen metabolism is an extremely encompassing macrocosm widely applicable in waste management, environmental control [1], and preservation of natural resources.
Physical environment
Tropical Rain Forests
Rain forests have a high level of biodiversity above and below ground, maintained within a relatively constant climate. This consistency is brought on by a lack of distinct changes in precipitation levels [1] and consistently high temperatures [2]. The dense canopy of treetops prevents much sunlight from reaching the forest floor. A lack of sunlight directly correlates to an abundance of broad-leafed shrubbery found at ground level[1]. Tropical rain forests flourish where rainfall exceeds evapotranspiration, a combination of evaporation and plant transpiration of water into the atmosphere, for eleven or twelve months a year [1]. No constraints on water supply exist under normal circumstances which permits vegetative plant growth to continue year-round [1]. Tropical rains contain high levels of nitrogen in the NH4+ form [3], significantly contributing to the nitrogen influx. Despite the abundance of nitrogenous compounds, uptake and use is a major rate limiting step in biological growth due to its general recalcitrance, or difficulty in degrading, in soils.
Soil
Tropical rain forests have a unique type of soil classified as an oxisol [2]. Oxisols contain low to moderate nutrient levels with high levels of eroded mineral compounds and humidification, and high biomass content [2]. Despite contrary logic, forest soils have reduced nutrient contents due to efficient microorganism decomposition and acid leaching by rain water. Much of the nitrogen cycling processes occurs in the top layers of soil where decomposing organic matter attracts microorganisms. Many recalcitrant compounds of essential nutrients are present in tropical oxisols. Soils sites with moderate nutrient content can be found in tropical rain forests as well, but are less abundant and scattered [2]. Oxisols contain both oxidized and reduced forms of nitrogen, of which concentrations vary independently of leaching [3].
Biological interactions
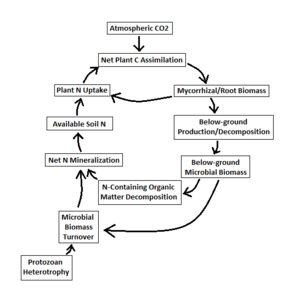
Interactions between microorganisms and plant root systems play a significant role in tropical rain forest soil environments. Such relationships cycle nitrogen from soil microbes to plants that otherwise could only be taken up in low quantities due to poor mobility, such as NH4+ [4]. Extraradical hyphae of arbuscular mycorrhizal fungi in conjunction with root systems support the soil [4] and prevent erosion in addition to nutrient support. In fact, mycorrhizal fungi are better adapted than plant roots for nitrogen mineralization due to their fast growth and penetrative hyphae [4]. Growing microbial communities in close proximity to plant roots increase mineralization of decaying organic material, which raises nutrient uptake rates of root systems [4].
As with any ecological community there is a food chain localized in rain forest soils; bacteria and fungi have predators such as protozoa and microarthropods that contribute to organic material turnover. Fungivorous microarthropods cause a remarkable variation in nitrogen mineralization by consuming soil fungi [5]. Bazzaz (1998)[6] indicates that “… all ecosystems have the same basic components: producers, consumers, decomposers, symbionts, and pathogens. They all cycle carbon, nutrients and water…” in reference to overall tropical forest sensitivity. These crucial relationships between organisms create the need for extreme biodiversity in a unique, niche-abundant system.
Microbial processes
Nitrogen Fixation
Nitrogen (N) fixation is the process of taking gaseous N, such as N2, and enzymatically reducing it into a “fixed” form, ammonia (NH3). Ammonia and ammonium (NH4+) are used interchangeably here since the latter is simply the ionized form of the former. Bacteria are the main nitrogen fixers in tropical rain forest soils, using nitrogenase in combination with other unique enzymes to carry out the necessary series of reactions [6]. Although N2 is the most abundant gas in Earth’s atmosphere, its chemical inertness makes it unusable by non-fixing organisms. Therefore, fixing N is an essential process for all organisms whether they can perform the process or not. Accordingly, bacteria, the main type of nitrogen fixers in rain forest soils, must use fixed N for their own biosynthetic processes. Consequently some soil microbes are strong competitors of plants for soil N, depleting available N to plants during community growth [7]. N fixation rates have been shown to be high in tropical rain forests compared to temperate forests due to blue-green algae (Cyanobacteria) present in soils [3].These bacteria can exist freely or in lichen symbioses with fungi, and are significant nitrogen fixers.
Nitrogen Mineralization
Nitrogen (N) incorporated into organic compounds must be removed during the decomposition of dead animals, plants, and microbes. N mineralization is a process performed by decomposers found ubiquitously in environments with decaying organic matter. By using organic molecules as substrates, enzymes produced by microorganisms are able to extract N ultimately as NH4+. Mineralization is an important process in tropical rain forest soils due to continuously accumulating dead plant and animal matter, as rain forests have a high density of biological entities [1]. Mineralization leads to N immobilization, implying that N is incorporated into microbial biomass. Robertson (1984)[8] finds that significant amounts of N are immobilized into microbial biomass in areas of new rain forest growth; this effect plateaus out as plant succession progresses. Immobilization starts the process of mineralization over again; fresh, dead microbial biomass is re-mineralized, pushing the N cycle forwards. Interestingly a direct correlation between high levels of photosynthesis and increased N mineralization has been observed, corresponding to bacterial populations present around plant roots [9]. An indicator of such changes is the amount of N taken up by bacterial-hunting protozoa populations, which increase along with these bacterial populations [9].
Denitrification
Denitrification is a process performed by specific groups of bacteria, such as species in the genus Pseudomonas, in rain forest soils. It involves the reduction of nitrate (NO3-) or nitrite (NO2-) into nitric oxide (NO-), nitrous oxide (N2O), or dinitrogen (N2) gases. Products of denitrification have adverse effects on the environment, as N2O is a potent greenhouse gas. Surprisingly tropical rain forest soils contain relatively low levels of NO3- [3] despite its water-soluble nature. Therefore denitrification plays less of a significant role in rain forest soils compared to processes producing and consuming NH4+, which is found at higher concentrations [3]. However, denitrification is no less essential to denitrifying bacteria and the tropical ecosystem as a whole: a very delicate balance between influx and efflux of nitrogen is in place.
Key Microorganisms
Bacteria
Gram positive bacteria are very abundant in tropical soils, specifically those in the Actinomyces genus [10].
Gram negative bacteria are fast growing and compete for simple substrates [10], which also limits their productivity in an environment consisting of complex carbon sources. Pseudomonas sp. are known to promote plant growth by inhibiting pathogenic infections, and are denitrifying bacteria[11]. Bacteria of the phylum Cyanobacteria (blue-green algae) form a significant nitrogen fixing population in rain forests [3].
Arbuscular mycorrhizal Fungi
Arbuscular mycorrhizal fungi are the most common fungal found in symbiosis with tropical crop roots [4], perhaps indicating a selective advantage. The Glomeromycota phylum holds all arbuscular mycorrhizal fungi [12].
Other Fungi
Trichoderma sp. and Fusarium sp. are two examples of root pathogens [12].
Doliocarpus sp., Trichilia tuberculata, and Alseis blackiana are three widely distributed tropical fungi [12].
Ascomycota sp. are abundant leaf-litter decomposers [12].
Eukarya
Protozoa are involved in bacterial biomass turnover as these organisms are largely heterotrophic [13]. Amoebae, flagellates, and ciliates constitute most of the microbial eukaryotes found in tropical rain forests [13].
Fungivorous microarthropods are also heterotrophic, contributing to fungal biomass turnover [5]. Oribitadia, Mesostigmata, and Collembola are the main genus’s found in tropical rain forest soils [5].
Current Research
In a series of studies of two forest ecosystems in Costa Rica, Eaton et al. (2012)[14] employed a variety of nucleic acid and conventional biomass measurement techniques to examine the role of soil microorganisms as indicators of environmental change. Categorizing soil microorganisms is a necessary step in continuing research on tropical rain forest biodiversity and cycling.
Leguminous trees are an abundant tropical rain forest inhabitant, and are responsible for nitrogen fixation. Some of these trees have root nodules in which rhizobial organisms reside and fix nitrogen. Pons et al. (2007)[15] used the 15^N natural abundance method to estimate amounts of nitrogen fixed by leguminous and non-leguminous trees in the tropical Guyana. Their overall objective was to discern differences in nitrogen fixation rates between species, perhaps establishing new information about what plants to use in nitrogen-poor soils for agricultural use.
References
1 Wright, J. S. "Phenological responses to seasonality in tropical forest plants". Tropical Forest Plant Ecophysiology, 1996 (440-460).
2 Swift, M. J., Andren, O., Brussaard, L., Briones, M., Couteaux, M. .-M., Ekschmitt, K., Kjoller, A., Loiseau, P. and Smith, P. “Global change, soil biodiversity, and nitrogen cycling in terrestrial ecosystems: three case studies”. Global Change Biology, 1998, 4:7 (729–743). DOI: 10.1046/j.1365-2486.1998.00207.x
3 Jordan, C., Caskey, W., Escalante, G., Herrera, R., Montagnini, F., Todd, R., Uhl, C. “The nitrogen cycle in a ‘Terra Firme’ rainforest on oxisol in the Amazon territory of Venezuela”. Plant and Soil, 1982, 67:1-3 (325-332). DOI: 10.1007/BF02182779
4 Cardoso, I. M., Kuyper, T. W. “Mycorrhizas and tropical soil fertility”. Agriculture, Ecosystems & Environment, 2006, 116:1–2 (72–84). DOI: 10.1016/j.agee.2006.03.011
5 Beare, M. H., Parmelee R. W., Hendrix, P. F., Cheng, w., Coleman, D.C., Crossley, D. A. “Microbial and Faunal Interactions and Effects on Litter Nitrogen and Decomposition in Agroecosystems”. Ecological Monographs, 1992, 62:4 (569-591)
6 Kim, J., Rees, D. C. “Nitrogenase and biological nitrogen fixation”. Biochemistry, 1994, 33:2 (389–397) DOI: 10.1021/bi00168a001
7 Bazzaz, F. A. “Tropical Forests in a Future Climate: Changes in Biological Diversity and Impact on the Global Carbon Cycle”. Climatic Change, 1998, 39:2-3 (317-336). DOI: 10.1023/A:1005359605003
8Robertson, P. G. “Nitrification and nitrogen mineralization in a lowland rainforest succession in Costa Rica, Central America”. Oecologia, 1984, 61:1 (99-104). DOI: 10.1007/BF00379093
9 Zak, D. R., Pregitzer, K. S., Curtis, P. S., Teeri, J. A., Fogel, R., Randlett, D. L. “Elevated atmospheric CO2 and feedback between carbon and nitrogen cycles”. Plant and Soil, 1993, 151:1 (105-117). DOI: 10.1007/BF00010791
10 Waldrop, M. P., Balser, T. C., Firestone, M. K. “Linking microbial community composition to function in a tropical soil” Soil Biology and Biochemistry, 2000. DOI: 10.1016/S0038-0717(00)00157-7
11 Drigo, B., Kowalchuk, G. A., van Veen, J. A. “Climate change goes underground: effects of elevated atmospheric CO2 on microbial community structure and activities in the rhizosphere”. Biology and Fertility of Soils, 2008, 44:5 (667-679). DOI: 10.1007/s00374-008-0277-3
12 Farrow, W. M. “Tropical Soil Fungi”. Mycologia, 1954, 46:5 (632-646)
13 Foissner, W. “Tropical Protozoan Diversity: 80 Ciliate Species (Protozoa, Ciliophora) in a Soil Sample from a Tropical Dry Forest of Costa Rica, with Descriptions of four New Genera and seven New Species”. Archiv für Protistenkunde, 1995, 145:1-2 (37–79). DOI: 10.1016/S0003-9365(11)80300-3
14 Eaton, W. D., Chassot, O. “Characterization of soil ecosystems in Costa Rica using microbial community metrics”. Tropical Ecology, 2012, 53 (185-195)
15 Pons, T. L., Perreijn, K., Van Kessel, C., Weger, M. J. A. “Symbiotic nitrogen fixation in a tropical rainforest: 15N natural abundance measurements supported by experimental isotopic enrichment”. New Phytologist, 2007, 173:1 (154–167). DOI: 10.1111/j.1469-8137.2006.01895.x