Phage Therapy for Drug-Resistant Pathogens
Introduction

By Katie Orefice
Phage therapy, or the use of bacteriophages to infect bacteria, is a defense against drug-resistant pathogens. Typically, antibiotics are used in order to combat bacterial infections and have had a lot of success. Although, when antibiotics are used often, bacteria is capable of developing a resistance to the drug, rendering it much less effective. A newer potential defense of pathogens is phage therapy. Phage therapy uses bacteriophages, which are viruses that target and infect bacteria. Each type of bacteria is susceptible to a number of bacteriophages [1]. When using a bacteriophage to target a pathogen, bacterial lysis often occurs, leading to the breakdown of a cell’s membrane and bursting of the cell [1]. This would be a successful result in targeting a pathogen. The use of phage therapy actually dates back to the early 1900s, even before antibiotics were discovered. Once antibiotics were widely used, phage therapy dissipated but now that bacteria are developing drug-resistant characteristics, the use of phage therapy has gained a renewed interest and is continuously expanding.
[1] [2] [3] [4] [5] [6] [7] [8] [9]
Bacteriophage Function
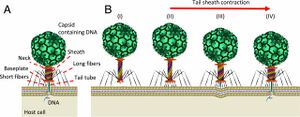
Bacteriophages are known as viruses that specifically infect bacteria. In terms of human health, bacteriophages are present in the intestinal community, which help with digestion, immune system function, and mental health [1].
In order for bacteriophages to function in combating bacteria, they must begin an infection cycle. To start, they attach to the surface of a host cell, which is allowed by certain proteins on the host cell surface, called cell-surface receptors [1]. This protein described actually performs a very important function for the host cell, but the bacteriophage has evolved to use it to its advantage, such as attachment. Once attached, the phage injects its genome into the cell through the cell envelope. Once the phage’s DNA is inside the cell, replication in a lytic cycle begins. As many progeny phages are assembled as possible in this lytic cycle [1]. Not only are the phage genomes replicated, but the corresponding enzymes and proteins are also assembled. Once enough progeny phages are created, the host cell lyses, meaning that the cell wall bursts which releases the progeny [1]. The phage then inserts its DNA into the host cell’s cytoplasm, leading to the expression of phage genes by the host cell RNA polymerase and ribosomes [1]. Phage genomes are continuously replicated, along with enzymes and ribosomes that then produce the phage capsid proteins. Lastly, the phage genome expresses an enzyme that lyses the host cell wall, which releases these completed virus particles [1]. With the destruction of the host cell wall, this leads to the destruction of the cell, and therefore the bacteria, making it clear that bacteriophages can be instrumental in the killing of bacteria.
Antibiotic Resistance and Phage Therapy Application
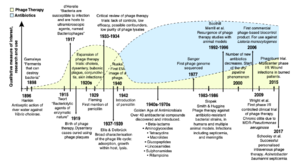
Antibiotics have been widely used to combat bacterial infections, although it is well known that bacteria are capable of developing resistance to certain antibiotic medications. This is a major threat to global health due to the increasing antibiotic resistance levels due to antibiotic overuse. There are various mechanisms that lead to antibiotic resistance in pathogens, such as drug target modification, molecular bypass, active efflux, and the chemical modification of the compound [2]. In drug target modification, it is possible that a point mutation occurs in specific genes, which can change the amino acid that is present, leading to an alteration in protein structure, preventing the binding of antibiotics and therefore antibiotic function [2]. In molecular bypass, microbes can avoid antibiotic action through specific avoidance mechanisms. For example, in vancomycin resistance, the substitution of an amide bond of D-Ala-D-Ala with an ester linkage gets rid of a hydrogen bond donor, causing electronic repulsion which therefore prevents binding of the antibiotic, one example of avoiding antibiotic function without mutations [2]. There is also active efflux, which is essentially the removal of antibiotics from the cell. Proteins are in charge of this removal, along with proton pumps [2]. Lastly, in chemical modification, enzymes catalyze the inactivation of specific antibiotics [2]. Through the various, complex mechanisms of antibiotic resistance, it is clear that other manners of pathogen defense will be necessary to combat these antibiotic-resistance bacteria.
One possibility when exploring alternatives to antibiotics in bacterial defense is phage (bacteriophage) therapy. Phage therapy was actually first described in 1907 by Félix d’Herelle in France, even before antibiotics were discovered [3]. It was explained that every type of bacteria is susceptible to a certain limited number of specific types of phages [3]. Due to the fact that when bacteria are infected by bacteriophages, it often leads to lysis, this technique was considered feasible for the treatment of bacterial infections, as long as the phage specific was to the pathogen [3]. There was some success and some failure when the experimentation began, as phages were used to treat various illnesses such as cholera or typhoid fever, by giving a patient phage in an open wound, or orally provided through aerosol or injection [3]. As soon as antibiotics were discovered and had success, phage therapy was placed aside, but it has become much more relevant today with the increase in antibiotic resistance.
Phage Therapy Approaches and Applications
There are various approaches to phage therapy delivery, one being monophage therapy, which points to the use of a single phage. Monophage therapy has seen success in the laboratory setting, specifically with animals [4]. Although it has been successful, this technique is hampered by a few drawbacks, one being that phages must be specific to the bacteria, so pinpointing one singular phage may cause difficulty [4]. Another disadvantage is the possibility of bacteria becoming resistant to the phage in monophage therapy. This highlights the importance of polyphage therapy, where multiple different phages are used in a phage cocktail [4]. The evolution of bacterial resistance can happen relatively quickly, and therefore it is important that this is avoided as much as possible [5]. A phage cocktail is often used, which combines phages in order to target multiple strains of the same bacterial species, or multiple species [5]. Although essential to avoid bacterial resistance, using multiple phages may lead to complexities in production or safety questions. Due to the fact that phages are specific to each bacteria, it is also important to not include a random mixture of phages, but instead an optimized phage usage that is specific to the knowledge of bacterial receptors [5]. One example of this would be using phages that each target a different receptor in the same bacterial host, which in turn will reduce the likelihood and speed of phage resistance. Another strategy to minimize resistance is by combining both monophage therapy and polyphage therapy in a sequential order [4]. By administering monophage and polyphage therapies at different times in treatment, the bacteria will be continuously exposed to new phages, making it harder to develop resistance, while also keeping the bacteria at a low level so that the immune system can take effect [4]. Currently, phage cocktails are available as over-the-counter medicine in Russia and Georgia for bacterial infections [4]. Two well-known generic phage cocktails, Pyophage (PYO) and Intestiphage have been used. Pyophage targets at least 6 common bacteria, while Intestiphage targets about 23 different enteric, or disease-causing, bacteria [4]. Although not yet approved in Western countries, the study of polyphage therapy effectiveness has increased and continues to be subjective to clinical studies.
It is often very effective in a clinical setting to use more than one medicinal therapy in fighting a pathogen. It has commonly been in practice to combine phage therapy with antibiotic use in order to combat bacterial infections. The term phage-antibiotic synergy (PAS) has been used to describe the combinations of phage therapy and antibiotic use [4]. This has been used in previous studies, showing that while using antibiotics, this could increase the bacterial production of lytic phages [4]. There is even more potential for this strategy for a variety of bacterial species in the order of treatment.
It has been demonstrated that treatment with bacteriophages prior to antibiotics may lead to the most efficient killing of bacteria [4]. Optimizing the timing of phage-antibiotic synergy may be successful in improving efficacy [4]. By using a combination of phages and antibiotics, this limits the amount of antibiotic used and therefore decreases the likelihood of developed resistance [4]. Secondly, a combination of the two treatments would give antibiotics a restored strength against bacteria [4]. The effectiveness of the combination treatment has been demonstrated in broiler chickens infected with E. coli [6]. With solely an antibiotic, specifically enrofloxacin, mortality was decreased by 65%. Phage treatment alone decreased mortality by 53% [6]. The combination of phage therapy and enrofloxacin resulted in no mortality, demonstrating the clear effectiveness of phage-antibiotic synergy treatment [6]. Another study observed a 10,000-fold greater reduction in bacterial load with phage and ciprofloxacin rather than each treatment alone in rats with P. aeruginosa [6]. There have been very few clinical trials experimenting with phage-antibiotic synergy, but the combination has demonstrated significant potential in the treatment of bacterial infections in vivo.
Genetic modification has demonstrated large potential in terms of the medical field, and specifically with bacteriophage treatment. In genetically modifying pre-existing biological systems to perform tasks that are not typically done are one way that phage therapy may progress in the future. In bioengineering phages, they can become more dynamic in circumstances such as expanded host range, switching host tropism, or modification of phage capsids [4]. One study was able to genetically modify the host range of E. coli phage T2 by incorporating long tail fiber genes of a different phage through homologous recombination [4]. In doing so, the host range of E. coli phage T2 expanded, while also keeping its success in lytic activity [4], demonstrating successful usage of bioengineering in phage therapy. Another study altered E. coli phage T7 to express the enzyme dispersin B, which can degrade a key component of bacterial biofilms [4]. This engineered virus was able to reduce biofilm cell counts by >100-fold compared to the original, wild-type phage [4]. It is clear that bioengineering technology can lead to successful treatment of bacterial infections, but further investigation of bioengineered phage safety and efficacy is required prior to its regulatory usage.
Phage bioengineering may also have an impact on vaccine development. In phage display, the vaccine sequence, known as the antigen, can be cloned into a phage capsid protein [4]. The antigen is then displayed on the phage surface and can then be demonstrated to the immune system, which would lead to the production of antibodies [4]. In this situation, the phage acts as a vaccine delivery system. Phages would likely be successful in vaccine delivery due to the fact that they are able to survive harsh environmental conditions [8]. The potential for phage-based vaccine development is promising in the use against various diseases caused by microbial infections [8].
Efficacy and Safety
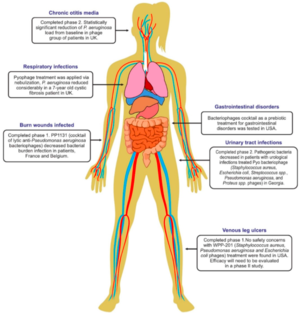
The first clinical trial of phage therapy conducted according to good manufacturing practices (GMP) and good clinical practices (GCP), or FDA approval was from 2015 to 2017 where burn wound infections of P. aeruginosa were treated in 25 patients involving a cocktail of 12 phages [4]. This led to no unwanted side effects, and less bacterial load in the wounds, but this did not work as well as the control, meaning that the phages did not help more than a typical antibiotic did. This could’ve been explained by the small sample size, the titer of phage decreased from manufacturing to usage, or the possibility that the bacteria were resistant to lower doses of phage, leading to the need for more future studies [4]. The oral administration of phages has also been conducted against E. coli, in healthy adults in 2005 and 2012, and again in 2017. There were no harmful effects from this oral administration. It was seen that the phages did not replicate in the gut of healthy and infected individuals, meaning there was little to no evidence of the phages or phage-specific antibodies in the bloodstream, meaning it was less successful [4]. Intravenous phage therapy, which is delivered through the veins, was studied in its effect on typhoid fever and S. aureus bacteria. With reports over the past 80 years, 1,000 patients were successfully treated with no significant side effects. One successful use of intravenous phage therapy occurred in 2017 with a patient who had necrotizing pancreatitis with an MDR A. baumannii infection [4]. Phage therapy was initiated when antibiotics were not working. The use of a phage cocktail, which has many types of phages was issued in order to avoid phage resistance, and was successfully tolerated by the patient [4].
It is possible that patients who are terminally ill without any alternative treatments may use phage therapy with “off-license” approval [4]. In 2016 with Tom Patterson who had been in a coma for months due an infection by drug-resistant Acinetobacter baumannii, the FDA granted emergency approval and a phage solution that targets A. baumannii was injected intravenously. Shortly after, Dr. Patterson was much improved and came out of his coma [3]. This success led to the approval of a clinical study to further examine the safety and efficacy of phage therapy for patients specifically with ventricular assist devices with Staphylococcus aureus infections in 2019 [3]. Other examples of emergency uses of phage therapy have been evident in other bacterial infections of Pseudomonas, P. aeruginosa, A. baumanii, and more [6]. One case in 2011 reported the treatment of a P. aeruginosa urinary tract infection associated with a bilateral ureteral stent [6]. After the use of antibiotics, the infection was recurring, and the use of a specific phage was identified for the specific bacteria. The phage cocktail, consisting of 6 different bacteria including P. aeruginosa was successful in reporting a 10-fold reduction of bacteria in urine after 5 days of the phage treatment [6].

More antibiotics usage cleared the infection and the urine remained sterile for one year, concluding observations [6]. Another case study of emergency phage treatment was with an aortic graft infection after surgical intervention to repair an aortic aneurysm with a graft [6]. The P. aeruginosa infection was treated with antibiotics, but resistance to the ciprofloxacin evolved during treatment that took place over 3 years. Phage was then combined with ceftazidime and applied topically. The treatment was successful and the recurrence of the P. aeruginosa was negative after two years without any antibiotic treatment [6]. This positive outcome was the result of the combination of antibiotics and phage that was specific to the bacteria.
The success of emergency phage treatment led to the beginning of clinical trials that went on to investigate the safety and efficacy of phage treatment. One small clinical trial investigated the use of a phage cocktail in treating an external ear infection of P. aeruginosa [9]. The phage cocktail was administered with no adverse effects reported. P. aeruginosa levels decreased by less than 10-fold for the phage group as compared to the placebo group, who received typical treatment [9]. Another trial involved the testing of two different phage cocktails to target E. coli, which causes diarrheal diseases involving individuals exhibiting symptoms [6]. One phage cocktail containing 11 phages were used, along with a commercial phage cocktail from Russia with at least 17 different phages [6]. Standard treatment was given to the control group. The results did not demonstrate significant differences in bacterial count between both groups, potentially due to phage particles that were unable to survive in the stomach environment [6]. Trials have indicated that phages work under specific conditions and have not demonstrated adverse effects [9] , highlighting the importance of continuing trials to learn more about the efficacy of phages and how they can be applied in different environments with various bacterial infections.
As phage therapy continues to progress and potentially become a major aspect of the clinical world, safety is a large consideration when dealing with a potential treatment for bacterial infections. There has been much success in emergency phage treatments, and other successes in clinical trials that have not shown adverse effects. Side effects that did occur were typically present in non-associated situations with the use of bacteriophages [7]. This may also be due to the exposure to phages that commonly occurs in drinking water and food on a daily basis, as phages are present in large amounts in these daily occurrences [7]. Finally, although bacteriophage treatment has largely proven to be safe, it is important that clinical trials leading to regulation of phage treatment involves these safety protocols.
Conclusion
Phage therapy demonstrates massive potential, not only in the medical field but in aspects of agriculture, ecology, and overall public health. Research on the applications of phage therapy is continuing and further depicts possibilities in terms of the expansion of antibiotic resistance. In terms of the medical field, antibiotics have been very successful in the treatment of bacterial infections, but as bacterial resistance is on the rise, so are alternatives of combating these pathogenic infections. Whether through the use of the combination of phage cocktails with antibiotics or genetically modified phages, bacteriophages offer many possibilities for the future of medicine and in combating pathogenic infections.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Slonczewski, J. L., & Foster, J. W. (2017). Viruses. In Microbiology an evolving science. New York: W.W. Norton & Company. Retrieved March 16, 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Wright, G. (2011, February 01). Molecular mechanisms of antibiotic resistance. Retrieved April 08, 2021. https://pubs.rsc.org/en/content/articlelanding/2011/cc/c0cc05111j/unauth#!divAbstract]
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 Slonczewski, J. L., & Foster, J. W. (2017). Environmental Influences and Control of Microbial Growth. In Microbiology an evolving science. New York: W.W. Norton & Company.]
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 4.14 4.15 4.16 4.17 4.18 4.19 4.20 4.21 4.22 4.23 4.24 4.25 4.26 Fernando L. Gordillo Altamirano, Jeremy J. Barr. Phage Therapy in the Postantibiotic Era. Clinical Microbiology Reviews Jan 2019, 32 (2) e00066-18; DOI: 10.1128/CMR.00066-18]
- ↑ 5.0 5.1 5.2 5.3 Fernando L Gordillo Altamirano, Jeremy J Barr, Unlocking the next generation of phage therapy: the key is in the receptors, Current Opinion in Biotechnology, Volume 68, 2021, Pages 115-123, ISSN 0958-1669, https://doi.org/10.1016/j.copbio.2020.10.002.]
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 Kaitlyn E. Kortright, Benjamin K. Chan, Jonathan L. Koff, Paul E. Turner, Phage Therapy: A Renewed Approach to Combat Antibiotic-Resistant Bacteria, Cell Host & Microbe, Volume 25, Issue 2, 2019, Pages 219-232, ISSN 1931-3128, https://doi.org/10.1016/j.chom.2019.01.014.]
- ↑ 7.0 7.1 7.2 Qadir, M. (n.d.). Phage therapy: A modern tool to control bacterial infections. Retrieved April 07, 2021, from https://www.researchgate.net/profile/M-Qadir/publication/267877606_Phage_therapy_A_modern_tool_to_control_bacterial_infections/links/552b741a0cf2e089a3aa30d3/Phage-therapy-A-modern-tool-to-control-bacterial-infections.pdf]
- ↑ 8.0 8.1 8.2 Aghebati-Maleki, L., Bakhshinejad, B., Baradaran, B. et al. Phage display as a promising approach for vaccine development. J Biomed Sci 23, 66 (2016). https://doi.org/10.1186/s12929-016-0285-9]
- ↑ 9.0 9.1 9.2 9.3 Vandenheuvel, D. (n.d.). Bacteriophage therapy: Advances in formulation strategies and human clinical trials. Retrieved April 08, 2021, from https://www.annualreviews.org/doi/10.1146/annurev-virology-100114-054915
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2021, Kenyon College.
