Pyrobaculum arsenaticum: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{ | {{Moore2013}} | ||
===Classification=== | ===Classification=== | ||
Latest revision as of 15:50, 22 February 2016
Classification
Higher order taxa
Archaea; Crenarchaeota; Thermoprotei; Thermoproteales;Thermoproteaceae
Species
Pyrobaculum arsenaticum
Description and significance
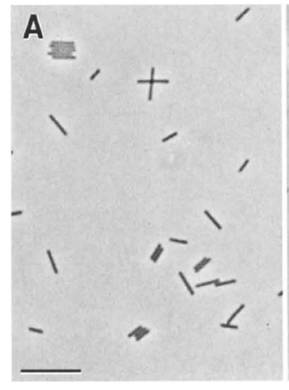
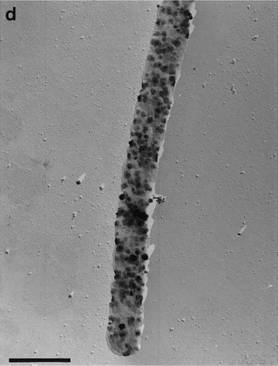
The Archaean Pyrobaculum arsenaticum is a free living microbe, which can be found within the hot springs of Pisciarelli Salfataras in Naples, Italy. P. arsenaticum is gram negative, and its cells are cylindrical shaped rods with rectangular ends. The average length of each cell is 4 μm, with most cells ranging from 3 μm to 7 μm. was the first Pyrobaculum arsenaticum hyperthermophylic microorganism shown to grow chemolithoautotrophically using arsenate as an electron acceptor with hydrogen. (1)
Genome structure
The genome of Pyrobaculum arsenaticum has been measured 2,121,076 base pairs long contained in a single circular chromosome. Further analysis has revealed 2,408 predicted genes and a C+G content of 58.3 mol%. (2)
Cell and colony structure
Cells of Pyrobaculum arsenaticum are cylinder shaped rods averaging 4 μm in length and 0.7 μm wide. While most cells range from 3 μm to 7 μm in length, cells up to a length of 20 μm have been observed. While colonies are growing, rods are often seen arranged in V-, X- or raft-shaped aggregates When colonies are grown with both acceptors present, thiosulfate and arsenate, a precipitate forms. The precipitate, known as realgar [As2S2], is seen as a yellow-orange flocculent mass in the culture medium.
Metabolism
This species is characterized as a hyperthermophylic, facultative autotroph. During anaerobic respiration, Pyrobaculum arsenaticum uses hydrogen as an electron source and reduces arsenate to form arsenite. As a lithoautotroph, CO2 is used as a carbon source. Elemental sulfur can also be utilized as an alternate electron acceptor for autotrophic growth. When grown organotrophically, Pyrobaculum arsenaticum uses arsenate and thiosulfate or arsenate and cysteine as terminal electron acceptor, resulting in the production of arsenic sulfide minerals called realgar. Pyrobaculum arsenaticum is the first species to indicate that such sulfur compounds could be generated biologically, which is evidence that the microbes may play a key role in the bio-geochemical cycling of arsenic and sulfur in hydrothermal areas. (3)
Ecology
Pyrobaculum arsenaticum was discovered in the year 2000 after it was isolated from the high temperature environment of hot springs, specifically those of Naples, Italy. Further studies have shown that Pyrobaculum arsenaticum grows optimally at 90C, and is able to grow at temperatures ranging from 68C to 100C. At 65C and 102C Pyrobaculum arsenaticum is unable to grow. Pyrobaculum arsenaticum is capable of growth in slightly saline enviroments, between 0% and 3% NaCl concentration.(1)
References
(1)Dworkin,Martin. The Prokaryotes: Vol. 3: Archaea. Bacteria: Firmicutes, Actinomycetes. Springer, 2006
(2)Ehrlich, H. 2009 Geomicrobiology. Fifth Edition. Taylor and Francis Group, LLC. 14:254-256
(3)Huber, R. et al. 2000. Respiration of arsenate and selenite by hyperthermophylic archaea. Systematic and Applied Microbiology. 23:305-314
(4)Huber R, Huber H, Stetter KO (2000) Towards the ecology of hyperthermophiles: biotopes, new isolation strategies and novel metabolic properties. FEMS Microbiol Rev 24:615–623
(5)Kegg Genome. http://www.genome.jp/kegg-bin/show_organism?org=pas
Edited by Robert Walsh of Dr. Lisa R. Moore, University of Southern Maine, Department of Biological Sciences, http://www.usm.maine.edu/bio