Reptile-Exotic-Pet-Associated-Salmonellosis
By Tomas Grant
Introduction
Salmonellosis is one of the most common foodbourne diseases and can be caused by the various serovars of the species S. enterica , and is found worldwide, causing 93.8 million cases of gastroenteritis and over 155,00 deaths annually worldwide (Gay et el, 2014). Understanding the pathology of foodbourne Salmonellosis is important however, there is an increasing concern for Reptile-Exotic-Pet-Associated-Salmonellosis (REPAS) , a major agent of infection through direct or indirect animal contact in people’s homes, veterinary clinics, farms, zoological gardens, and other professional, public and private settings. (Rabsch et, al, 2014). There are a number of reports that describe the prevalence of Salmonella spp. in reptiles ranging from lizards, snakes, and turtles, and multiple serovars have been identified and shown to be associated with each of these organisms (Sylvester et al, 2014). Reptiles have been shown to be asymptomatic carriers of Salmonella and have been shown to have a natural interaction with the bacteria, with these bacteria being natural inhabitants of reptile gut microflora, shed at various rates throughout the reptile’s lifecycle. Reptiles are often unrecognized sources for the disease in humans and there are estimates that indicate reptile-associated salmonellosis is responsible for 3-11% of all human salmonellosis cases within both Canada and the United States (Rabsch et al, 2014).
Classification
Salmonella spp. are rod-shaped, flagellated, and facultative anaerobic bacteria which belong to the family Enterobacteriaceae, and as apart of the phylum Proteobacteria are Gram-negative. Within the genus Salmonella there are two distinct species; S. enterica and S. bongori , but the majority of the diversity lies within the more than 2,600 servovars of the species S. enterica . The three main serovars within this species are (1) S. typhi, the cause of systemic infections and typhoid fever, (2) S. Enteritidis, a major food cause of food poisoning associated with poultry farming, and (3) S. typhimurium, which is not fatal in humans, but may cause gastroenteritis, but also more serious cases including septicemia, meningitis, and subnormal empyema (Rabsch et, al, 2014).
Higher order taxa:
Kingdom:Bacteria
Phylum: Proteobacteria
Order: Gammaproteobacteria
Suborder: Enterobacteriales
Family: Enterobacteriaceae
Genus: Salmonella
Species:
Salmonella enterica subsp. I serovar Typhimurium (S. typhimurium LT2), S. enterica subsp. enterica serovar Typhi (S. typhi CT18), S. enterica subsp. enterica serovar Typhi Ty2 (S. typhi Ty2)
NCBI: Taxonomy Genome: S. enterica S. typhi Ty2 S. typhimurium| (McClelland et al, 2001)Epidemiology
Transmission of Salmonella
The main pathway of transmission for Salmonella enterica spp., which is an invasive primary pathogen, is by the fecal-oral route which usually begins with the ingestion of contaminated water or food, so that the bacterium is able to encounter the intestinal epithelium, spreading throughout the gastrointestinal tract causing gastroenteritis (Figure 1). The bacteria must reach the small intestine after being ingested and then must adhere to and then break through the stomach lining, where it is capable of reproducing and then subsequently spreading to other tissues in the body. Humans typically acquire the organisms through either direct transmission by handling a reptile, and via indirect transmission by coming into contact with any object that has been contaminated by a reptile. Other sources of transmission include aquariums, terrariums, cages, as well as the water that most reptiles like turtles and amphibians live in (CDC, Jan 2013). Clothing that has come into direct contact with reptiles has been associated as another source of transmission from one infected individual, and also bites and claw scratches have been shown to be effective at transmitting Salmonella spp.
Salmonella has ben shown to be able to survive for long periods of time in the environment and prefers those environments where it is humid and warm, and has been isolated for prolonged periods from a multitude of surfaces that have been contaminated by reptile fecal matter (CDC, Nov 2013). It is of important note to highlight the fact that the CDC found that Salmonella has been reported to “survive 115 days in pond water, 89 days in tap water, within dried reptile feces, and from aquariums and terrarium water up to 6 weeks after removal of a turtle from the habitat (CDC, Jan 2013). The disease can be transmitted to other individuals, both human and animal alike in fecal matter, with the organisms being shed continually throughout the duration of the infection. Shedding has been shown to last anywhere from several days to multiple weeks, with the ability of certain individuals to become asymptomatic carriers for several months. It has been shown that non-typhoidal Salmonellosis infection patients have the ability to shed the bacteria in their feces for over a year, and antibiotic treatment has been shown to typically prolong the shedding of these organisms during this time period (CDC, Jan 2013). Also, over 90% of reptiles may carry Salmonella and these animals may carry up to five different serotypes, increasing the risk associated with reptiles as a whole. Also,it is important to note that recent studies have shown that shedding of Salmonella within the feces is higher in reptiles that are kept in captivity when compared to wild reptiles (Rabsch et al, 2014), which is especially important because these “pet” reptiles are kept in closer proximity and are in closer contact to humans. These findings alone show the danger associated with such a long living organism and help demonstrate the prevalence of the disease Salmonellosis and the dangers associated with Salmonella as a world-wide issue.
Pathology and Immune Response
Clinical Signs of Infection
Patients infected by Salmonella typically present a multitude of symptoms including an acute onset of fever, abdominal pain, cramping, as well as diarrhea with or without blood (typically associated with inflammation of the large bowel), as well as vomiting and severe nausea, but there is a wide spectrum of severity of the illness which may be indicated by only a few or a multitude of symptoms (Fabrega and Vila, 2013). The disease usually manifests after the ingestion of >45,000 bacteria in contaminated food, water, or other sources of infection and non-typhoidal, self-limiting, Salmonella gastroenteritis requires a period of incubation ranging from 6 to 72 hours depending on susceptibility of the host (CDC, Nov 2013). Whether or not Salmonella remains within the intestine or disseminates to other major organs depends mainly on the host’s immune response as well as the virulence factors the strain possesses.
Pathogenesis of Salmonella serovar S. Typhimurium
The pathogenesis triggered by the Salmonella serovar S. Typhimurium has been studied extensively and knowledge about its multitude of virulence mechanisms for successfully invading host tissue cells has been compiled. These organisms have a large armamentarium of virulence factors all regulated by an extremely complex regulatory meshwork, responsible for coordinating and synchronizing all the time sensitive elements involved. This regulatory mechanism is extremely important because it guarantees expression of each virulence element, but it also facilitates a communication between all of the determinants, ensuring the invading bacteria responds appropriately at all stages in a temporal hierarchal method. These elements include Salmonella pathogenicity islands (SPI’s), as well as other virulence determinants, including those encoded within the pSLT virulence plasmid, as well as adhesions, flagella, and biofilm forming proteins which all contribute to several stages of the disease (Fabrega and Vila, 2013).
The first step in the infection pathway of Salmonella Typhimurium involves host ingestion of Salmonella found in contaminated food or water as mentioned before, and the subsequent invasion of the stomach which presents the first obstacle to successful infection: the highly acidic pH of the host organism’s stomach. In order to overcome this obstacle, S. Typhimurium activates the acid tolerance response (ATR) system, which provides an inducible pH-homeostatic function in order to maintain the organism’s intracellular pH at levels higher than those found in the stomach (Lahiri et al, 2010). Upon entry into the small intestine the organism must adhere to and then cross the intestinal mucus layer, the second line of defense against pathogens, and then must adhere to intestinal epithelial cells, causing the appearance of the infection in the form of large cytoskeletal rearrangements within host tissue cells. An important feature of these bacteria are the presence of flagella which gives Salmonella an increased chance at encountering the intestinal epithelium, leading to subsequent adhesion and invasion of host cell tissues. The intestinal epithelium is the primary interface between the internal and external environment, and the adhesion of Salmonella is a key factor in its pathogenicity, or its ability to cause disease. Approach and attachment are enhanced by the motility of the organism, which are driven by several virulence factors including multiple adhesins and fimbriae.
The internal modifications induced within host cells serves to disrupt normal epithelial brush borders and induces the formation of membrane ruffles. These ruffles are responsible for engulfing the adhered bacterium in large vesicles called Salmonella-containing vacuoles (SCVs) (Broz et al, 2012). Engulfment of Salmonella is mediated by virulence elements encoded within SP1-I, which encodes for several effector proteins responsible for triggering the invasion of epithelial cells within the membrane, and regulates actin cytoskeletal rearrangements which leads to the intake of these bacteria (Figure1- 2,3,5). (Fabrega and Vila, 2013) These SVC’s are the most important determinants of Salmonella pathogenicity because they represent the intracellular compartments in which these organisms can replicate,and highlights the facultative intracellular nature of these bacteria. SVC’s arise during the early endocytic pathway, but must direct changes within host endocytic trafficking and function to avoid direct fusion of SVC’s with secondary lysomsomes and halt activity of lysosomal enzymes. An important mechanism for the maintenance of the integrity of the SVC membrane is the Salmonella induced formation of an F-actin meshwork which surrounds bacterial vacuoles, a process known as vacuole-associated actin polymerization (VAP) (Fabrega et al, 2013). This not only protects the bacterial SVC’s, but allows for the migration of these vacuoles to the perinuclear position near the Golgi apparatus, which allows for the interception of host exo- and endo-cytic transport vesicles to obtain nutrients essential for supporting bacterial replication (Broz et al, 2012).
While initially forming the SVC’s, the invading Salmonella bacterium induces a secretory response within the intestinal epithelium that aims to recruit and transmigrate phagocytes located in the submucosal space across the membrane and into intestinal lumen, followed by the reconstitution of the once disrupted epithelial brush border. Those phagocytes now in the intestinal lumen can be used to engulf individual cells and transport them directly across the membrane. A number of SVC’s also migrate to the basolateral membrane, release their internal cells into the submucosa, and then engulfed by phagocytes. More specifically three phagocytic types have been reported to interact directly with invading Salmonella bacteria including (i) neutrophils, (ii) inflammatory monocytes, and (iii) dendritic cells (Figure 1- steps 6 and 7) (Fabrega and Vila, 2013). These bacteria are then internalized again within an SCV allowing for further replication, and the migration of infected phagocytes allows for the spread of the invading bacteria, and subsequent circulation in the host’s bloodstream to several tissues including the spleen, liver, and in extreme cases the brain.
Importance of PLEKHM1 in Salmonella Containing Vacuole Biogenesis and infection
As mentioned before, the Salmonella containing vacuoles formed during the pathogenic pathway taken by Salmonella is an important factor in ensuring invading bacteria are capable of replicating and continuing infection. An important step in the formation and maintenance of this compartment is the avoidance of the host endolysosomal pathway and subsequent fusion with the lysosome. Current research is aimed at examining the mechanisms by which Salmonella manipulates the endocytotic pathway and is interested in looking at the molecular basis by which this manipulation occurs. Salmonella has been shown to secrete a number of effector proteins, including SifA, through a specialized secretion system into the cytosol, which works to hijack the host endosomal system and helps generate the SVC. Sifs, orSalmonellainduced filaments, require a continuous supply of membranes and are central to the survival of the bacteria inside of the host, and is a key factor along with maintenance of the integrity of the SVC. An important enzyme in this pathway is Rab7 GTPase which is targeted by Salmonella and used to recruit host membranes through mechanisms which remain unknown, and the take over of this enzyme allows for SVC maturation. The Rab family of small guanine nucleotide binding proteins are crucial for mediating membrane trafficking within host cells, and include a number of proteins including kinases, tethering factors, and motor proteins (McEwan et al, 2015).
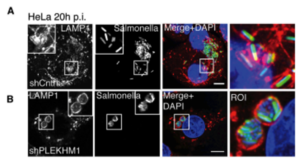
A study by McEwan et al, 2015 was interested in examining the role of Pleckstrin homology domain-containing protein 1 (PLEKHM1) in the recruitment of phagosomes and other membranes for maintenance of the SVC. PLEKHM1 is a lysosomal adaptor and it was speculated that there was an interaction between SifA and PLEKHM1 and other host proteins for the recruitment of Rab7. The research examined two different domains of PLEKHM1, plekstrin homology domains PH1 and PH2, and using cell culture (SILAC)-based mass spectrometry analyses they found that direct interaction with SifA is achieved through the second PH domain (PH2) and that PLEKHM1 itself has direct interaction with active Rab7 GTPase (McEwan et al, 2015). PLEKHM1 is targeted by Salmonella, and the PLEKHM1-SifA interaction is required for recruitment of the endolysosomal membrane pools that are essential for the growth of the SVC and proliferation in primary cells and tissues of the host. The depletion of PLEKHM1 caused a profound defect in SCV morphology, with multiple bacteria accumulating in enlarged structures and significantly hinders Salmonella proliferation in multiple cell types and mice (Figure 3) In the figure you can se a clear difference between PLEKHM1 competent cells (Figure 4, 3rd panel top row), versus PLEKHM1 deficient cells (Figure 4, 3rd panel bottom row) with noticeable differences in the actin filament network surrounding the SVC's, indicating a loss of the ability to recruit host membranes in PLEKHM1 deficient tissues. These results also highlight the differences in bacterial concentration between the cells, with PLEKHM1 cells showing cells bunching up within very small and packed SVC's as a result of the loss of this lysosomal adaptor's function and ability to interact with Rab7 GTPase and recruit host membranes. These results taken together indicate that Salmonella utilizes a complex containing PLEKHM1 and Rab7 to mobilize phagolysosomal membranes to the SCV, and highlight the importance of PLEKHM1 i in provides a critical interface between infection by the pathogen and the host endolysosomal system.
Antibiotic Resistance and Treatment
Treatment for salmonellosis can involve a multitude of strategies and dependson the patient’s symptoms and the severity of the illness .For individuals with gut-limited non-typhoidal salmonellosis (NTS) infections, they typically exhibit substantial fluid loss and must undertake oral or intravenous rehydration to treat fluid and electrolyte imbalances, and symptoms typically last for 5 to 7 days after treatment. Typical treatment for invasive Salmonellosis involves a cocktail of antibiotics including but not limited to ampicillin, trimethoprim-sulfamethoxazole (TMP-SMZ), and flouroquinolones. Antimicrobial therapy is typically used for enteric fever, septicemia or focal extra intestinal infections, which may require surgery and an extended treatment with antibiotics. Antibiotics can also increase the period of shedding of these organisms and the excessive use antibiotics has caused an increase in the development of new antibiotic resistant strains (CDC, Jan 2013). Also, antibiotics are extremely useful when there is a need for the interruption of shedding within feces in order to manage outbreaks of salmonellosis in hospitals.
The armamentarium of virulence factors that serovars of the Salmonella enterica species has allowed for an increased number in antibiotic resistance strains, and more specifically there has been a rise in the number of S. enteric subsp.I serovar Typhimurium resistant strains isolated around the world (Sylvester et al, 2013). S. Typhimurium has been shown to be resistant to ampicillin and TMP-SMZ, leading to a decreased use of both antibiotics. Even more concerning to public health is the appearance of multidrug resistant strains, which shows resistance to multiple antimicrobial agents, specifically those isolates within S. Typhimurium isolates exhibiting multi drug resistance (>55%) (Fabrega and Vila, 2013). The spread of the MDR phenotype is caused by the replication of resistant clonal strains, which typically carry several genes within their genomes which confer ACSSuT (ampicillin, chloramphenicol, streptomycin, sulfonamides, and tetracycline) resistance type as a result of horizontal gene transfer. Also, certain organisms possess hybrid plasmids which can also disseminate and confer antibiotic resistance in a similar fashion, with strains resistant to ampicillin, sulfonamides, tetracycline, and chloramphenicol detected in Spain, the United Kingdom, and other European countries (Sylvester et al, 2013).
Risks Associated With S. enterica infections
Risk Factors
There are a multitude of risks associated with non-typhoidal salmonella (NTS) infections and the severity and complications associated with the disease depends on host susceptibility as well as host-pathogen interactions. Salmonella infections within immunocompetent hosts causes self-limiting diarrhea which has an untreated mortality rate of approximately 0.1%. However, there a multitude of risk factors for non-typhoidal salmonella gastroenteric disease including age, alterations within the endogenous gut flora, achlorhydria (low/absent production of gastric acid in the stomach), previous gastric surgery, diabetes. More importantly, there is an increased risk for complications in an immunocompromised host including the possibility for more severe and invasive nature of the disease within these individuals.

More serious cases of salmonellosis have been reported and approximately 5% of individuals with NTS develop bacteremia, or the presence of bacteria in the blood, which is a serious and potentially fatal issue (Fabrega and Vila, 2013). This disease is more likely to manifest itself in young children, immunocompromised individuals, or those individuals suffering from comorbid medical conditions like HIV, malaria, or malnutrition. The development of bacteremia within these hosts allows Salmonella the opportunity to spread to other tissues throughout the host, increasing the likelihood of developing a focal infection. These infections include septic arthritis, osteomyelitis, cholangitis, meningitis, and even pneumonia. A recent study by Pees et al, conducted in Germany, highlights the risk associated with Salmonellosis within children within a number of households with reptiles present. Recent reports clearly indicated an increase in the number of Salmonella infections in children younger than three years old with serovars associated with reptiles (Figure 3). The aim of the study was to establish possible connections between captive reptiles and salmonellosis in children by both examining children with salmonellosis and all of the organisms kept in their respective homes. Out of a total of 19 examined households, an identical serovar of Salmonella to that of the salmonellosis-affected child was found in at least one reptile as confirmed by biochemical typing, indicating a major role of reptiles in the transmission of Salmonella to children (Pees et al, 2013).
Disease Prevention Measures
There are a number of disease prevention measures that can be implemented to avoid the contraction and spread of Reptile-Exotic-Pet-Associated-Salmonellosis, but the simplest step is disinfection. Many disinfectants including 1% sodium hypochlorite, 2% glutaraldehye, 70% ethanol, iodine-based disinfectants, formaldehyde, and phenolics are able to successfully kill these bacteria (CDC Jan 2013). Salmonella organisms are also susceptible to both moist (121 degrees Celsius for >10 min.) and dry heat (160-170 degrees Celsius for a minimum of 1 hr.).
Besides simple disinfection, the Center for Disease Control and Prevention (CDC) has issued recommendations for the handling of reptiles by their owners, and some states also have implemented laws requiring owners of pet stores to communicate the increasing risks to those customers who wish to purchase reptiles like lizards, snakes, and turtles. With the increase in popularity of reptiles as mainstream pets, there was a subsequent increase in incidence of Salmonellosis cases. Over the ten year span from 1991-2001, the estimated number of households with reptiles doubled from approximately 850,000 to 1.7 million, and this increase is consistent with trends seen today with an estimated 3% of households within the US having at least one reptile (Rabsch et al, 2014). This increase in the number of reptiles per household, coupled with the increased rates of REPAS within the United States has prompted a push for greater awareness of the potential risks associated with having reptiles in close human contact.
A few standard recommendations for disease prevention of REPAS directly from the CDC insist that individuals handle reptiles or reptile cages should immediately and vigorously wash their hands with soap and water. They also advise that those individuals who are at increased risk for infection or other complications of salmonellosis avoid contact with reptiles including children less than 5 years of age as well as immunocompromised individuals. A general precaution to parents is that reptiles should be kept out of households with children that are younger than a year of age because of the increased risk of infection. These represent only a few of the basic precautions that pet owners within the United States and around the world should adhere to in order to help lower the risk of infection by Salmonella and the onset of salmonellosis in men, women, and children alike. This bacteria and its servers are invasive by nature and represent pathogens that are capable of host cell invasion with potentially fatal consequences. Reptiles represent organisms that are capable of being asymptomatic carriers for these disease causing pathogens and as such pet owner should be well aware of the dangers associated with owning these animals and the necessary precautions that should be taken. For further information on recommendations made by the CDC to avoid Reptile-Exotic-Pet-Associated-Salmonellosis visit: CDC Reptiles, Amphibians and Salmonella and CDC- "Reptile-Associated-Salmonellosis"
References
”Receptors Adhere Selectively: Microbes." [1]
McClelland et al. 2001.Complete genome sequence of 'Salmonella enterica serovar Typhimurium LT2. Nature 413: 852-856 [2]
Gay et al. 2014. Salmonella serotypes in reptiles and humans, French Guiana. Veterinary Microbiology doi:10.1016/j.vetmic.2014.01.024. [3]
“Reptile-Associated Salmonellosis”, Centers for Disease Control (CDC), January 2013 [4]
”Reptiles, Amphibians, and Salmonella “ Centers for Disease Control (CDC), November 2013 [5]
Rabsch, W et al. Chapter 7:The Zoonotic Agent Salmonella. Zoonoses -Infections Affecting Humans and Animals: Focus on Public Health. 2014. [6]
W.R.B. Sylvester et al, 2014. Prevalence, serovars and antimicrobial susceptibility of Salmonella spp. from wild and domestic green iguanas (Iguana iguana) in Grenada, West Indies. Zoonoses Public Health. doi: 10.1111/zph.12093: 436-41. [7]
T. Whitten et al, 2014. Reptile-Associated Salmonellosis in Minnesota, 1996-2011. Zoonoses Public Health. doi: 10.1111/zph.12140: 199-208 [8]
A. Fabrega and J. Vila, 2013. Salmonella enterica Serovar Typhimurium Skills To Succeed in the Host: Virulence and Regulation. Clinical Microbiology Reviews. doi: 10.1128/CMR.00066-12. (26): 308-341. [9]
A. Lahiri et al, 2010. Visiting the cell biology of Salmonella infection. Microbes and Infection 12: 809-818. [10]
P. Broz et al, 2012. Innate immune response to Salmonella typhimurium, a model enteric pathogen. Gut Microbes 3:2, 62-70. [11]
Hans-Willi Mittrucker and Stefan H.E. Kaufmann. 2000. Immune response to infection with Salmonella typhimurium in mice. Journal of Leukocyte Biology 67: 457-463. [12] CDC: Disease Information:Salmonellosis [13]
McEwan et al, 2015. PLEKHM1 Regulates Salmonella-Containing Vacuole Biogenesis and Infection. Cell Host and Microbe 17: 58-71. [14]
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2015, Kenyon College.
