Rio Tinto
Introduction
The Rio Tinto (red river in Spanish) is a unique ecosystem due to its natural acidity (mean pH is 2.3) and high concentration of heavy metals (Fe, Cu, Zn). The peculiar abiotic factors have endowed the Tinto with an eclectic collection of microbes encompassing all three domains of life. This article will feature the contribution by bacteria to the geomicrobiological cycle which is instrumental to the generation and maintenance of the extreme conditions of the ecosystem [1]. The river itself measures 100 km in length and traverses three defined zones: originating in the historical mining region of the Iberian Pyritic Belt, running south-southwest through a transitional zone and reaching the Gulf of Cádiz, at Huelva [2].
Chemical environment
Paramount to the Tinto system is its constant acidic pH all along the river owing to the high concentration of solubilized ferric iron [1][2][4][6]. The buffer capacity of Ferric iron can be appreciated using Le Chatelier’s principle. When the river is diluted by rain or tributaries, the equilibrium reaction favors products, whereas when evaporation is taking place the equilibrium shifts to the left:
Fe3+ + H2O ⇌ Fe2+ + ½O2 + 2H+ (1)
Exploration of the Tinto ecosystem has favored the notion that the extreme chemical environment is a consequence of metabolizing microorganisms, and not the result of thousands of years of mining activity in the area as was formerly believed [1][4]. The basin of the river supports a thriving community of chemolithotrophic microorganisms responsible for the iron and sulfur cycles operating in Rio Tinto [1]. Pyrite (FeS2) is an important chemolithotrophic substrate because both of its constituents, sulfide and ferrous iron can be used by sulfer- and iron-oxidizing bacteria as a source of energy [1]. The thiosulfate mechanism by which Fe(II) and S2– are liberated from pyrite is initiated by Fe3+ mediated hydrolysis of Fe–S bonds, and highlights the interaction between substrates of the iron and sulfur cycles [8][9].
Microbial diversity
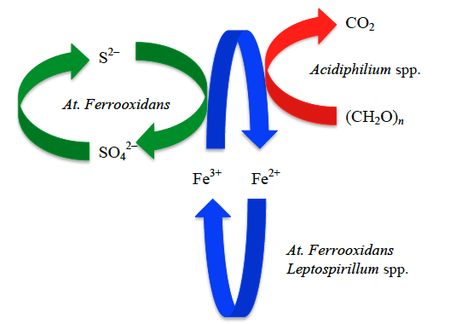
The prokaryotic diversity of the Tinto ecosystem is apparently dictated by the iron cycle. This is supported by the fact that members of three bacterial genera, all conspicuously associated with the iron cycle comprise 80% of the diversity of the water column [1][4]. The Rio Tinto is a model ecosystem for Acidithiobacillus ferrooxidans, Leptospirillum spp., and Acidiphilium spp.
Acidithiobacillus ferrooxidans
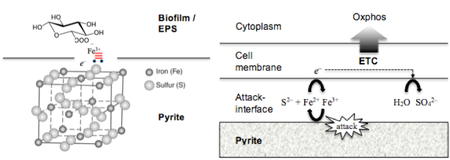
The antiquated view that the initial step in the bioleaching of pyrite, in which the oxidation of disulfide to thiosulfate by ferric iron occurs independent of microbes, has been replaced by a more comprehensive process involving acidophiles [10]. A direct contact mechanisms between ferric iron associated with extracellular polymeric substances (EPS) excreted by At. Ferrooxidans and the insoluble mineral ore has been demonstrated [3][10]. The equation for the direct contact induced thiosulfate mechanism (reaction (2)):
Fe3+ + S2– + 3H2O → SO4 2– + Fe2+ + H+ (2)
Having adapted to the extreme conditions of pH and high concentration of toxic heavy metals of the Tinto system, specialized chemolithotrophic organisms enjoy a luxury not shared by their land-dwelling counterparts [11]. At. ferrooxidans have unlimited access to iron and sulfidic electron donors, and have developed a means for capitalizing on the reserve of these resources in Rio Tinto. Attachment to pyrite is mediated by EPS-complexed iron(III) ions, indicating the importance of biofilms in bacterium-substratum interaction [1][3][10].
Sulfate is one of the products obtained from oxidative attack on insoluble metal sulfides and has been shown to be an important electron acceptor used for dissimilatory reduction in acidic environments [4]. However in the Rio, sulfate-reducing activity is scarce as a consequence of the availability of ferric iron in the system [4]. In addition, a vast supply of iron(III) ions are needed for the initial attack step in the biooxidation of pyrite, further necessitating the requirement for a reliable supply of ferric iron [1].
Leptospirillum spp.
Species within the genus Leptospirillum spp. are obligate chemolithotrophs, and many play a crucial role in the recycling of iron(III) ions in acidic environments [1][4][7]. The maintenance of the iron cycle in the Rio is of critical importance to the success of the ecosystem, and is reliant on ferric iron in solution. Consequently, hydrolysis of ferric iron (reaction (1)) lowers the pH to promote its water solubility, and in turn, microbes respiring with iron(II) as an electron donor produce Fe3+ by extracellular electron transfer, thereby competing the cycle. In certain parts of the river the water column is deep enough that sections become completely anoxic, and provides a niche in which At. ferrooxidans oxidize iron anaerobically using nitrate as an electron acceptor (reaction (3)) [1]. Whereas the dominant oxidizers of ferrous iron throughout the oxic regions, which constitute the majority of the river, have been shown to be members of Leptospirillum spp. (reaction (4)) [7][9].
Fe2+ + NO3 → Fe3+ +H2O (3) anoxic
Fe2+ + O2 + 3H2O → Fe3+ + H2O (4) oxic
Certain biochemical properties of Leptospirillum spp. make them well adapted for survival in Rio Tinto and signify their importance to the iron cycle. Leptospirillum spp. have a lower pH optimum than At. ferrooxidans, and can tolerate higher concentrations of heavy metals [9]. Norris et al. have shown a KM for Fe2+ of Leptospirillum spp. well below that of At. ferrooxidans, cementing the belief that the two groups employ different ferrous-iron-oxidizing systems and suggests that the mechanism used by Leptospirillum spp. is better suited for the Tinto ecosystem as long as oxygen is present [6].
Acidiphilium spp.
While At. ferrooxidans have been touted at the interface between the iron and sulfur cycles, Acidiphilium spp. members form the link between the carbon and iron cycles operating in the Rio [4][7]. Consistent with the theme of specialized prokaryotic metabolic processes servicing the Tinto community, Acidiphilium spp. have the unique capacity to degrade organic molecules by using ferric iron as an electron acceptor in both aerobic and anaerobic microenvironments [2][4]. Under the acidic conditions in the Rio, dissolved Fe(III) is available in solution, and the redox potential for the Fe3+/F2+ half-reaction (+0.77 V) is comparable to the redox potential for the reduction of O2/H2O (+0.82 V) [5]. This offers a logical explanation for the unusual ability of Acidiphilium species to respire a variety a substrates coupled to the oxidation of Fe3+ both in the presence and absence of oxygen, and is likely of ecological significance in the cycling of iron and organic matter at oxic-anoxic boundaries in sediment [5].
The big picture
The discovery and elucidation of extremophiles such as those thriving in Rio Tinto has fashioned the notion that life is robust and provided the framework for the budding field of astrobiology. As an important terrestrial analogue for Mars, exploration of the Rio has typified some extant microbial communities that could plausibly sustain life on another planet. Anaerobic iron oxidizers (At. ferrooxidans) and iron respiration (Acidiphilium spp.) are conceivable microbial metabolisms that could support growth on Mars, and ferric iron in solution has been shown to absorb UV radiation thus protecting organisms growing in waters from radiation and oxidative stress [1]. That said, many more questions than answers remain, lending room for further study; albeit with an optimistic outlook.
References
(1) Amils, R., González-Toril, E., Fernández-Remolar, D., Gómez, F., Aguilera, A.,Rodríguez, N., Malki, M., García-Moyano, A., Fairén, A. G., de la Fuente, V., and Sanz, J. L. 2007. “Extreme Environments as Mars Terrestrial Analogs: The Rio Tinto Case”. Planetary and Space Science. 55:. DOI: 10.1016/j.pss.2006.02.006.
(2) Amils, R., González-Toril, E., Fernández-Remolar, D., Gomez, F., Rodríguez, N., and Duran, C. 2002. “Interaction of the Sulfur and Iron Cycles in the Tinto River Ecosystem”. Reviews in Environmental Science and Biotechnology. 1:. DOI: 10.1023/A:1023232002312.
(3) Gehrke, T., Telegdi, J., Thierry, D., and Sand, W. 1998. “Importance of Extracellular Polymeric Substances from Thiobacillus ferrooxidans for Bioleaching”. Applied and Environmental Microbiology. 64.
(4) González-Toril, E., Llobet-Brossa, E., Casamayor, E. O., Amann, R., and Amils, R. 2003. “Microbial Ecology of an Extreme Acidic Environment, the Tinto River”. Applied and Environmental Microbiology. 69:. DOI: 10.1128/AEM.69.8.4853-4865.2003.
(5) Küsel, K., Dorsch, T., Acker, G., and Stackebrandt, E. 1999. “Microbial Reduction of Fe(III) in Acidic Sediments: Isolation of Acidiphilium cryptum JF-5 Capable of Coupling the Reduction of Fe(III) to the Oxidation of Glucose”. Applied and Environmental Microbiology. 65.
(6) Norris, P. R., Barr, D. W., and Hinson, D. 1988. “Iron and Mineral Oxidation by Acidophilic Bacteria: Affinities for Iron and Attachment to Pyrite”. In Norris, P. R. and Kelly, D. P. (ed.), Biohydrometallurgy. Science and Technology Letters.
(7) Sánchez-Andrea, I., Rodríguez, N., Amils, R., and Sanz, J. L. 2011. “Microbial Diversity in Anaerobic Sediments at Río Tinto, a Naturally Acidic Environment with a High Heavy Metal Content”. Applied and Environmental Microbiology. 77:. DOI: 10.1128/AEM.00654-11.
(8) Sand, W., Gehrke, T., Jozsa, P. G., and Schippers, A. 2001. “(Bio)chemistry of Bacterial Leaching - Direct vs. Indirect Bioleaching”. Hydrometallurgy. 59:. DOI: 10.1016/S0304- 386X(00)00180-8.
(9) Sand, W., Rohde, K., Sobotke, B., and Zenneck, C. 1992. “Evaluation of Leptospirillum ferrooxidans for Leaching”. Applied and Environmental Microbiology. 58.
(10) Schippers, A. and Sand, W. 1999. “Bacterial Leaching of Metal Sulfides Proceeds by Two Indirect Mechanisms via Thiosulfate or via Polysulfides and Sulfur”. Applied and Environmental Microbiology. 65.
(11) White, D. 2007. “The Physiology and Biochemistry of Prokaryotes”. Oxford University Press, New York.