South Pacific Gyre
Overview
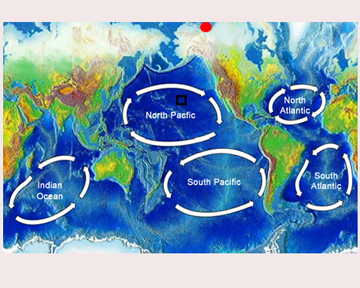
By Kjetil Odden
The South Pacific Gyre (SPG) is one of five oceanic gyres in the global oceans. It is not only the largest defined oceanic province on Earth [1], but also the most remote, sitting further from any continent than any other oceanic region [2]. The SPG covers approximately ten percent of our planet’s ocean surface [3], and is distinguished by some of the clearest water on Earth [4][5][6] as well as several unique and extreme oceanographic properties, which are common to such oligotrophic areas of water [1]. These properties include extremely low nutrient content [2] – which leads to low productivity [1][2][4][5] – as well as extremely low chlorophyll concentrations [2][4][5]. This combination of facts has made it common to refer to the SPG, as well as the other oligotrophic gyres, as a “biological desert” [1][2][6][3].
Despite this, there is ample life in both the sea and the sediment under the gyre that continues to be researched. The sedimentary microbial communities are of particular interest, as they play a crucial role in global biochemical cycles. [7]
[3][8] Due to the immense size and remoteness of the SPG, these microbes – while extremely important – are still severely understudied. [3] There have been many studies undertaken to improve our understanding of these communities, but their composition and makeup remains largely unknown. [7]
[6] Here we will attempt to concisely and efficiently cover what is known about microbial life in the SPG, how and why it is so important, and where more work will continue to be necessary in the future.
About the Environment
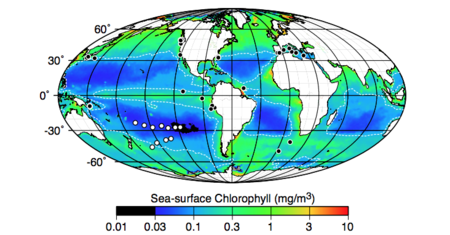
The exact geographic confines of the SPG are not always clearly or consistently defined. The most commonly-cited boundaries are the region from 20º S to 45º S, with the swell in the East Pacific and the start of Cretaceous oceanic crust in the west marking the east-west margins. [6] This places the center of the gyre farther from any continent or highly-productive ocean region than any other location on Earth. [1] The low-chlorophyll middle section of the gyre, which comprises the vast majority of its surface area, is just over double the size of North America. [1] The SPG is surrounded by regions of subduction zone volcanism, which often leads to emission of explosive ash. [9] These regions encircle the Pacific plate and are bounded by the Andes to the east and New Zealand – or more accurately, the eastern margin of the Australian plate – to the west. [9] The area is a continuous slab of basaltic oceanic crust which has a thin sedimentary covering ranging from 100m down to as low as one meter. [5]
The isolation of the SPG from the continents means that the center of the gyre is not impacted by an influx of nutrients or pollution from those land masses, as most regions of the oceans are. [6] This is likely an important contributing factor in the extremely low rates of respiration throughout the gyre. [1][2][4] The low respiration rates have several outcomes – namely, interstitial waters throughout the sediment are oxic, and the organic flux from the waters to the sea floor is extremely low. [1] The sub-seafloor community is even thought to be the low-level end member in sub-seafloor biomass and respiration distributions globally. [1][4] While this sounds extreme, it should also be pointed out that the other major ocean gyres resemble the SPG quite closely in many ways. [9][5] This means that while the SPG may in fact be the end member, as much as half of the world’s oceans likely approach those end member values, meaning the distribution is quite uneven and could be misleading. [1] It also should be mentioned that some border regions of the SPG – i.e., away from the vast, low-chlorophyll center – are quite different in a few key ways. Most significantly, the southern edge of the SPG holds a much higher sea-surface chlorophyll content. [5] This produces higher rates of productivity and respiration, as well as interstitial sub-seafloor waters that are mostly anoxic. [5]
Since most research on microbes in the gyre focuses on the sedimentary members, and since that is what we are most interested in here, a few more specific points should be made about the sediment column. The sediment in the SPG has some of the lowest organic burial rates in the ocean, as well as what researchers have called some of the lowest cell concentrations ever encountered in marine sediment. [5][10] The sediment itself accumulates extremely slowly – about 0.1 to 1.0 meters per million years. [1] These are among the lowest rates to occur anywhere on Earth’s surface. [1] The sedimentation rate is lowest at greater depths and slightly higher on the eastern side of the SPG, where the water depth is shallower. [1] It also slightly higher in the western and southern regions where chlorophyll concentrations are not as depleted as the central section. [1][5] Of course, the rates there are still very low compared to what is observed in most marine sediment columns. Within the sediment, however, there is life, and important life at that. The living community is one of low biomass and low metabolic activity. [1][2][5][11]
Due to its oligotrophic nature and the factors already discussed, oxygen not consumed in the top few centimeters of the sediment will diffuse down to much greater depths. [4] In shallow sediment, oxygen is the principal net terminal electron acceptor. [5] On a larger scale, nitrogen fixation, as mentioned previously, is the major source of nutrients for the microorganisms. [2] Researchers have even estimated that the SPG accounts for over ten percent of global pelagic nitrogen fixation, which translates to about five or six percent of all new nitrogen input to the world’s oceans. [2]
Known Microbial Ecology
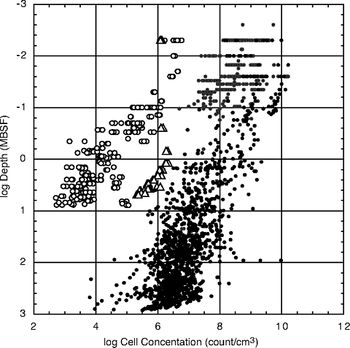
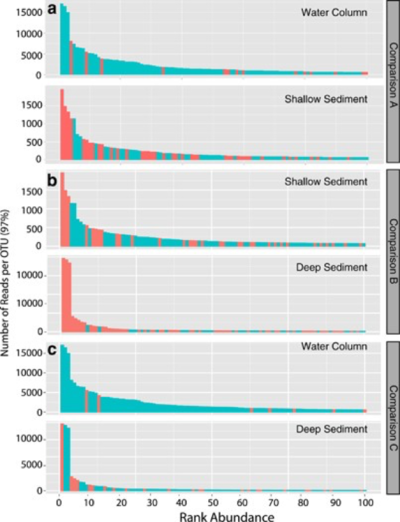
As has already been discussed, the microbial ecology of the SPG is shaped by its unique and extreme conditions that cause low productivity low cell counts. In many ways, the overall microbial community is quite similar to what has been observed in other oceanic gyres. [11][3] These oligotrophic oceans are often inhabited primarily by small phytoplankton and heterotrophic bacteria. [8] These are usually very small (under 3 micrometers) in length and have adapted to survive in limited-nutrient conditions. [8] This is largely the case in the SPG as well; however, this will be less of a focus beyond this point as we will discuss sub-seafloor life. At the same, there is only very limited research on specific communities within the sub-seafloor, so we will continue to lean into the better-researched aspect of microbial life in the waters of the gyre, as well. This is especially important as recent studies have shown that the abundance of bacterial taxa in the seafloor, and the sedimentary communities in general, are colonized via the overlying water column. [11]
Most primary production in the SPG is sustained by active regeneration processes, which fuel up to 95% of the biological nitrogen demand. [12] The sediment column in the gyre’s seafloor is highly stratified, which is a necessary precursor to many of the subsequent processes and characteristics discussed. [8] Despite the extremely low organic burial rate, microbes have been found to occur throughout the sediment column. [1][2] This has primarily been based off sediment core samples, and indicates that biomass has persisted in the sediment for at least 70 million years. [1] At a range of different depths, cell abundances have been found to be three to four orders of magnitude lower than at the same depths in nearly all other researched sub-seafloor communities. [1] Additionally, oxygen fluxes prove presence of an active microbial community in the sediment as well. [1] It is inferred from the consistent and evenly-distributed oxygen fluxes that the porewater is oxic throughout the sediment column, and that the sedimentary community is predominantly aerobic. [1][2][5] This also implies that the flux of oxygen may sustain aerobic life in the underlying basalt, which is up to over 100 million years old. [1]
The respiration rates within the sedimentary community are one to three orders of magnitude lower than previously researched sites. [1] As a result, this community is mostly aerobic which is yet another feature unique from other marine sediment communities. [1][5] The recorded presence of dissolved oxygen, nitrate, phosphate, and inorganic carbon throughout the sediment column across the gyre also indicates that microbial life is not limited by the availability of electron acceptors. [5] It also has led researchers to conclude that life is not limited by the major nutrients of C, Ni, or P in the sedimentary environment. [5] Regarding the electron donors, about half used by the sub-seafloor community are provided by radiolysis of water; the other half is provided by buried organic matter. [1][5]
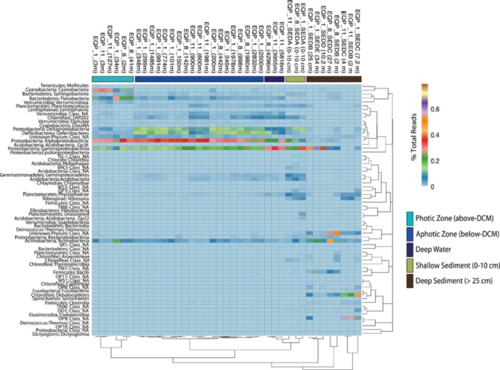
Finally, we will touch on some specific methods and specific compositions and distributions of communities. Phylogenetic analysis has been done to determine dominant species – Proteobacteria and cyanobacteria were found to be predominant. [6] Betaproteobacteria were detected, but only in the center of the gyre. The same researchers also performed cultivation of heterotrophic microbes, finding that nearly 20% of phylogenetically distinct bacteria isolates at least at the species level. [6] These results suggest that distribution and diversity of microbes in the SPG are indeed closely related to its oligotrophic oceanographic features. [6]
From synthesis of multiple studies, it has been estimated that the SPG (from sub-seafloor to surface) is dominated by 20 or so major bacterial clades. [3] In the central part of the gyre, the dominant organism is Procholorococcus, which shows low abundance in the upper waters and is more frequent in the irradiance zone. [3] The surface waters, while of less interest to us, are very important; they are dominated by SAR11, SAR86, and SAR116. [3] There were distinct distribution patterns observed for these clades as well as for Actinobacteria. [3][8]
In some of the first successful attempts at resolving the identities of microbes in the sub-seafloor, it was confirmed that much of the sediment seems to be analogous to that of the deep waters above it. [11] In particular, Alphaproteobacteria, Gammaproteobacteria, and Deltaproteobacteria were all found to dominate the upper layers of the sub-seafloor sediment. [11] Farther down, the deep sediment layers were found to be comprised primarily of Chloroflexi, Actinobacteria, and members of another phylum of yet unknown lineage. [11]
Key Microbial Players
- Alphaproteobacteria
- A class of bacteria within the phylum Proteobacteria
- Gram-negative – as with all proteobacterias
- In the Pacific gyres, most of these are found in the sub-seafloor, but not very deep into the sediment. [11][3]
- They show a distinct distribution throughout the SPG throughout the deep water column as well, which extends into the sediment. [11][3]
- SAR11
- This is an order within the class of alphaproteobacteria
- They are possibly the most abundant bacterium in the world [8]
- SAR86 and SAR 11, together
- Both orders here are prevalent in the waters rather than the sediment. [3][8]
- Together they make up nearly 70% of gene sequences in the SPG. [8]
- Their potential activity was found to increase with depth, which is the opposite of Prochlorococcus. [11]
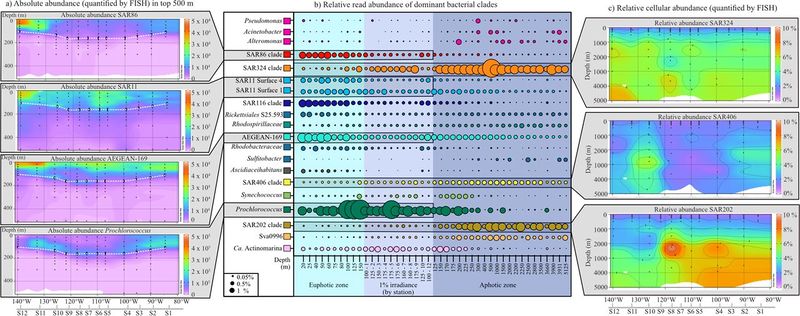 Comprehensive look at bacterial compositions throughout the SPG, including absolute abundances of SAR11, SAR86, Prochlorococcus, and one order of Alphaproteobacteria (a)... also gives relative abundance and depth distribution of the 20 dominant bacterial clades mentioned in the previous section(b). [6]
Comprehensive look at bacterial compositions throughout the SPG, including absolute abundances of SAR11, SAR86, Prochlorococcus, and one order of Alphaproteobacteria (a)... also gives relative abundance and depth distribution of the 20 dominant bacterial clades mentioned in the previous section(b). [6]
- SAR11
- Actinobacteria
- Prochlorococcus
- A genus of small marine cyanobacteria
- Belong to photosynthetic picoplankton
- Major ocean producers – probably the most abundant photosynthetic organism in the world. [14]
- Unusual pigment due to chlorophyll [14]
- These microbes are not found in the sedimentary layers of the sub-seafloor, but rather in the waters throughout the SPG. [6][11] In particular, they are dominant in the central area of the gyre. [6][11][3]
- They serve a massively important role throughout the SPG as the dominant photosynthetic organism. [3][8]
Plastic Pollution
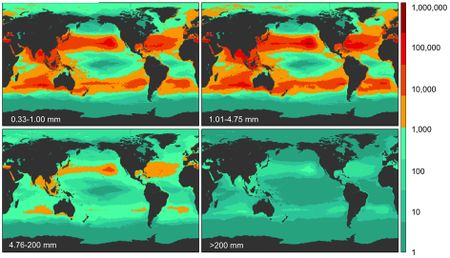
The existence of massive collections of garbage and plastic in ocean gyres has been known for years. In particular, the Pacific garbage patch in the North Pacific Gyre has been well-documented and well-studied. [15][16] In the last several years, there began to more research done on the nature of these patches, and it was assumed that one would exist in the South Pacific Gyre as well. [17][16] In 2017, the assumptions were confirmed. A research trip through the gyre recorded extensive evidence for a massive center of plastic pollution in the SPG. [17] The patch cannot be picked up by photographs or satellites, as the plastic quickly breaks down into tiny pieces, as a result of extreme exposure to the Sun. [17][16] This is generally true for all subtropical gyres with plastic problems – they do not have massive piles of plastic in the way we might imagine – but it is even more pronounced in the SPG, likely due to the nature of plastic that is ending up in the waters from the developing nations around it. [17][16] While this may sound like a good thing, it is not. The plastic is not degrading, or becoming organic matter, but rather simply becoming even easier for animals to swallow. [17] Put simply, it is an absolutely massive problem, but one the scope of which cannot be addressed here. As far as how microbes relate, there is a connection to this massive pollution center. When the particles of plastic become small enough, they can actually be ingested and metabolized by microorganisms, primarily through processes to form carbon dioxide. [17][16] It is unclear exactly how this patch has and/or will continue to affect microbial communities in the SPG, but it is clear that any ill effects could be devastating to the larger-scale geochemical processes that depend on them. [17] More work is clearly needed on this relatively new discovery as we seek to understand what it does and what we can do to manage it.
Conclusion
The massive, remote, and ultra-oligotrophic South Pacific Gyre can be characterized by extreme oceanographic conditions; low chlorophyll, respiration, and productivity; and even a newly-found plastic pollution collection. Despite it all, the SPG is home to active microbial communities of bacteria that are crucial to the workings of global geochemical cycles. With so little known and so much to learn, it is clear that the SPG will be a hub for significant and exciting research for decades to come.
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 1.22 1.23 D’Hondt et al.: Subseafloor sedimentary life in the South Pacific Gyre. PNAS 2009.
- ↑ 2.00 2.01 2.02 2.03 2.04 2.05 2.06 2.07 2.08 2.09 2.10 Halm et al.: Heterotrophic organisms dominate nitrogen fixation in the South Pacific Gyre. Nature 2011.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 Reintjes et al.: On-Site Analysis of Bacterial Communities of the Ultraoligotrophic South Pacific Gyre. ASM 2019.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 Ferdelman et al.: Oxygen penetration deep into the sediment of the South Pacific Gyre. Biogeosciences discussions 2009.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 D’Hondt et al.: IODP Expedition 329: Life and Habitability Beneath the Seafloor of the South Pacific Gyre. Scientific Drilling 2013.
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 6.8 6.9 Yin et al.: Spatial Variations in Microbial Community Composition in Surface Seawater from the Ultra-Oligotrophic Center to Rim of the South Pacific Gyre. PLOS ONE 2013.
- ↑ 7.0 7.1 Hondt et al.: Metabolic Activity of Subsurface Life in Deep-Sea Sediments. Science 2002 295:2067.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 8.8 8.9 West et al.: Distinct Spatial Patterns of SAR11, SAR86, and Actinobacteria Diversity along a Transect in the Ultra-oligotrophic South Pacific Ocean. Frontiers in Microbiology 2016.
- ↑ 9.0 9.1 9.2 Dunlea et al.: Dust, volcanic ash, and the evolution of the South Pacific Gyre through the Cenezoic. AGU 2015.
- ↑ Kallmeyer et al.: Global distribution of microbial abundance and biomass in subseafloor sediment. PNAS 2012.
- ↑ 11.00 11.01 11.02 11.03 11.04 11.05 11.06 11.07 11.08 11.09 11.10 11.11 Walsh et al.: Bacterial diversity and community composition from seasurface to subseafloor. Nature 2016.
- ↑ Raimbault and Garcia: Evidence for efficient regenerated production and dinitrogen fixation in nitrogen-deficient waters of the South Pacific Ocean: impact on new and export production estimates. LMGEM 2008.
- ↑ Servin et al.: Evidence Excluding the Root of the Tree of Life from the Actinobacteria. MBE 2007.
- ↑ 14.0 14.1 Life at the Edge of Sight. Harvard University Press 2017.
- ↑ Kaiser: The Dirt on Ocean Garbage Patches. Science 2010.
- ↑ 16.0 16.1 16.2 16.3 16.4 De Wolff, Kim: Gyre Plastic: Science, Circulation and the Matter of the Great Pacific Garbage Patch. UC San Diego 2014.
- ↑ 17.0 17.1 17.2 17.3 17.4 17.5 17.6 Eriksen et al.: Plastic pollution in the South Pacific subtropical gyre. MPB 2013.
