Talimogene Laherparepvec: A groundbreaking viral therapy for late stage melanoma: Difference between revisions
No edit summary |
|||
| Line 55: | Line 55: | ||
3. Liu, B. L., Robinson, M., Han, Z., Branston, R. H., English, C., Reay, P., & ... Coffin, R. S. (2003). ICP34.5 deleted herpes simplex virus with enhanced oncolytic, immune stimulating, and anti-tumour properties. <i> Gene Therapy, 10(4), </i> 292. <https://www.nature.com/articles/3301885>. | 3. Liu, B. L., Robinson, M., Han, Z., Branston, R. H., English, C., Reay, P., & ... Coffin, R. S. (2003). ICP34.5 deleted herpes simplex virus with enhanced oncolytic, immune stimulating, and anti-tumour properties. <i> Gene Therapy, 10(4), </i> 292. <https://www.nature.com/articles/3301885>. | ||
9. Johnson, D. B., Puzanov, I., & Kelley, M. C. (2015). Talimogene laherparepvec (T-VEC) for the treatment of advanced melanoma. <i> Immunotherapy </i>, 7(6), 611-619. | |||
<https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4519012/pdf/nihms709872.pdf> | |||
<br><br>Authored for BIOL 238 Microbiology, taught by [mailto:slonczewski@kenyon.edu Joan Slonczewski], 2018, [http://www.kenyon.edu/index.xml Kenyon College]. | <br><br>Authored for BIOL 238 Microbiology, taught by [mailto:slonczewski@kenyon.edu Joan Slonczewski], 2018, [http://www.kenyon.edu/index.xml Kenyon College]. | ||
Revision as of 17:53, 23 April 2018
Classification
Baltimore classification: dsDNA
Family: Herpesviridae
Virion: Enveloped
Capsid symmetry: Icosahedral
Replication site: Nucleus and cytoplasm
Overview of Talimogene Laherparepvev (T-VEC)
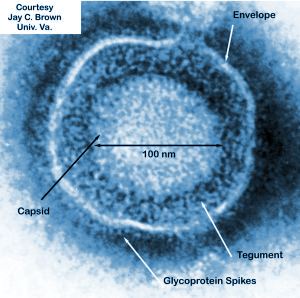
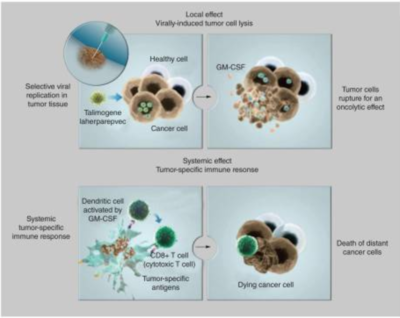
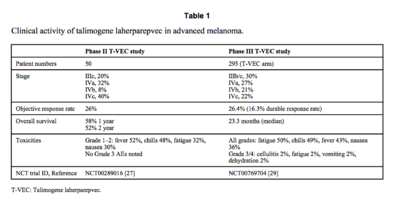
By Alex Fazioli
Double brackets: [[
Filename: PHIL_1181_lores.jpg
Thumbnail status: |thumb|
Pixel size: |300px|
Placement on page: |right|
Legend/credit: Electron micrograph of the Ebola Zaire virus. This was the first photo ever taken of the virus, on 10/13/1976. By Dr. F.A. Murphy, now at U.C. Davis, then at the CDC.
Closed double brackets: ]]
Other examples:
Bold
Italic
Subscript: H2O
Superscript: Fe3+
Section 2
Include some current research, with at least one figure showing data.
Section 3
Include some current research, with at least one figure showing data.
Section 4
Conclusion
References
1. Andtbacka, R. H., Kaufman, H. L., Collichio, F., Amatruda, T., Senzer, N., Chesney, J., ... & Milhem, M. (2015). Talimogene laherparepvec improves durable response rate in patients with advanced melanoma. Journal of clinical oncology, 33(25), 2780-2788. <https://s3.amazonaws.com/academia.edu.documents/41260637/Talimogene_Laherparepvec_Improves_Durabl20160115-22239-yubl2z.pdf?AWSAccessKeyId=AKIAIWOWYYGZ2Y53UL3A&Expires=1522701871&Signature=rttGQ7Dqvp0CPvJGeXHU5jkWlU4%3D&response-content-disposition=inline%3B%20filename%3DTalimogene_Laherparepvec_Improves_Durabl.pdf>.
2. Pol, J., Kroemer, G., & Galluzzi, L. (2016). First oncolytic virus approved for melanoma immunotherapy. Oncoimmuneology, 5(1), e1115641 (3 pages). <https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4760283/>.
3. Liu, B. L., Robinson, M., Han, Z., Branston, R. H., English, C., Reay, P., & ... Coffin, R. S. (2003). ICP34.5 deleted herpes simplex virus with enhanced oncolytic, immune stimulating, and anti-tumour properties. Gene Therapy, 10(4), 292. <https://www.nature.com/articles/3301885>.
9. Johnson, D. B., Puzanov, I., & Kelley, M. C. (2015). Talimogene laherparepvec (T-VEC) for the treatment of advanced melanoma. Immunotherapy , 7(6), 611-619. <https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4519012/pdf/nihms709872.pdf>
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.
