The Effects of Global Climate Change on Soil Respiration
INTRODUCTION
A critical topic of climate change research examines how global warming, from elevated anthropogenic CO2 emissions, affects biological mechanisms in soil microbes that regulate carbon cycling. Soil respiration is a biological process in microbes that convert organic carbon to atmospheric CO2 and is considered to be one of the largest global carbon fluxes on Earth [1]. Due to the temperature sensitivity in soil respiration, it is not surprising to see massive changes in Earth’s global carbon balance from global warming [2]. Changes in environmental carbon fluxes holds strong relevance in human society, including changes in agricultural land use, atmospheric ozone concentration and preservation of natural resources [2].
EFFECTS OF GLOBAL WARMING IN SOIL
Temperature
Over the past century, global atmospheric temperatures have increased by approximately 0.5 degrees Celsius [3]; a phenomenon many scientists believe to be caused by elevated greenhouse gas emissions [4]. Increased soil temperature as a result of global warming has been shown to promote temperature dependent carbon cycling pathways in soil microbes, including decomposition and nutrient mineralization [5] [6]. In the case of decomposition, organic carbon substrates are often complex and require a high temperature activation energy, thus an increase in soil temperature will provide sufficient activation energy for rapid substrate degradation via enzymatic reactions by soil microbes [6]. Consequently, global warming will promote the release of carbon dioxide from soil microbes, resulting in a positive feedback cycle, amplifying the effects of global warming [7]. This theory was tested in a recent experiment that measured the response of soil microbes under an imposed soil warming environment. The results showed a 45% mean increase in carbon dioxide emission from promoted soil respiration activity [8].
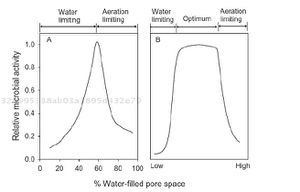
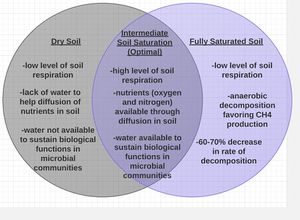
Soil Moisture
Soil moisture is another important factor that regulates CO2 efflux from soil respiration [9]. Various models from previous research have shown optimal levels of heterotrophic respiration in wet but unsaturated soil [9] [10]. Fully saturated soils result in anaerobic conditions that depress aerobic microbial activity [9] [10]. Anaerobic conditions inhibit carbon dioxide production in soil in two ways. Firstly, the main product of decomposition during aerobic decomposition is carbon dioxide, whereas methane is the main product of anaerobic decomposition [10]. Secondly, the rate of anaerobic decomposition is 30-40% compared to the rate of aerobic decomposition [10]. Therefore, not only is CO2 generation depressed by anaerobic conditions, but also the rate of decomposition is reduced [10]. Similarly, dry soil conditions limit microbial activity and prevent nutrient diffusion, both of which are unfavorable for soil respiration [10]. Increased levels of infrared radiation on Earth’s surface from global warming have elevated the rate of evaporation [11], causing the depletion of water in soil and ultimately decreasing soil respiration activity [9] [12].
MECHANISMS
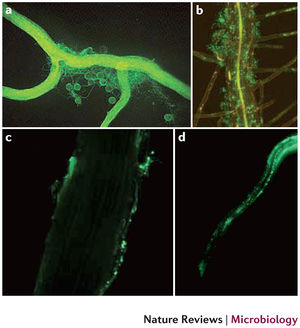
Rhizosphere Respiration
Interactions between plant root systems and microorganisms play an integral role in soil respiration. Rhizosphere respiration is a symbiotic relationship between plant roots and rhizobacteria that stimulate CO2 production in soil [9]. One way plant roots assist rhizobacteria is by providing large fluxes of carbon rich molecules from the release of long carbohydrate chains and enzymes when the root sheds dead material [13]. Soil microbes have adapted to this niche by forming biofilms on root surfaces to optimize organic carbon intake for CO2 production [13]. In return for the supply of carbon, rhizobacteria have the ability to enhance plant growth by mineralizing normally poorly soluble nutrients, such as iron and phosphate via the secretion of siderophores [14]. Upon binding with Fe3+ ion, siderophores form a complex that can be taken up by plant root receptors [15]. Iron is then reduced to its soluble form Fe2+ within the plant root cell [15]. It is predicted that global climate change will increase primary production in plants, resulting in an increase of carbon release by roots into the soil, subsequently promoting rhizosphere respiration [13].
Tricarboxylic Acid Cycle
The tricarboxylic acid cycle is a fundamental process shared by plants, fungi, animals and bacteria that oxidizes six carbon sugars into CO2 and H2O. This process requires oxygen as the terminal electron acceptor and accounts for the majority of microbial soil respiration [9]. The TCA cycle is a CO2 generating process within the mitochondria that works in conjunction with glycolysis, the pentose phosphate pathway and electron transport chain, during aerobic respiration [9]. Since the TCA cycle can only operate under aerobic conditions where oxygen is available as the terminal electron acceptor, CO2 output by TCA cycle in soil is inhibited under anaerobic conditions [9].
KEY MICROORGANISMS
Gram Positive Bacteria
Actinobacteria are gram positive bacteria that are commonly found in tropical soils [16]. Among this group are rhizobacteria called Actinomycete Streptomyces which secrete antibiotics and toxins around rhizospheres to prevent growth of other potentially pathogenic rhizobacteria from establishing [17].
Fungi
Arbuscular mycorrhizal fungi are obligate symbionts that rely on land plant root systems to provide them with hexose and in return, the fungi supplies phosphates and nutrients to the plant. [18]
FUTURE RESEARCH DIRECTION
A carbon profile can be obtained by taking soil core samples and analysing CO2, methane and nitric oxide levels throughout the phases of climate change [7]. Ancient soil samples can be used as references to compare carbon cycling pathways before and after the impacts of global warming. However, the carbon cycle is a complex system that interacts with other nutrient cycles, making analysis of results often cumbersome [5].
References
[1] Schlesinger, William H., and Jeffrey A. Andrews. "Soil Respiration and the Global Carbon Cycle" Biogeochemistry 48.1 (2000): 7-20. Print.
[2] Bardgett, Richard D., Chris Freeman, and Nicholas J. Ostle. "Microbial Contributions to Climate Change through Carbon Cycle Feedbacks." The ISME journal 2.8 (2008): 805-14. Print.
[3] Folland, C.K., Karl, T.R., Kinnikov. “Observed Climate Variations and Change” Climate Change: the IPCC Scientific Assessment. 1990. 195-238. Print.
[4] Kirschbaum, Miko U. F. "The Temperature Dependence of Soil Organic Matter Decomposition, and the Effect of Global Warming on Soil Organic C Storage." Soil Biology and Biochemistry 27.6 (1995): 753-60. Print.
[5] Shaver, Gaius R., et al. "Global Change and the Carbon Balance of Arctic Ecosystems." BioScience 42.6 (1992): 433-41. Print.
[6] Davidson, E.A., Janssens, I. A., "Temperature Sensitivity of Soil Carbon Decomposition and Feedbacks to Climate Change." Nature 440.7081 (2006): 165-73. Print.
[7] Johnston, Carol A., et al. "Carbon Cycling in Soil." Frontiers in Ecology and the Environment 2.10 (2004): 522-8. Print.
[8] Hagedorn, Frank, et al. "Short-Term Responses of Ecosystem Carbon Fluxes to Experimental Soil Warming at the Swiss Alpine Treeline." Biogeochemistry 97.1 (2010): 7-19. Print.
[9] Luo, Yiqi, Xuhui Zhou, and Inc ebrary. Soil Respiration and the Environment. Boston: Academic Press, 2006. Print.
[10] Pete Falloon, et al. "Direct Soil Moisture Controls of Future Global Soil Carbon Changes: An Important Source of Uncertainty." Global Biogeochemical Cycles 25.3 (2011) Print.
[11] Manabe, Syukuro, PCD Milly, and Richard Wetherald. "Simulated Long-Term Changes in River Discharge and Soil Moisture due to Global Warming." Hydrological Sciences Journal/Journal des Sciences Hydrologiques 49.4 (2004): 625-. Print.
[12] Curiel Yuste J. Baldocchi, D.D., Gershenson, A., Goldtein, A., Misson, L., Wong, S., “Microbial soil respiration and its dependency on carbon inputs, soil temperature and moisture. “Global Change Biology. 2007. 13, 2018-2035. Print.
[13] Cardon, Zoe G., and Julie Lynn Whitbeck. The Rhizosphere: An Ecological Perspective. Amsterdam: Elsevier Academic Press, 2007. Print.
[14] Van Loon, L. C. “Plant Responses to Plant Growth-Promoting Rhizobacteria.” European Journal of Plant Pathology, 2007. Print.
[15] Harrington, J.M., Crumbliss, A.L., “The redox hypothesis in siderophore-mediated iron uptake” Biometals. 2009. 22: 679-689. Print.
[16] Waldrop, M. P., T. C. Balser, and M. K. Firestone. "Linking Microbial Community Composition to Function in a Tropical Soil" Soil Biology and Biochemistry 32.13 (2000): 1837-46. Print.
[17] Premjit, S. “Characteristics of Soil Ecology” Applied Soil Ecology. 2007. 66-67. Print.
[18] Márquez, A. J., J. Stougaard, and Inc ebrary. Lotus Japonicus Handbook. Dordrecht: Springer Netherlands, 2005. Print.