The sociality of Myxococcus xanthus: Evidence of social group formation and interactive intelligence
By: Damaris Garduno
Introduction:
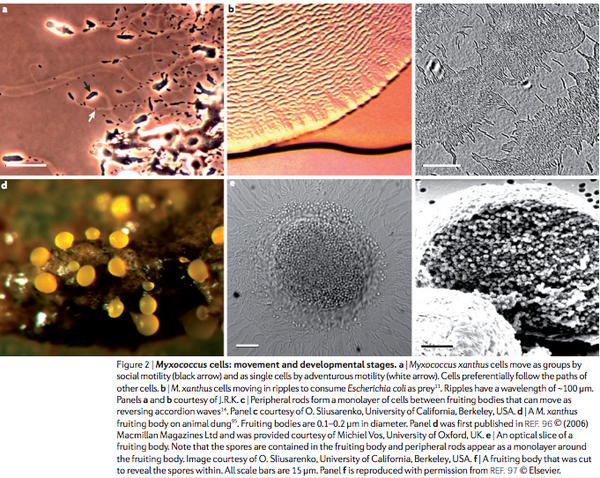
The intelligence and socialization of microbes is not studied in the same way as that of humans. Microbes lack the neuron network for scientists to do so. Like ethnographers, the scientific community has observed and quantified certain microbial social behaviors and lack thereof. Myxococcus xanthus inter-bacterial interaction is especially interesting since they feature spore-producing fruiting bodies, predator customs, and swarming motility (Fig.1a-1f) (Wartel et al., 2013). What type of social or antisocial behaviors do they show under varying levels of stress and amongst different combinations of bacterial community members? Scientists have been debating the true answer to this question for at least 40 years. The following is a compilation of the experimental results that can help us answer this question.
Background Information:
Myxococcus xanthus belongs to the Deltaproteobacteria, Myxococcales order, which are gram-negative rod-shaped bacterium that may inhabit soil and plants alone or be embedded in the feces of herbivores (Fig. 1d) (Wall, 2014). In the early 1960s, scientists became fascinated by the rare development cycle of bacteria like Myxococcus xanthus. It was hard to believe that unicellular microorganisms could be capable of such complex multicellular behaviours (Hagen, 1978; Claessen et al., 2014). Several years and experiments later, the scientific community is now starting to understand and support the view of prokaryotic organisms as social creatures, not only capable but impressively successful at evolving a diverse set of morphological, chemical, and genomic features that have helped them thrive cooperatively in the face of environmental and ecological change (Claessen et al., 2014). Researchers have been delving into identifying and discovering the modalities and purpose of specialized mechanisms developed by microbes, such as predatory rippling, swarming, fruiting body formation, and sporulation (Zusman et al., 2007). Furthermore, these features do not act in isolation, rather, the collective decision-making processes and hierarchies needed to trigger expressions of colonial behavior have been observed to be moderated in tandem by ecological stress, and intraspecific and interspecific signals, such as proteins (Hendrata & Birnir, 2010; Carmen et al., 2013; Wei et al., 2011; Remis et al., 2013, Pathak et al., 2012 a/b, Fiegna & Velicer, 2003; Balagam et al., 2014; Perez et al., 2014; Swapna et.al, 2014; Wartel et al., 2013; and Dziewanoska et al., 2014). We will explore the depictions of social group formation by first outlining the Myxococcus xanthus developmental life cycle in order to then attempt to understand the interactive intelligence employed before, during, and after this bacteria grows into unique multicellular structures.
Social group formation-a diverse developmental life cycle:
There is not just one single way that Myxococcus xanthus grows. Like humans, its developmental biology is shaped concurrently by an extensive genome and ever changing environmental influence.
Swarming

Under favorable food and spatial conditions, it is common to see these microorganisms under a microscope grow by swarming, a directional dispersion of small organized intraspecific clusters away from the colony’s center and towards dispersed nutrients (Fig. 2a) (Hendrata & Birnir, 2010). Cooperatively, myxobacteria scavenge for micromolecules such as detritus or decomposed soil and digest macromolecules or microbes such as Escherichia coli and the legume symbiont, Sinorhizobium meliloti (Rosenberg, 1977; Perez et al., 2014; Mcbride et al.,1996). Although they lack tentacle or tail-like flagella, movement is still achieved through different gliding motility mechanisms performed independently or in cooperative swarms. They move on solid growth substrates at a rate of two to four micrometers per minute, a slow but sufficient speed (Zusman et al., 2007; Balagam, 2014).
Predation patterns
Swarms of predatory Myxococcus xanthus kill and consume an impressively wide range of prey by releasing bioactive toxic secondary metabolites, exo-enzymes, and lytic compounds (Perez, 2014; Vos & Velicer, 2009). The advantage to feeding cooperatively is having a larger genetic representation that is more likely to have the ability to encode the lipases, proteases, nucleases and produce other digestive enzymes or antibiotics imperative to the lysing and digesting of prokaryotic and eukaryotic organisms (Rosenberg, 1977; Perez et al., 2014). Besides their expansive genomic digestive allocation and collaboration, myxobacteria have a distinct way of capturing their prey, rarely modeled by other species. Past studies have termed the hunting patterns as bacterial wolfpack attacks and predatory ripples (Fig. 1b). These strategies are popular areas of research as they may be manipulated by scientists looking to create natural antibiotic models, assisting in prolonged efforts of curing human diseases without too many non-natural compounds (Berleman & Kirby, 2009). Berleman et al. (2006) were the first to explore how rippling motility- a water wave like movement performed when cells organize into moving groups, forming a series of ripples – was initiated during fruiting body formation which is induced by starvation, and predatory activity contrastly induced by nutrition-filled environmental conditions. Before Berleman et al. published their paper, experiments run by Kim & Kaiser (1990), Gronewold & Kaiser (2001), and Stevens & Sogaard-Anderson (2005) endorsed the C-signal model that starvation-induced CsgA proteins would be placed on mycobacterial cell surfaces that in turn were read by neighbor cell receptors, transmitting the ripple movement signal laterally through a mysterious surface protein placement and lucky receptor recognition. The study conducted by Berleman et al. (2006) analyzed strain, growth, macromolecular and substrate conditions that successfully induced rippling and their significant effect on cellular predation and development success. The results elucidated the source of rippling as a response, not specifically to the previously proposed peptidoglycan CsgA, but instead to the presence of any macromolecular substrates or prey. Why does rippling then still occur during fruiting body conditions, an environment perceived to be scarce in macromolecular substrates and prey? Berleman et al. (2006) hypothesize that scavenging lysed cell debris may be responsible for the minimal rippling activity seen at the beginning of fruiting body formation. Their model states that detection of any macromolecule by Myxococcus xanthus stimulates the accordion-like, oscillatory, rippling cellular movement that sweeps through its surroundings, ensuring that no energetically favorable substrate is left behind. This traffic-jam phenomenon of colliding waves of myxobacteria cells reflecting off one another and resulting in repetitive passages over certain areas (Fig. 2a-2c) is tracked mathematically by Hendrata & Birnir (2010). They used equations to generate simulations orienting our visualization of Myxococcus xanthus food availability as the main driver of directionality and motility. Although these studies are convincing and stress the importance of intricacies of biological systems, this year, Perez et al. (2014) further challenged the misconceptions of rippling, pioneering the idea that rippling is not always the key element of predatory strategies. Their evidence presents rippling as an unnecessary tactic for some efficient predation attacks.
Specialized motility: Perez et al. 2014
As soil bacterium, Myxococcus xanthus prey upon substrates and microbial organisms in the soil community. This occurrence could upset prokaryotic and eukaryotic identities that have roles within inter-kingdom interactions essential to sustaining our planet (Perez et al., 2014). Past studies have shed light upon myxobacteria rippling resulting from the presence of food and have hypothesized it to be essential to efficient predation. This study further strengthens the significance of other elements, beyond just the presence of prey, that influence the tactic myxobacteria decide to employ in order to execute the most efficient predation action. In this case, the action elected by a predator is at the mercy of the prey’s make-up. Perez et al. (2014) observed two effective predatory tactics performed by Myxococcus xanthus that when co-cultured with the soil bacterium and nitrogen-fixing legume symbiont, Sinorhizobium meliloti, vary depending on levels of rhizobial exopolysaccharide galactoglucan (EPS II). On the macroscopic level, this is important because EPS II levels are thought to highly influence plant defense response modulation that enhances ecologically advantageous soil rhizobia survival. Based on prey characteristics, the most efficient motility mechanism is employed. The first step in both myxobacteria predatory tactics always involves direct contact with the prey in which their colony is penetrated and cells are personally lysed. Myxococcus xanthus social motility(S) and adventurous motility (A) impaired mutants are either partially or completely unable to prey upon Sinorhizobium meliloti. The S and A solid surface gliding techniques prove to be a greater asset to myxobacteria than the famous predatory rippling mechanism alone. Therefore, Myxococcus xanthus specializes its motility, from rippling to gliding, according to prey identification. This further cements the advanced competence of Myxococcus xanthus to make conscious informed choices about its behaviour during its lifetime and instantly, rather than just across generations, by judging the types of environmental organisms or conditions around it.
Starving conditions lead to fruiting-body formation and sporulation
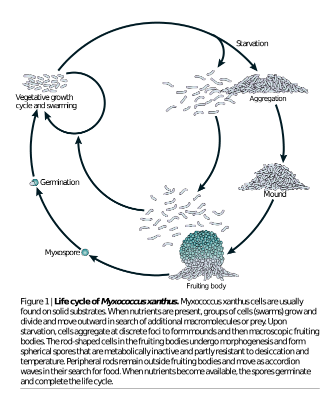
Myxococcus xanthus takes advantage of medium, oxygen, and larger organismal prey when it is available in its surroundings. This biofilm swarming strategy becomes inefficient as soon as food becomes scarce (Hendrata & Birnir, 2010).This behavior is similar to that modeled by nomadic human groups that take advantage of areas across terrains based on food availability and seasonal conditions, coming back together and becoming stationary to cooperatively survive through seasons of nutrient depletion. Myxococcus xanthus will also reprogram their lifestyle in starving conditions. These environmental conditions inhibit growth to which Myxococcus xanthus respond by applying a new developmental process involving even larger intraspecific cooperation than that of swarming, spore-bearing fruiting body formation (Fig. 3) (Claessen 2014). Myxococcus xanthus will aggregate centrally to form a large mound composed of unicellular intraspecific microbe layers in order to restart cooperative feeding (Hendrata & Birnir, 2010; Shimkets, 1999). Rather than sitting as a vegetative multicellular mound expecting prey to pass by, Myxococcus xanthus scavenge by gliding across surfaces. Thousands of Myxococcus xanthus of all abilities, such as non-motile and motile, set aside their differences to be able to communicate and coordinate smoothly enough to successfully capture a meal (Fig. 1b) (Shimkets 1999).
Whitworth (2008) and Jelsback & Sogaard-Anderson (2002) present the fruiting body formation as an aggregation center that progressively builds for 24 hours, containing 10^5 densely packed cells destined to differentiate into new social roles (Fig. 1e and 1f). Fruiting body formation is regulated by signal transduction pathways with a mixture of signal components like proteins such as Ser/Thr kinases. Each member of the fruiting body is synchronized so that genetic expression is moderated skillfully, with only certain genes turned on or off at beneficial points of this new development. For example, with the help of intercellular signaling, tasks are divided among subpopulations of the colony: one of every ten cells is likely to morphologically transform from its natural rod shape to spherical spores with the power to withstand desiccation and various temperature levels. Three out of every ten cells will differentiate into peripheral rods that move around and between fruiting bodies scouting for food (Fig. 1c). The remaining members of the macroscopic fruiting body will have the fate of programmed cell death [altruism] that provides nutrients assisting in the morphogenesis and maturation of the rod shaped cells to new spherical spores, especially during food droughts so that when the nutritional setting is adequate, these spores are ready to germinate and conclude this developmental life-cycle (Whitworth, 2008; Nariya & Inouye, 2008; Wireman & Dworkin 1977). The exact signals responsible for delegating the assignments and selection of subpopulation tasks have yet to be confirmed but the sociality theories that have arisen from accumulating knowledge of Myxococcus xanthus will be explored after reviewing the pros and cons of unicellular organisms developing into multicellular colonies with a harmonized mentality, and therefore harmonized action.
The Microbial Game Theory: Is a temporary microbial social caste system worth it?
You win some
The balance or asymmetry of net gain and loss of all colony members exercising altruistic behavior and microbial cooperation strategies as proficient or defective social microbes could and has been analyzed in terms of the game theory, traditionally used to make gambling or economic decisions for humans (Velicer 2003).
A temporary microbial social caste system may prove itself to be advantageous for Myxococcus xanthus under harsh conditions but can also be detrimental or unfavorable to certain game players. On the bright side, differentiation helps Myxococcus xanthus continue to grow in emaciated states, carry on prey consumption, and still have a fighting chance at battling predators that thrive in the conditions under which Myxococcus xanthus cannot. The range of movement types for the organism to utilize also expands from just (A) adventurous motility to (S) social motility and possibly a combination or alteration of the two. Combining A and S motility types allows for a broader range to conduct swarming, in turn, implementing more meticulous field investigation for food (Shi & Zusman 1993).
A single Myxococcus xanthus cell is more likely to stand strong in the face of environmental disruptions as a member of a colony rather than as a lone unit. Because each member has a vast genome that is not always necessarily the same, there is more likely to be at least one mutation or expression difference that has the potential to make the collective even stronger. Rosenberg et al. (1977) noted the positive repercussions of elevated cell-density on Myxococcus xanthus growth rate and stress resistance. The new spores produced from fruiting body aggregates are much more likely to survive than a unicellular organism. Alone, Myxococcus xanthus glides at a retarded pace, and under starving conditions may find it close to impossible to spot even the leftover crumbs of macromolecules or substrates that have already been lysed.
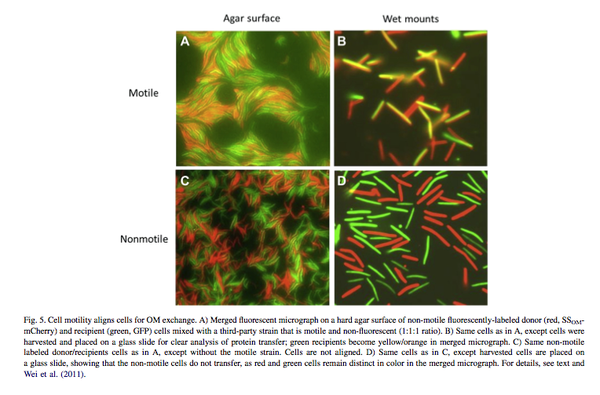
On the other hand, myxospores, which form as a result of differentiation, can disperse freely and quickly. Because of this, Myxococcus xanthus may exploit patches of food that have not yet been discovered (Claessen et.al, 2014). In the long run, these specialized spores will be in better shape than the feeble, malnourished cells that started the fruiting body formation. By dispersing, competition is no longer centralized but is still organized so that kin spores can disperse side-by-side after budding from a fruiting body mass (Gardner, 2006). Realistically, an enormous mass of cells is rarely composed of only kin and hardly ever clone cells. This increases the probabilities of less genetically fit individuals within a mound to benefit from their neighbouring mound cells, which have genetic components essential for survival in environments where they would otherwise die. Less fit cells may get to the point where they may even be lucky enough to convert from a vegetative role to a more active character in their bacterial society. Wei et. al (2011) implement fluorescent labeling and microscopy to track role conversions that occur via protein transfer. Figure 4 demonstrates the experimental results, clearly distinguishing cellular transformations through the presence of hybridized pigmentation of cells when non-motile and motile cells coinhabit an environment (Fig. 4A & 4B). The experimental results also model the lack of role conversions when nonmotile cells exist in isolation (Fig. 4C & 5D).
You lose some
Velicer et al. (2000) aptly named the less fit, less contributing, asocial team members of Myxococcus xanthus in cooperation events as “cheaters”. The reasoning behind this is that they receive developmental benefits from the energy and time intensive work of socially effective group performers without providing anything in return. One of Velicer et al.’s (2000) initial experiments tested the general response of socially effective Myxococcus xanthus populations to the introduction of genetically defective Myxococcus xanthus microbes. Response variables included productivity levels and notable signs of member inclusion or exclusion. Antisocial Myxococcus xanthus mutants were antisocial in the sense they were genotypically incapable of fruiting-body development, had ancestors that did not evolve under social environments for 1,000 generations, and also carried mutations that, past literature, had listed as vital developmental pathway points. Despite such unattractive traits for socially proficient, cooperative event-driven progenitors, these cheating soil bacterium achieve and are allowed to have disproportionate reproductive success (Velicer et al. 2000). Repression of developmentally defective Myxococcus xanthus seems like the most pragmatic choice for the general good of the bacterium population. Not only do asocial Myxococcus xanthus not contribute towards productivity, they negatively impose a load on the backs of the other bacteria: as cheaters increase in frequency, group productivity levels decrease, placing the mean fitness of all socially proficient and non-proficient individuals at risk of fitness loss (Velicer et al., 2000). According to the findings presented by Velicer et al. (2000), if cheaters persist to represent a large percentage of the population, the ability to undergo cooperative interactions and purposes may dramatically decrease and they will all eventually disappear.
Velicer continued his research on Myxococcus xanthus cheater consequences with Fiegna. Together, they focused on quantifying the competitive fates over cycles of development and growth of socially impaired, antisocial Myxococcus xanthus cheaters when mixed with social Myxococcus xanthus. Social Myxococcus xanthus have been termed the ‘proficient cooperator’ wild type. Velicer’s initial claim about social collapse leading to extinction was supported by the results of this research. When antisocials rose to high numbers, productivity was dismal enough to cause substantial population crashes and outright extinction was recorded (Fiegna & Velicer 2003).
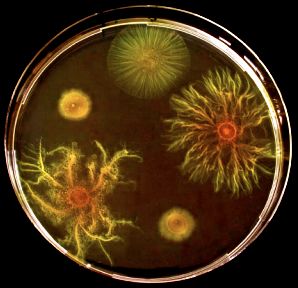
Although the benefits for cooperative behavior adaptation are immediately effective for survival, recent research has unmasked the long term evolutionary disadvantages. Fortunately, Myxococcus xanthus may not be inevitably doomed; there is hope. In 2006, Feigna et al. observed the ability of proficient cooperators to stand up for themselves by sticking together. Proficient cooperators form cliques that flee from the cheater-infected swarms or fruiting bodies to form a utopic, cheater-free colony. This intention limits the reproductive success of bacterial social parasites and, even better, have evolved new genotypes to resist cheater exploitation. Additionally, some socially defective bacterium are able to re-evolve cooperative physiological and/or morphological components. Velicer’s work with Yu (2003) tracked the evolution of cooperative swarming of experimental non-cooperative Myxococcus xanthus lineages through genetic modifications, deleting a gene necessary for the motility needed to grow best on soft agar. S motility(S+) is the most effective motility for swarming on soft agar while A motility(A+) is the best for hard agar. Figure 5 visually demonstrates the remarkable ability of candidate myxobacteria cheaters(evolved lineages E7 & E8) to develop primitive cooperative behaviors on their own, because of social bacteria(ancestral bacteria A1: A+S- & A2: A+S- with A motility genotypes, and wildtype A+ S+ motility genotypes) but not apart from them on soft agar petri dishes.
Methods of interactive intelligence:
How can such small organisms implement such diverse and complex behaviors?
Since 1978, researchers like Hagen et al. started exploring the idea of interdependence of microorganisms through Myxococcus xanthus mutant pairwise mixtures. In isolation, non-fruiting Myxococcus xanthus mutants were unable to sporulate. However, in the presence of appropriate complementary mutants, or the wild-type strains, patterns of functional sporulation and fruiting body formation surprisingly appeared. In order to forego the developmental cycle steps to become a multicellular structure capable of combating stress, bacterial cells somehow send signals to compensate for defective-fruiting community members. This compensation mechanism was termed “the signal hypothesis”. Before and after that, study results of Myxococcus xanthus non-motile mutant pairwise mixtures, also suggested synergistic communication that enabled non-motile mutants to become transiently motile in the presence of the wild-type and select mutant types (Hodgkin & Kaiser, 1977; Velicer & Yu, 2003). These sort of discoveries were the first signs of bacterial linguistic communication and social intelligence. Now that 37 years have passed, the resources available to biologists have made it so these types of experiments are designed to uncover finer and more comprehensive evidence of interspecific and intraspecific cellular interactive competence, and how exactly it may be regulated.
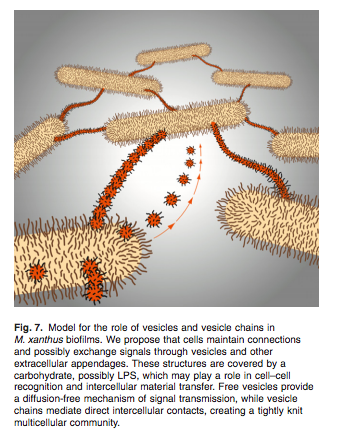
Before cellular devices became the big hit for communication, telephones had wires that traveled all the way from the caller to the person who picked up the phone in order for a message to get across. Bacterial social networks may be formed through a method of communication that mirrors this contact strategy, except, instead of wires, Myxococcus xanthus constructs outer membrane vesicle chains and tubes as extensions making it possible to connect with cells both near and far (Fig. 6) (Remis et.al 2014). A wide range of techniques were used to investigate further about the function and anatomy of these chain and tubular extensions including: Transmission Electron Microscopy (TEM) and cryo-TEM, High Pressure Freezing(HPF) -TEM, Focused Ion Beam (FIB)/Scanning Electron Microscopy(SEM), TEM tomography, time-lapse fluorescence microscopy, quantification of prey cell lysis, vesicle and vesicle chain quantification, in-gel tryptic digestion and protein identification, and Fluorophore-assisted carbohydrate-electrophoresis (FACE) for monosaccharide composition analysis (Remis et al., 2013). Every year a new experimental technique is developed and applied to continue to present new theories attempting to explain how such small organisms can implement such diverse and complex behaviors, supporting or questioning other theories. Naturally, we scientists will never have a permanent answer but every report mentioned above and to come will serve as a foundation for learning more and more about the sociality of Myxococcus xanthus.
References:
28.Velicer, G. J. (2003). Social strife in the microbial world. Trends in Microbiology, 11(7), 330-337.
34.Whitworth, David E. Myxobacteria. Multicellularity and differentiation. ASM Press, 2008.