Thermus: Difference between revisions
No edit summary |
No edit summary |
||
| Line 1: | Line 1: | ||
{{Curated}} | |||
{{Biorealm Genus}} | {{Biorealm Genus}} | ||
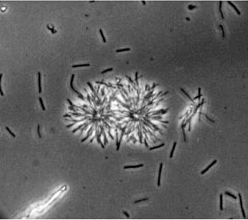
[[Image:rightstrain.jpg|thumb|350px|right|Strains of thermophilic species isolated from the Tanganyika Lake. The left sample is an example of a vesicular strain while the sample at right shows a more "rotund" bodied cell. From the [http://www-archbac.u-psud.fr/projects/tanganydro/_Sampling.html Tangandro Expedition.]]] | |||
==Classification== | ==Classification== | ||
Latest revision as of 20:52, 6 August 2010
A Microbial Biorealm page on the genus Thermus
Classification
Higher order taxa:
Bacteria; Deinococcus-Thermus; Deinococci; Thermales; Thermaceae
Species:
T. antranikianii, T. aquaticus, T. brockianus, T. caldophilus, T. filiformis, T. igniterrae, T. kawarayuensis, T. nonproteolyticus, T. oshimai, T. rehai, T. scotoductus, T. thermophilus,T. thermophilus HB27, T. yunnanensiT. sp., T. sp. manikaranii , T. spp.
Description and Significance
Thermus bacteria cells are a Gram-negative bacteria and are "either immotile or possess flagella and are motile" (R. Huber et al. 2004). Thermus is both more active at higher temperatures and more thermostable than most other enzymes. Go here to learn about Thermus thermophilus in vitamin B(12) synthesis.
Genome Structure
Thermus is assumed to have a pilus-like structure for conjugation, though the molecular basis of the DNA uptake is still unknown. In the three loci of the T. thermophilus genome, twelve genes encoding preplin-like proteins were found. Mutational analysis suggests that one locus (containing four encoding preplin-like protein genes) is essential for natural transformation (A. Friedrich et al. 2002).
Cell Structure and Metabolism
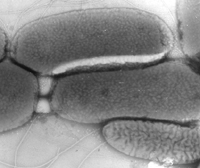
Thermus has an optimum growth temperature of between 70°C and 75 °C (min: 37-45°C, max: 79°C), though some have lower growth temperatures of about 60°C (min: 35-40°C, max: 70°C). High temperature Thermus pH ranges between 7.5 and 8.0, though strains grow as low as 5.1 and as high as 9.5 (R. Huber et al. 2004 ). No strain of Thermus appears to be capable of fermentation.
Thermus strains use several substrates for growth, including carbohydrates, amino acids, carboxylic acids, and peptides (R. Huber et al. 2004). Because of the diversity in habitat from which Thermus strains are isolated, the nutritional diversity also has a large variety. Thermus strains often use monosaccharides as a source for single carbons, though typically pentoses is not metabolized. Thermus strains do not necessarily use the constituent monosaccharide of the disaccharides being used. Most Icelandic strains use sucrose and maltose, though two strains use glucose. The proteins hydrolyzed vary by the isolates as well; while elastin, fibrin, and casein are used by many high-temperature strains of Thermus, nitrate is used as a terminal electrany strains, some are unable to hydrolyze each substrate (R. Huber et al. 2004). For mon acceptor and Themus grows anaerobically in the presence (but not in the absence) of it. Metabolic studies have shown Thermus aquaticus to be a complete tricarboxylic acid cycle, giving a glyoxylate bypass as well with isocitrate lyase and malate synthase (Pask-Hughes).
Ecology

Thermus-like bacteria have been found in both shallow and deep-sea marine hydrothermal systems, as well as in low saline solfataric springs. The Thermus strain T. aquaticus was first isolated in Yellowstone National Park from neutral and alkaline springs and has since then also been isolated from hot springs found in Iceland. Currently, they have been isolated from high temperature (55-100°C) and weak acid to alkaline pH (5-9) habitats, though strains of Thermus were isolated from sites as acidic as pH 3.9 in New Zealand (R. Huber et al. 2004).
References
Alfredsson G. A. ; J. K. Kristjansson; S. Hjorleifsdottir; K. O. Stetter; 1988. "Rhodothermus marinus, gen. nov., sp. nov., a thermophilic, halophilic bacterium from submarine hot springs in Iceland." J. Gen. Microbiol. vol. 134, pp. 299–306.
Lunar and Planetary Institute. 2002. Yellowstone Hot Springs Expedition.
Rehse, P.H.; Kitao, T.; Tahirov, T.H. "Acta Crystallography." (2005). D61, 913-919.
Tangandro Expedition. 1996. "Tangandro." Institut de Génétique et Microbiologie.
