User:S4289005
Thomas Clarkson, Bench D, 23/09/16.
Classification
Higher order taxa
Bacteria – Bacteria – Bacteroidetes – Bacteroidetes – Bacteroidales – Porphyromonas - gingivalis
Species
The species is Porphyromonas gingivalis, and it has a number of different type strains. These are 2561T, ATCC 33277T, BCRC 14417T, CCRC 14417T, CCUG 25893T, CCUG 25928T, CIP 103683T, Coykendall 2561T, DSM 20709T, JCM 12257T, KCTC 5121T, NCTC 11834T, Slots 2561T, Slots' 2561T, Slots' strain 2561T[1].
Description and significance
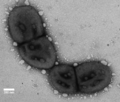
Porphyromonas gingivalis, is a gram-negative rod bacteria. It is non-motile, and found on and within the gingival epithelial cells in the oral cavity. It is dark-pigmented, asaccharolytic, and requires iron from heme for growth[2]. Its relative importance comes from its implication as the most common cause of periodontitis, with a number of articles demonstrating its role as the aetiological agent. Overall, around 85% of diseased tissues have been shown to house P. gingivalis[2]. Additionally it is not often found in healthy tissue, and the depth of the surface pits it forms from infection is strongly correlated to numbers of P. gingivalis present at the site[2]. The bacteria cannot be found in the environment, due to its specific metabolic requirements, It can however be cultured on a blood agar plate[2].
Genome structure
The P. gingivalis strain, 2561T has a total of 41 sequences in its genome[1]. The longest among these is 9878 base pairs long and codes for a DnaK operon, the total genome is 2 354 886 base pairs long[1]. Of these genes, 6 of them are related to 16RNA in some form. A number of the others DNA sequences are related to the virulence factors required for pathology, particularly fimbriae for which 7 of the genes code for[1]. A literature review found no evidence of plasmids of any sort within the genome of P. gingivalis. Furthermore, it was confirmed that they have no cryptic plasmids, however researchers have succeeded in introducing plasmids to their genome[3]. Other researchers sequenced the entire genome of strain TDC60, which is a particularly virulent strain, and found that it had a single circular genome 2 339 898 bp long, indicating no plasmids were present[4].
Cell structure and metabolism
P. gingivalis is a gram negative, rod-shaped bacteria. As a result it has a thinner cell wall with an outer membrane largely composed of LPS. It is also an obligate anaerobe. On account of this, as well as its niche environment on the gingiva, it has a number of adapted metabolic pathways to enable growth. One of the main features is that it requires iron from heme for growth, which it can sequester from its host using gingipains[5]. Additionally, P. gingivalis cannot grow without vitamin K. the bacteria is asaccharoltyic, meaning that it can’t break down sugars. This is not necessary because the environments it lives in are often lacking in sugars. Such as within the pits it forms, at the bottom of which minimal sugars are present, therefore it produces energy by breaking down amino acids[2].
Ecology
P. gingivalis makes up a major component of the human oral microbiome, and has successfully colonised the epithelial cells of the gingiva[6]. It is a compulsively anaerobic species, so cannot survive in the presence of oxygen. It can often be found in what is termed a ‘red complex’ in conjunction with Treponema denticola, and Tannerella forsythia. This complex is strongly associated with periodontal disease[7]. Despite its well-researched connection with pathology, P. gingivalis is found in numbers of patients not displaying clinical pathology[6]. In general, bacteria colonising the mouth are tolerant towards and tolerated by the immune system, because inflammation leading to loss of teeth will eventually kill both the host and the bacteria[8]. This is why some of the bacteria do not produce a negative reaction in the host.
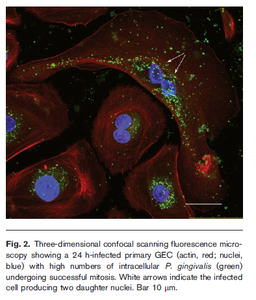
P. gingivalis colonises epithelial cells in the gingival compartment, which sometimes detect the bacteria and release inflammatory cytokines, however no particular apoptosis is noted in colonised cells[6]. The bacteria is able to adhere to the epithelial cells due to its fimbriae, and enter the cells by binding to the β1 integrin receptor[6]. It is capable of intracellular replication. Infected cells as with colonised cells do not display apoptosis, as the typical apoptotic pathways are inhibited by P. gingivalis while it replicates. In this way the bacteria is able to maintain viability within the epithelial cells for large amounts of time[6]. The mouth seems to be the only habitat that this bacteria can survive in, as there is no evidence of free-living P. gingivalis[8]. This makes sense, as the bacteria has very restrictive metabolic requirements. It is also suggested that the surface of the tooth may be required for colonisation, which is another factor reducing the ability of P. gingivalis to replicate outside the oral cavity. The evidence supporting this hypothesis is that the bacteria has not been cultured from edentulous babies or elderly[8]. Other mammalian species are colonised by P. gingivalis, however the strains are genetically dissimilar and distinct.
Pathology
P. gingivalis is able to causes disease in humans due to a number of virulence factors allowing it to evade the immune system and colonise regions within the host. It has three main categories of virulence factors: colonisation, host evasion, and host attacking[5]. Below is a list of the different virulence factors for each[5]:
Colonisation:
- Fimbriae (Also important in evading host)
- Hemagglutinin
- Outer membrane proteins
- Gingipains (help sequester iron and promote bacterial growth)
Evading Host:
- Capsule (antigenic and phase variation makes an acquired immune response difficult)
- LPS (a potent immunogenic endotoxin)
- Complement Proteases (reduce the effectiveness of a complement immune response)
- biofilm
Damaging Host:
- Proteinases
- Collagenase
- Lytic enzymes
These virulence factors, combined with the predisposition of P. gingivalis for colonising the gingiva can often result in periodontitis. This is a disease for which P. gingivalis has been heavily implicated. Periodontitis is a disease which results in the degradation of supporting periodontal tissues, this leads to poor attachment of the enamel to bone[9]. This reduced connection means that patients often experience tooth loss. This is the main observed form of pathology resulting from P. gingivalis infections. The cause of this degradation is chronic inflammation, which attacks the gingiva, the periodontal ligament, and the alveolar bone which connects the jaw to the teeth[10]. Periodontitis is essentially an extreme form of gingivitis, which a large portion of the population experiences. Gingivitis presents as light inflammation of the gingiva. In periodontitis the inflammation goes much deeper into the tissue, resulting in the formation of deep pits filled with P. gingivalis. It is this inflammation which attacks the gingival support structures around the teeth. The resultant breakdown of fibre and resorption of alveolar bones can lead to the movement and loss of teeth[10].
Researchers have found that there is a connection between P. gingivalis causing periodontitis and type II diabetes[10,11]. However it is postulated this connection is only because type II diabetes results in susceptibility to P. gingivalis infections. Other researchers have found possible connections between P. gingivalis and other diseases such as cardiovascular disease and artherosclerosis[12]. Borgnakke et al. (2013) surveyed a great deal of literature with the proposal that periodontitis is often connected to general health of the patient, their search uncoverered a great deal of information to suggest that this might be the case[13]. The formation of a biofilm by this bacteria is of significance to its virulence, and this is considered to be a potential target for therapeutic treatment[7]. LPS also plays a large role in the development of periodontitis, interaction with cells in peridontic lesions leads to a large immune response as LPS is a potent endotoxin and highly immunogenic[7].
Application to biotechnology
P. gingivalis generally relies upon teeth for colonisation of the mouth, and is strictly anaerobic, therefore it exists as a very specialised bacteria in the oral cavity[7]. On account of this, as well as their metabolic requirements, they are difficult to culture in large numbers. They do not make any commercially relevant metabolic products, therefore most of the biotechnology surrounding this species is aimed at removing them from periodontitis patients, rather than utilising them for production. There are currently a number of chemical treatments available which can target P. gingivalis, but most of these result in undesirable side effects such as vomiting or tooth-staining[14]. As a result there is an increasing demand for naturally produced biomaterials which are biocompatible, biodegradable, and do not produce undesirable side effects. There are a number of materials such as hydrogels, which fit this role and additionally can be chemically modified or loaded with drugs in order to slowly release the treatment[15]. One of the major materials being investigated for this application is chitosan, it displays a number of desirable characteristics such as biodegradability, and antimicrobial properties[14]. Pullulan is another naturally formed compound being investigated, as it is already being used in some dental care products[14].
Extensive research seems to indicate genetically modified P. gingivalis has never been used to produce useful bacterial products. Furthermore there is no evidence that any of the unique cellular products from P. gingivalis have ever been converted into a useful technology. Therefore, the only biotechnology relevant to P. gingivalis, is technology targeted at the bacteria. In addition to the compounds mentioned previously, there are a number of other research projects for detecting P. gingivalis, such as by using nanobodies[16].
Current research
There is currently a great deal of research underway regarding P. gingivalis, with most of the literature surrounding its role in periodontitis. Therefore the most common type of papers are investigating different methods for the prevention of P. gingivalis infection. The relative abundance of this sort of research is due to the large burden of periodontitis on society, with 40-60% of the adult population having the mild form of the disease, and 10% having an advanced form[10]. Some researchers are investigating treatments to prevent/remove the growth of P. gingivalis biofilms from the teeth and gingiva. Wang et al. (2016) looked into composite materials that would reduce the growth of a number of bacterial biofilms including P. gingivalis, they recorded the number of colony forming units with after each treatment[17]. Their results indicated that a treatment of 3% DMAHDM and 3%MPC resulted in a four orders of magnitude reduction in biofilm growth relative to the control used[17].
Khalad et al (2014) have been studying the difference in host cytokines and chemokines expression when the disease is present. They observed that TGF-β1 was consistently elevated in patients with P. gingivalis, whereas CXCL8, IL-6, and IL-2 were all under expressed in patients with periodontitis[18]. This research could potentially lead to new diagnostic tests for early detection of the disease. In addition to looking into new innovative solutions to prevent periodontitis, some researchers are carrying out randomised clinical trials. Cosgarea et al. (2016) investigated the treatment of 3-7 days of amoxicillin and metronidazole following periodontal surgery in chronic periodontitis patients[19]. The treatment resulted in significant improvements in the patients health and the three and six month mark. Overall there is a large amount of research underway regarding P. gingivalis, with most of it aimed at preventing or treating periodontitis.
References
1. StrainInfo. 2016. Taxon Passport Porphyromonas gingivalis. http://www.straininfo.net/taxa/6891;jsessionid=72D629E303BDD72F6608E86FA40AF430.straininfo2. Accessed 21st September
2. Bostanci N, Belibasakis GN. 2012. Porphyromonas gingivalis: an invasive and evasive opportunistic oral pathogen. FEMS Microbiology Letters 333:1-9.
3. Maley J, Shoemaker NB, Roberts IS. 1992. The introduction of colonic-Bacteriodes shuttle plasmids into Porphyromonas gingivalis: Identification of a putative P. gingivalis insertion-sequence element. FEMS Microbiology Letters 93:75-81.
4. Watanabe T, Maruyama F, Nozawa T, Aoki A, Okano S, Shibata Y, Oshima K, Kurokawa K, Hattori M, Nakagawa I, Abiko Y. 2011. Complete Genome Sequence of the Bacterium Porphyromonas gingivalis TDC60, Which Causes Periodontal Disease. Journal of Bacteriology 193:4259-4260.
5. Pandit N, Changela R, Bali D, Tikoo P, Gugnani S. 2015. Porphyromonas gingivalis : Its virulence and vaccine. J Int Clin Dent Res Organ 7:51-58.
6. Yilmaz Ö. 2008. The chronicles of Porphyromonas gingivalis: the microbium, the human oral epithelium and their interplay. Microbiology 154:2897-2903.
7. Mysak J, Podzimek S, Sommerova P, Lyuya-Mi Y, Bartova J, Janatova T, Prochazkova J, Duskova J. 2014. Porphyromonas gingivalis: Major Periodontopathic Pathogen Overview. Journal of Immunology Research 2014:8.
8. Asikainen S, Chen C. 1999. Oral ecology and person-to-person transmission of Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis. Periodontology 2000 20:65-81.
9. Griffen A, Becker M, Lyons S, Moeschberger M, Leys E. 1998. Prevalence of Porphyromonas gingivalis and Periodontal Health Status. Journal of Clincal Microbiology 36:3239-3242.
10. Preshaw P, Bisset S. 2013. Periodontitis Oral Complication of Diabetes. Endocrinology and Metabolism Clinics 42:849-867.
11. Ojima M, Takeda M, Yoshioka H, Nomura M, Tanaka N, Kato T, Shizukuishi S, Amano A. 2005. Relationship of Periodontal Bacterium Genotypic Variations With Periodontitis in Type 2 Diabetic Patients. Diabetes Care 28:433-434.
12. Desvarieux M, Demmer RT, Rundek T, Boden-Albala B, Jacobs DR, Sacco RL, Papapanou PN. 2005. Periodontal Microbiota and Carotid Intima-Media Thickness. The Oral Infections and Vascular Disease Epidemiology Study (INVEST). 111:576-582.
13. Borgnakke WS, Glick M, Genco RJ. 2013. Periodontitis: The canary in the coal mine. The Journal of the American Dental Association 144:764-766.
14. García AR, Wong LJG, Star JV, González MSF, Niño KA. 2012. In vitro evaluation of biopolymers as delivery system of plant extracts on cultures of Porphyromonas gingivalis and Agreggatibacter actinomycetemcomitans, p 479-483, Microorganisms in Industry and Environment doi:doi:10.1142/9789814322119_0104. WORLD SCIENTIFIC.
15. Slaughter BV, Khurshid SS, Fisher OZ, Khademhosseini A, Peppas NA. 2009. Hydrogels in Regenerative Medicine. Advanced Materials15 21:3307-3329.
16. Skottrup PD, Leonard P, Kaczmarek JZ, Veillard F, Enghild JJ, O’Kennedy R, Sroka A, Clausen RP, Potempa J, Riise E. 2011. Diagnostic evaluation of a nanobody with picomolar affinity toward the protease RgpB from Porphyromonas gingivalis. Analytical Biochemistry 415:158-167.
17. Wang L, Xie XJ, Imazato S, Weir MD, Reynolds MA, Xu HHK. 2016. A protein-repellent and antibacterial nanocomposite for Class-V restorations to inhibit periodontitis-related pathogens. Materials Science & Engineering C-Materials for Biological Applications 67:702-710.
18. Khalaf H, Lönn J, Bengtsson T. 2014. Cytokines and chemokines are differentially expressed in patients with periodontitis: Possible role for TGF-β1 as a marker for disease progression. Cytokine 67:29-35.
19. Cosgarea R, Juncar R, Heumann C, Tristiu R, Lascu L, Arweiler N, Stavropoulos A, Sculean A. 2016. Non-surgical periodontal treatment in conjunction with 3 or 7 days systemic administration of amoxicillin and metronidazole in severe chronic periodontitis patients. A placebo-controlled randomized clinical study. Journal of Clinical Periodontology 43:767-777.
20. The Brain Bank. 2015. Diabetes & Periodontitis – A Deadly Combination. http://thebrainbank.scienceblog.com/2015/07/26/diabetes-periodontitis-a-deadly-combination/. Accessed 23rd September.
21. Moreno SM, Contreras A. 2013. Functional differences of Porphyromonas gingivalis fimbriae's determining periodontal disease pathogenesis: A Literature Review, vol 44.
This page is written by Thomas Clarkson for the MICR3004 course, Semester 2, 2016