Viruses as Pest Control: the coevolution of the Myxoma virus and its long-eared hosts
By Ilana Richter
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2018, Kenyon College.
Background
European colonists introduced rabbits to Australia at the end of the 1700s [1]. European rabbits (Otyctolagus cuniculus) are native to southern Europe and northern Africa, but are considered an invasive pest species around the world. The rabbit population in Australia grew exponentially due to the lack of natural predators and warm climate that enabled year-round breeding. Australia was home to an estimated 10 billion rabbits by the 1920s [2]. The Australian rabbit population caused widespread vegetation depletion, leading to soil erosion [3]. The rabbits caused massive destruction of crops and pastures for livestock, resulting in a significant loss of agricultural productivity and harming the economy. Rabbits competed for resources with native herbivores, and themselves became food for the increasing population of native predators. These factors heavily contributed to the decline of native Australian biodiversity decline [4]. Australian farmers attempted to use various chemical pesticides, but those also proved detrimental to the ecosystem. In the 1950s, scientists proposed using the recently-discovered Myxoma virus to curb the rabbit population. This experiment in biological warfare ultimately failed to eliminate the rabbit infestation, but became an important experiment on the coevolution of a pathogen and its host.
Myxoma virus
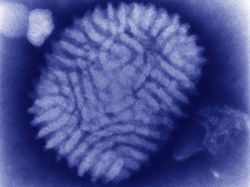
Myxoma viruses are poxviruses that live commensally inside lagomorphs in North and South America[5]. South American Myxoma resides inside the tapeti ((Sylvilagus brasiliensis). Californian Myxoma resides inside the brush rabbit (S. bachmani). The virus only causes benign skin tumours in its reservoir hosts. The virus is vectored by mosquitoes and fleas, but it can also be spread through direct contact with an infected animal[6]. Myxoma virions are enveloped and rectangular (pictured in Figure 1). The envelope includes lipids derived from the host cell and glycolipids synthesized from the viral genome. Like many poxviruses, they contain a bioconcave core[7]. Myxoma has a nonsegmented genome consisting of a single molecule of linear double-stranded DNA[8]. The viral genome consists of 171 unique genes and terminates with inverted repeats and hairpin structures. Virions replicate within the cytoplasm of the host cell and reproduce by budding out of the host cell's membrane. The Myxoma virus infects fibroblasts, lymphocytes, and mucosal cells[9]. The virions produce many proteins that are designed to attack or avoid the immune system’s of European rabbits. The M-T7 protein of the pathogen binds to the IFN-gamma protein of the rabbit host. This binding inhibits an immunoregulatory cytokine protein. [10]The M11L gene inhibits apoptosis, allowing the Myxoma virion to survive in a larger range of environments. [11]The Myxoma virus mimics tumor necrosis factor receptors (a type of cytokine), reducing the host’s ability to trigger an immune or inflammation response[12]. Myxoma also downregulates MHC molecules on the surface of infected cells, preventing recognition by cytotoxic T-cells.
The Myxoma virus is notable because it demonstrates extremely high host specificity. The Myxoma virus is highly infectious in rabbits, but is unable to replicate or cause symptoms in any other species. Additionally, it is only known to cause severe pathology in European rabbits and their descendents. A 2009 experiment examined the effect of the South American strain of the Myxoma virus on two species of North American rabbits. One species (S. nuttallii) developed skin lesions, but did not develop classic myxomatosis. The virus remained in significant levels in their systems for over 20 days. The other rabbit species (S. audubonii) displayed almost no evidence of systemic infection. The researchers also tested the effect of California Myxoma. This strain was able to replicate in both hosts, but did not cause myxomatosis nor reach transmissible titres at the inoculation sites in either species. [13]
Host species
European Rabbits (Oryctolagus cuniculus) are lagomorphs native to continental Europe. An adult typically weighs 3-5 lbs, and males are typically larger than females. They are typically grey-brown in color. The European breeding season is between January and August, because of the warm temperature[14].The females are pregnant for one month and are known to mate immediately after giving birth. Females birth multiple litters per breeding season, each consisting of 3-7 kits. Rabbit kits are born naked, deaf, and blind. They are completely dependent on their mothers for the first 21-25 days after birth. Young rabbits reach sexual maturity at 3-5 months of age. Wild rabbits have an average lifespan of nine years.
Rabbits prefer to construct warrens (burrows) in grassland with trees or shrubbery. Their diet consists of grass and plant shoots. They ingest their own feces and utilize hindgut fermentation to extract the maximum amount of nutrients from plant matter. In Europe, their natural predators include foxes, wolves, domestic dogs, domestic cats, lynxes, weasels, badgers, and large birds of prey. Rats often eat rabbit kits. Rabbits have been known to die of fright when pursued by a predator or otherwise extremely stressed[15].
Humans have been hunting rabbits for thousands of years. Every breed of domestic rabbit is descended from the European rabbit. Yong, Ed. “People Have Believed a Lie About Rabbit Domestication for Decades.”[16]. Rabbits multiply rapidly and destroy vegetation, especially when introduced to new environments. They have been introduced to islands around the world including the United Kingdom, Hawaii, San Juan, and New Zealand.
Governments throughout history have taken countermeasures to prevent the spread of invasive rabbits. The first record of invasive rabbits was documented in the first century, BCE. The Romans introduced rabbits to the Balearic Islands. The rabbits ate the crops, causing famine, and even destroyed infrastructure by tunneling. Emperor Augustus sent an army of men and ferrets to exterminate the rabbits[17].
Pathology
Most lagomorphs are susceptible to the Myxoma virus, but the virus is easily targeted by the immune systems of other lagomorph species. The reservoir hosts of the virus (wild American rabbits) do not usually exhibit symptoms, except for benign skin tumors. However, infected European rabbits contract the deadly disease myxomatosis. Domestic rabbits are also susceptible to this disease, as all domestic rabbits are descended from European rabbits. Vaccines against myxomatosis are commonly administered to domestic rabbits around the world, except in the United States. [18].
The first clinical sign of acute myxomatosis is conjunctivitis, which later progresses into total blindness. The rabbit will then become feverish and listless, becoming unable to eat or sleep. Early symptoms of the disease include edema and hemorrhage of the capillaries and soft tissues. Swollen heads and genitals are characteristic of infected rabbits (pictured in Figure 2). As the disease progresses, it causes fever, loss of appetite, and insomnia. Finally, the respiratory system malfunctions, leading to coma then death of the host[19]. Death occurs between two days and two weeks after symptoms appear. Less acute myxomatosis often manifests as development of mucinous skin lesions and fibrous tumors, especially at the site of infection. The Myxoma virus produces extracellular proteins that specifically evade a European rabbit’s immune response[20].
Experimental use in Australia
Conventional methods of rabbit elimination had failed to solve the problem. Poisoning rabbits and burning their warrens were expensive and laborious options, and ultimately incapable of destroying rabbits at a higher rate than they could reproduce [21]. The desperate government of New South Wales offered a £25,000 reward for “any method of success not previously known in the Colony for the effectual extermination of rabbits.” Over one thousand proposals were submitted [22]. The idea of using a biological control agent was unconventional yet promising, and Myxoma was a perfect candidate. The Myxoma virus only affects rabbits, but it had an incredibly high mortality rate.
Preliminary safety tests were performed in the 1930s (pictured in Figure 3), and myxomatosis positive rabbits were successfully released in Australia in 1950 [23]. The experiment immediately saw dramatic results. The Myxoma virus eliminated over 80% of the rabbit population over two years. [24]. Myxomatosis has a mortality rate of 99.8% upon release. Australia’s agricultural industry rapidly recovered. Mosquitoes do not live in arid deserts, so the virus could not spread across the entire country, but the program was still initially hailed as a resounding success [25]. However, the rabbit population soon began to recover. The rabbit population rebounded to almost 300 million by 1991 [26]. What had changed?
On coevolution
The highly lethal Myxoma virus placed the rabbit population under extreme selective pressure. The virus killed the vast majority of the susceptible rabbit population, leaving only somewhat resistant rabbits behind. These resistant rabbits reproduced and created resistant offspring. Over time, the population became more resistant to myxomatosis. However, as the rabbits gained resistance, the disease evolved so the rabbits were no longer resistant to its new form. By the 1990s, the virus had evolved the ability to totally deactivate the host’s immune system[27]. This severe immunosuppression allowed opportunistic infections by gram-negative bacteria, which resulted in the demise of the host.
A pathogen that has just been introduced to a species is often extremely lethal. This level of virulence is unsustainable, because if the parasite kills its host before it can spread to a new host, the parasite will also die. Evidence shows that such pathogens quickly evolve to become less virulent and more transmissible in a new host species[28] At first, this was the case with myxomatosis. The strain of Myxoma that was introduced to Australia, Strain I, was almost 100% lethal to infected rabbits within two weeks. Within a few years, it had mutated into multiple strains with lower mortality rates. Strain II had a mortality rate of 95-99% within two weeks. Strain III had a mortality rate of 70-95% within one month. Strain IV had a mortality rate of 50%-70% within 50 days. Finally, Strain V had a mortality rate of less than 50% [29]. This supported the belief that parasites evolve to become less virulent or even benign over time ex: the Myxoma virus lives within the bodies of American rabbits without causing them any harm. However, the Australian myxomatosis experiment provides strong evidence that nature does not necessarily conform to this pattern[30]. As rabbit populations evolved resistance to the milder strains, the virus coevolved to become more virulent again. Virulence and transmission were strongly negatively correlated in this instance because the virus spread through vectors. An infected rabbit needed to be bitten by a flying insect before dying. The rabbit must have born a high viral load in order for the virus to adhere to the mouthparts of the insect[31]. Myxoma strains with high mortality rates only remained in the rabbits’ systems for a few days before death. The window for transmission was brief in the most lethal strains. However, mild strains also did not remain in the rabbits’ systems for long, because they were managed by the hosts’ immune systems. The dominant strain (III) in Australia was of relatively moderate virulence. Instead of continuing to decrease in virulence Myxoma virus seems to have stabilized at a very high mortality rate (~75%).
Australia was not the only country to use myxomatosis to mitigate their rabbit infestation; the United Kingdom and France also experimented with viral pest control. Initially, the virus devastated rabbit populations, but rabbits soon developed immunity. Researchers compared the DNA of European rabbits collected before the Myxoma outbreaks with modern resistant populations. They discovered that the genomes had changed identically across all three locations, allowing the scientists to determine which genes conferred immunity to myxomatosis [32].
Genomic analysis
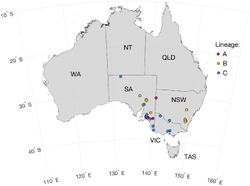
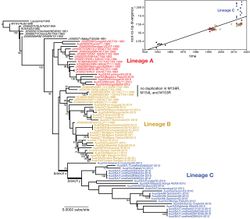
A recent study on the molecular phylogeny and evolution of the Myxoma virus was conducted by Dr. Peter Kerr and published in the Journal of Virology in 2018. His team analyzed 49 Myxoma genome sequences isolated in Australia between 2008 and 2017, as well as one archival sample from 1990[33]. They discovered that Australian Myxoma had evolved into three distinct lineages since 1950 (Figure 4). A previous study determined that the evolutionary rate of the Myxoma virus was relatively high and remarkably reliable. This held true for the first two lineages. The third, diverse, widespread lineage exhibited an elevated rate of evolutionary change (ie: more non-synonymous mutations) between 1996 and 2012 (Figure 5). The authors did not find evidence of mutations in DNA replication or proof-reading genes that could then lead to more mutations. There was little evidence for abnormal genetic recombination. The wild Australian rabbit population significantly declined during this time period due to the release of RHDV and a prolonged drought. The authors believe that these new selection pressures accelerated the evolution of the Myxoma virus. These factors likely combined to reduce the number of rabbits that were susceptible to Myxoma infection, creating extreme selection pressure upon the virus to evolve rapidly[34]. In addition, the Spanish rabbit flea (Xenopyslla cunicularis) became an established vector of Myxoma during this time period. The researchers surmised that the fleas replaced mosquitoes as the main vectors, changing the geographic transmission patterns of the virus. The genetic determinants of virulence in wild viral strains are multifactorial and not easily explained by knock-out studies. Their study did not conclude which genetic mutations were most impacted by the changed environment, but they suggested two genes to focus on in future investigations[35].
Aftermath
The Myxoma virus was also introduced to France in 1952. However, the European rabbit is a critical component of the ecosystem in its native range. Rabbits are an integral part of the diets of 29 predator species, including the endangered Iberian lynx[36]. Myxomatosis and RHD have decimated the rabbit population and subsequently threatened their predator populations. Many attempted strategies have failed to repair the state of the rabbit population. All future management strategies must include the goal to vaccinate wild rabbits against myxomatosis in order to succeed[37]. Unfortunately, this is not practical with current vaccines, because they have to be administered to each rabbit individually. Scientists are currently working on developing a vaccine that can be horizontally transmitted between rabbits without further human intervention[38]. Twenty strains of Myxoma have been isolated from wild rabbits. One isolate, 6918, had mutated from the original Myxoma strain to disrupt several known virulence factor genes[39]. This isolate appears to be a promising candidate for horizontal transmission of myxomatosis resistance through the wild rabbit population. Ideally, the vaccine should be administered to the population during a two-month window between the birth of vulnerable rabbit kits and before the start of annual epidemics[40].
When myxomatosis failed to eradicate Australia’s rabbit infestation, the Australian government approved use of a second biological weapon against the rabbits: RHDV. Rabbit hemorrhagic disease viruses (RHDV) are caliciviruses that also primarily affect European rabbits. RHD affects more species of lagomorph than does myxomatosis. RHD is also highly lethal, with a mortality rates of 70-100%[41]. RHDV virions are non-enveloped and spherical. They have a linear, positive sense RNA genome of ~6400-8500 base pairs. RHDV replicates inside hepatic cells, causing necrosis and liver failure and hepatitis. Symptoms include lethargy, fever, internal bleeding, coma, and death[42]. However, rabbits often die without showing clinical symptoms. Some rabbits do not experience symptoms at all and become carriers of the disease. Transmission mainly occurs through direct contact with infected animals, but the disease can also be vectored by insects. The first pathogenic strain of RHDV was first reported in China in 1984. A second strain of RHDV (RHDV-2) was discovered in France in 2010. This strain was notable because rabbits who had been vaccinated for RHDV-1 were not immune. Both strains have since spread through rabbits and hares around the world.
In 1991, scientists tested the pest control potential of this new virus, on the same island used for the Myxoma trials (Figure 3). The virus escaped the island and spread throughout the continent within five years[43]. RHDV proved to be lethally effective, especially in arid regions. New Zealand declined to import the virus, but it was illegally introduced anyways. After years of testing, a new strain of RHDV was deliberately released in Australia in 2017 to target the cold, wet regions of the country[44]. So far, RHD viruses have managed to successfully decrease the rabbit population that rebounded after they evolved resistance to Myxoma, which subsequently evolved to become less virulent. Only time will tell whether RHDV will face the same fate as Myxoma.
References
- ↑ "Rabbits – fact sheet". www.environment.nsw.gov.au. Retrieved 2020-04-21.
- ↑ Zukerman, Wendy. “Australia's Battle with the Bunny.” ABC Science, ABC, 8 Apr. 2009, www.abc.net.au/science/articles/2009/04/08/2538860.htm.
- ↑ "Environmental Damage caused by Rabbits" (PDF). Archived from the original (PDF) on 18 December 2012.
- ↑ Cooke, Brain D. 2012. "Rabbits: manageable environmental pests or participants in new Australian ecosystems?". Wildlife Research. 39 (4): 280. doi:10.1071/WR11166
- ↑ Kerr, Peter. 2013. "Viral Infections of Rabbits". Veterinary Clinics of North America: Exotic Animal Practice. 16 (2): 437–468. doi:10.1016/j.cvex.2013.02.002
- ↑ Upton, C., Mossman, K., and McFadden, G. "Encoding of a homolog of the IFN-gamma receptor by myxoma virus." Science. 1992; 20:1369-1372.
- ↑ Su, J., Wang, G., Barrett, J., Irvine, T., Gao, X., and McFadden, G. 2006. Myxoma virus M11L blocks apoptosis through inhibition of conformational activation of Bax at the mitochondria. J. Virol 80: 1140-51.
- ↑ Mahy, Brian W J; Van Regenmortel, Marc H (2008), The Encyclopedia of Virology, I (3rd ed.), San Diego, CA: Academic Press, p. 157
- ↑ Kerr, Peter. 2013. "Viral Infections of Rabbits". Veterinary Clinics of North America: Exotic Animal Practice. 16 (2): 437–468. doi:10.1016/j.cvex.2013.02.002
- ↑ Upton, C., Mossman, K., and McFadden, G. "Encoding of a homolog of the IFN-gamma receptor by myxoma virus." Science. 1992; 20:1369-1372.
- ↑ Su, J., Wang, G., Barrett, J., Irvine, T., Gao, X., and McFadden, G. 2006. Myxoma virus M11L blocks apoptosis through inhibition of conformational activation of Bax at the mitochondria. J. Virol 80: 1140-51.
- ↑ Mahy, Brian W J; Van Regenmortel, Marc H (2008), The Encyclopedia of Virology, I (3rd ed.), San Diego, CA: Academic Press, p. 157
- ↑ Silvers, L., Barnard, D., Knowlton, F., Inglis, B., Labudovic, A., Holland, M., Janssens, P., Van Leeuween, B., and Kerr, P. 2010. Host-specificity of myxoma virus: Pathogenesis of South American and North American strains of myxoma virus in two lagomorph species. Veterinary Microbiology 141: 289-300.
- ↑ Lockley, R. M. 1976. The Private Life of the Rabbit. Readers Union Group of Book Clubs.
- ↑ Hoffman, R.S.; Smith, A.T. 2005. "Order Lagomorpha". In Wilson, D.E.; Reeder, D.M (eds.). Mammal Species of the World: A Taxonomic and Geographic Reference (3rd ed.). Johns Hopkins University Press. pp. 205–206.
- ↑ The Atlantic, Atlantic Media Company, 14 Feb. 2018, www.theatlantic.com/science/archive/2018/02/eh-whats-up-doc/553304/
- ↑ López Seoane, V. (1861) Fáuna Mastológica de Galicia, ó historia natural de los mamíferos de este antiguo Reino, aplicada á la medicina, á la agricultura, á la industria, á las artes y al comercio. Manuel Mirás, 544 pages.
- ↑ Cavadini, P., Barbieri, I., Lavazza, A., and Capucci, L. 2010. Molecular characterization of SG33 and Borghi vaccines used against myxomatosis. Vaccines 28: 5714-5420.
- ↑ Kerr, Peter. 2013. "Viral Infections of Rabbits". Veterinary Clinics of North America: Exotic Animal Practice. 16 (2): 437–468. doi:10.1016/j.cvex.2013.02.002
- ↑ Stanford, M., Werden, S., and McFadden, G. "Myxoma virus in the European rabbit: interactions between the virus and its susceptible host." Veterinary Research 2007; 38: 299-318.
- ↑ Zukerman, Wendy. “Australia's Battle with the Bunny.” ABC Science, ABC, 8 Apr. 2009, www.abc.net.au/science/articles/2009/04/08/2538860.htm.
- ↑ Andrews, Stefan. 2017. “In 1950, a Disease Called Myxomatosis Was Introduced to Rabbits for Population Control in Australia and It Wiped out 500 Million Rabbits in Two Years.” The Vintage News. www.thevintagenews.com/2017/03/30/in-1950-a-disease-called-myxomatosis-was-introduced-to-rabbits-for-population-control-in-australia-and-it-wiped-out-500-million-rabbits-in-two-years/.
- ↑ “The Virus That Stunned Australia's Rabbits.” CSIRO, Internet Archive- Wayback Machine, 25 Feb. 2011, web.archive.org/web/20111125204128/www.csiro.au/science/Myxomatosis-History.
- ↑ Andrews, Stefan. 2017. “In 1950, a Disease Called Myxomatosis Was Introduced to Rabbits for Population Control in Australia and It Wiped out 500 Million Rabbits in Two Years.” The Vintage News. www.thevintagenews.com/2017/03/30/in-1950-a-disease-called-myxomatosis-was-introduced-to-rabbits-for-population-control-in-australia-and-it-wiped-out-500-million-rabbits-in-two-years/.
- ↑ Zukerman, Wendy. “Australia's Battle with the Bunny.” ABC Science, ABC, 8 Apr. 2009, www.abc.net.au/science/articles/2009/04/08/2538860.htm.
- ↑ Andrews, Stefan. 2017. “In 1950, a Disease Called Myxomatosis Was Introduced to Rabbits for Population Control in Australia and It Wiped out 500 Million Rabbits in Two Years.” The Vintage News. www.thevintagenews.com/2017/03/30/in-1950-a-disease-called-myxomatosis-was-introduced-to-rabbits-for-population-control-in-australia-and-it-wiped-out-500-million-rabbits-in-two-years/.
- ↑ Yong, Ed. “The Next Chapter in a Viral Arms Race.” The Atlantic, Atlantic Media Company, 14 Aug. 2017, www.theatlantic.com/science/archive/2017/08/rabbit-virus-arms-race/536796/.
- ↑ Read, Andrew F., and Peter J. Kerr. “Infographic: Evolving Virulence.” TheScientist, The Scientist Magazine, 30 Sept. 2017, www.the-scientist.com/multimedia/infographic-evolving-virulence-30813.
- ↑ Fenner F. & Marshall I.D. (1957). - A comparison of the virulence for European rabbits (Oryctolagus cuniculus) of strains of myxoma virus recovered in the field in Australia, Europe and America. J. Hyg., Camb., 55, 149-191.
- ↑ Yong, Ed. “The Next Chapter in a Viral Arms Race.” The Atlantic, Atlantic Media Company, 14 Aug. 2017, www.theatlantic.com/science/archive/2017/08/rabbit-virus-arms-race/536796/.
- ↑ Fenner F., Day M.F. & Woodroofe G.M. (1956). - Epidemiological consequences of the mechanical transmission of myxomatosis by mosquitoes. J. Hyg., Camb., 54, 284-303.
- ↑ Davis, Josh. "Darwin's rabbit is revealing how the animals became immune to myxomatosis". Natural History Museum. Retrieved 14 August 2019.
- ↑ Kerr, P, John-Sebastian Eden, Francesca Di Giallonardo, David Peacock, June Liu, Tanja Strive, Andrew F. Read, Edward C. Holmes. 2018. Punctuated evolution of myxoma virus: rapid and disjunct evolution of a recent viral lineage in Australia. Journal of Virology. doi: https://doi.org/10.1101/465583
- ↑ Kerr, P, John-Sebastian Eden, Francesca Di Giallonardo, David Peacock, June Liu, Tanja Strive, Andrew F. Read, Edward C. Holmes. 2018. Punctuated evolution of myxoma virus: rapid and disjunct evolution of a recent viral lineage in Australia. Journal of Virology. doi: https://doi.org/10.1101/465583
- ↑ Kerr, P, John-Sebastian Eden, Francesca Di Giallonardo, David Peacock, June Liu, Tanja Strive, Andrew F. Read, Edward C. Holmes. 2018. Punctuated evolution of myxoma virus: rapid and disjunct evolution of a recent viral lineage in Australia. Journal of Virology. doi: https://doi.org/10.1101/465583
- ↑ Ferrer M, Negro J. The near extinction of two large European predators: super specialists pay a price. Conserv Biol. 2004;18:344–349. doi: 10.1111/j.1523-1739.2004.00096.x.s
- ↑ García-Bocanegra I, Astorga RJ, Napp S, Casal J, Huerta B, Borge C, Arenas A. Myxomatosis in wild rabbit: design of control programs in Mediterranean ecosystems. Prev Vet Med. 2010 Jan 1; 93(1):42-50.
- ↑ Spiesschaert, B., McFadden, G., Hermans, K., Nauwynck, H., & Van de Walle, G. R. (2011). The current status and future directions of myxoma virus, a master in immune evasion. Veterinary research, 42(1), 76. https://doi.org/10.1186/1297-9716-42-76
- ↑ Bárcena J, Pagès-Manté A, March R, Morales M, Ramírez MA, Sánchez-Vizcaíno JM, Torres JM. Isolation of an attenuated myxoma virus field strain that can confer protection against myxomatosis on contacts of vaccinates. Arch Virol. 2000; 145(4):759-71.
- ↑ Merchant JC, Kerr PJ, Simms NG, Hood GM, Pech RP, Robinson AJ. Monitoring the spread of myxoma virus in rabbit Oryctolagus cuniculus populations on the southern tablelands of New South Wales, Australia. III. Release, persistence and rate of spread of an identifiable strain of myxoma virus. Epidemiol Infect. 2003 Feb; 130(1):135-47.
- ↑ "Rabbits – fact sheet". www.environment.nsw.gov.au. Retrieved 2020-04-21.
- ↑ Cooke, Brain D. 2012. "Rabbits: manageable environmental pests or participants in new Australian ecosystems?". Wildlife Research. 39 (4): 280. doi:10.1071/WR11166
- ↑ "Rabbit Calicivirus Disease (RCD)" (PDF). CSIRO
- ↑ "Rabbit biocontrol: RHDV1 K5 national release". Department of Primary Industries and Regional Development. Government of Western Australia. 21 June 2017. Archived from the original on 21 October 2017