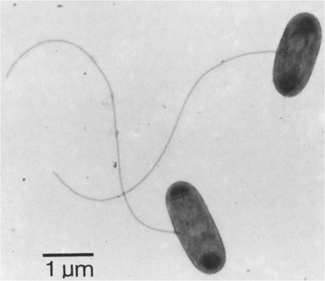
Sulfur-Reducing Bacteria and Archaea
Sulfur-Reducing Bacteria and Archaea
Introduction:
Sulfur-reducing bacteria (SRB) and archaea are a taxonomically diverse group of prokaryotes (Finster et al. 1997b; Schauder et at. 1993) which use elemental sulfur (S⁰) as their terminal electron acceptor (TEA) by reducing it to hydrogen sulfide (H2S) (Pfennig, and Biebl 1976), to generate energy.
The first SRB species to be discovered was Desulfuromonas acetoxidans which was isolated by Pfennig, and Biebl in 1976, from anaerobic sulfide containing marine mud sediments.
SRB are either strictly anaerobic, or microaerophilic, gram-negative, non-sporulating, flagellated vibrio and or rod shaped cells (Alain et al. 2009; Yoneda et al. 2012; Brock et al. 2009; Janssen et al. 2002; Pfennig, and Biebl 1976). SRB are only able to grow on a limited number of simple organic and inorganic carbon sources, including acetate, ethanol, carbon dioxide, or propionate (Yoneda et al. 2012; Sorokin et al. 2010; Alain et al. 2009; Brock et al. 2009).
There exist certain strains of various other respiratory types which are able to facultatively reduce S⁰. These include most sulfate-reducing bacteria (Brock 2009) and some dissimilatory Fe(III) reducing bacteria (Yoneda et al. 2012; Lovley et al. 1995). There also exist some SRB who are capable of microaerophilic O2 reduction e.g. Geovibrio thiophilus sp. (Janssen et al. 2002) and other S⁰ reducing species of Geobacter and Desulfuromonas which are also able to reduce nitrate or fumarate (Lovley et al. 1995).
Metabolism:
In terms of energetics the reduction of S⁰ to H2S is not very favorable, only producing a small amount of Gibbs free energy (Pfennig, and Biebl 1976). Regardless S⁰ reducers are able to obtain energy by catabolizing metabolic reactions which couple the oxidation of H2 to the reduction of S⁰ (as TEA) to provide energy for ATP synthesis via oxidative phosphorylation (Brock et al. 2009). Some SRB such as Desulfuromonas use the energy from S⁰ reduction to oxidize acetate to CO2 to produce growth substrates (Pfennig, and Biebl 1976). Desulfuromonas have high levels of cytochromes analogous to cytochrome c3 (Brock et al. 2009) which are thought to mediate electron transport to S⁰ (Pfennig, and Biebl 1976).
Unlike sulfate-reducing bacteria SRB are unable to attach sulfate to ATP to activate adenosine phosphosulfate (APS), which is required to reduced to sulfite, thus are unable to reduce sulfate (Brock et al. 2009).
Phylogeny:
There exist a wide variety of bacteria and archaea which respire via the reduction of S⁰ (Finster et al. 1997b).
SRB belong to the phylum Proteobacteria which can be separated into two distinct physiological groups based on classes divisions (Finster et al. 1997b). The first group comprising genera from the Deltaproteobacteria class are obligate anaerobes, which are able to completely oxidize organic substrates to CO2; they include Desulfuromonas, Desulfurella, Desulfuromonas, Geobacter, and Pelobacter (Finster et al. 1997b). The second group comprising genera from the Gamma and Epsilon Proteobacteria classes are microaerophiles, which are not able to completely oxidize organic substrates to CO2; they include Wolinella, Campylobacter, Shewanella, Sulfurospirillum, and Geospirillum bamesi (Finster et al. 1997b).
S⁰ reducing archaea are quite phylogenetically diverse at the phylum level they include but are not limited to the following orders: Thermococcales, Thermoproteales, Pyrodictales, Sulfolobales, Pyrodictales, and Sulfolobales (Schauder et at. 1993).
Ecology:
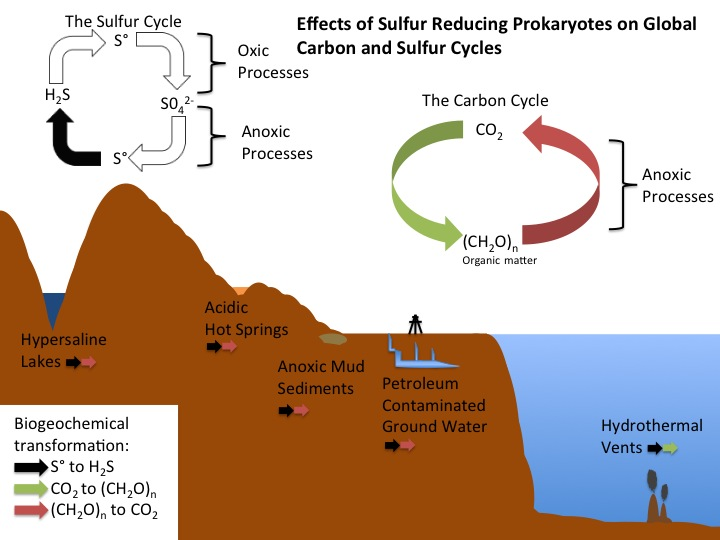
Sulfur reducing bacteria and archaeaare ubiquitously distributed throughout sulfur containing marine and terrestrial environments (Brock et al. 2009), and occupy the ecological niche of respiring S⁰ in anaerobic (Sorokin et al. 2010), or microaerophilic environments (Alain et al. 2009). It is of note that SRB tend to live in many of the same habitats as sulfate-reducing bacterial (Brock et al. 2009).
Environments from which SRB have been isolated include acidic hot springs (Yoneda et al. 2012), hydrothermal vents (Alain et al. 2009), hypersaline lakes (Sorokin et al. 2010), submarine thermal springs (Belkin et al. 1985), anaerobic marine mud sediments (Pfennig, and Biebl 1976), and anoxic mud from freshwater sulfur springs (Finster et al. 1997a).
Since certain environments in which sulfur reducing bacteria and archaea live are extremophilic thus pH and temperature optima can be used to classify S⁰ reducers into extremely, and moderately acidophilic subgroups (Schauder et at. 1993). The former consists only of archaea of various genera including Acidianus, Desulfurolobus and Stygiolobus living in low pH (2 to 3), and high temperature (80⁰ to 90⁰C) environments (Schauder et at. 1993). The latter group consists of the moderately acidophilic archaea, and SRB living in slightly alkaline (pH up to 8.5), and moderate temperature (30⁰ to 80⁰C) environments (Schauder et at. 1993). The high diversity and abundance of extremophilic S⁰ reducers may be due to S⁰ being more soluble at high temperature, and therefore more easily accessible as an electron acceptor to groups living in high temperature environments (Schauder et at. 1993).
In anoxic mud sediment environments SRB often form associations with other bacteria that oxidize H2S to S⁰ notably green sulfur bacteria (GSB) who provide the SRB with S⁰ (Pfennig, and Biebl 1976). SRB then reduce the S⁰ back to H2S which the GSB can use as their electron donor (Brock et al 2009).
In hydrothermal vent communities Epsilonproteobacteria SRB can be found as free-living organisms on or around vent chimneys, or plumes, or as endosymbionts of animals such as tube worms and shrimp (Alain et al. 2009; Brock et al. 2009). The metabolic S⁰ reduction performed by SRB helps to detoxify harmful sulfide compounds, allowing their endosymbiotic host organisms to live in otherwise toxic environments (Brock et al. 2009). Due to their high abundance and specialized metabolic activities Epsilonproteobacteria SRB are thought to play an important role in the deep-sea vent sulfur biogeochemical cycle (Alain et al. 2009).
Human effects:
There exist many cases of human affected or engineered environments in which sulfur reducing prokaryotes are found. Such examples include, but are not limited to engineered bioreactors (Sorokin et al. 2010), petroleum contaminated ground water (Kodama et al. 2007), and acid mine drainage sites (Riefler et al. 2008). In one such wet-land [acid mine drainage] treatment site, SRB were found to significantly increase alkalinity of the acidic sulfur containing sludge (Riefler et al. 2008).
The SRB genus Sulfurospirillum is of bioremedial interest as certain strains have been found to reduce a variety of anthropogenic compounds from polluted sites. Such Sulfurospirillum strains are able to reduce either trichloroethylene to cis-dichloroethene, selenate to elemental selenium, or arsenate to arsenite (Kodama et al. 2007), (Luijten et al. 2003). Sulfurospirillum’s ability to reduce these pollutants could potentially be important for bioremediation of soils polluted with chlorinated compounds and metal ions (Luijten et al. 2003).
References:
(1) Alain, K., Callac, N., Guégan, M., Lesongeur, F., Crassous, P., Cambon-Bonavita, M. “Nautilia abyssi sp. nov., a thermophilic, chemolithoautotrophic,sulfur-reducing bacterium isolated from an East Pacific Risehydrothermal vent.” International journal of Systematic and Evolutionary Microbiology. 2009, DOI: 10.1099/ijs.0.005454-0
(2) Brock, T., Madigan, M., Martinko, J., Dunlap, P., Clark, D., “Brock Biology of Microorganisms.” Pearson Benjamin Cummings. 12th ed. 2009, ISBN: 0-132-55077-3
(3) Belkin, S., Jannasch, H. “A new extremely thermophilic, sulfur-reducing heterotrophic, marine bacterium” Archives of Microbiology, 1985, DOI: 10.1007/BF00408055
(4) Biebl, H., Pfennig N. “Growth of sulfate-reducing bacteria with sulfur as electron acceptor.” Archives of microbiology, 1977, DOI: 10.1007/BF00446664
(5) Finster, K., Coates, J., Liesack, W., Pfennig, N. “Desulfuromonas thiophila sp. nov., a new obligately sulfur-reducingbacterium from anoxic freshwater sediment.” International journal of systematic bacteriology, 1997a, DOI: 10.1099/00207713-47-3-754
(6) Finster, K., Liesack, W., Tindall, B. “Sulfurospirillum arcachonense sp. nov., a new microaerophilic sulfur-reducing bacterium” Soc General Microbiol, 1997b, DOI: 10.1099/00207713-47-4-1212
(7) Janssen, P., Liesack, W., Schink, B. “geovibrio theophilus sp. nov., a novel sulfurreducens bacterium belonging to the phylum deferribacteres.” International Journal of Systematic and Evolutionary Microbiology, 2002, DOI: 10.1099/ijs.0.02116-0
(8) Kodama, Y., Ha, L., Watanabe, K. “Sulfurospirillum cavolei sp. nov., a facultatively anaerobicsulfur-reducing bacterium isolated from an underground crude oilstorage cavity” International Journal of Systematic and Evolutionary Microbiology, 2007, DOI: 10.1099/ijs.0.64823-0
(9) Lovley, D., Phillips, E., Lonergan, D., Widman, P. “Fe(III) and S0 reduction by Pelobacter carbinolicus.” Applied and environmental microbiology, 1995, DOI: 1070-6291
(10) Luijten, M., Weert, J., Smidt H., Boschker, H., de Vos, W., Schraa, G., Stams, A. “Description of Sulfurospirillum halorespirans sp nov., an anaerobic, tetrachloroethene-respiring bacterium, and transfer of Dehalospirillum multivorans to the genus Sulfurospirillum as Sulfurospirillum multivorans comb. nov.” International Journal of Systematic and Evolutionary Microbiology, 2003, DOI: 10.1099/ijs.0.02417-0
(11) Pfennig, N., Biebl, H. “Desulfuromonas acetoxidans gen. nov. and sp. nov., a new anaerobic,sulfur-reducing, acetate-oxidizing bacterium.” Archives of microbiology, 1976, DOI: 10.1007/BF00416962
(12) Riefler, R., Krohn, J., Stuart, B., Socotch, C. “Role of sulfur-reducing bacteria in a wetland system treating acid mine drainage” The Science of the total environment, 2008, DOI: 10.1016/j.scitotenv.2008.01.024
(13) Schauder, R., M ller, E. “Polysulfide as a possible substrate for sulfur-reducing bacteria.” Archives of Microbiology, 1993, DOI: 10.1007/BF00252224
(14) Sorokin, D., Muyzer, G. “Desulfurispira natronophila gen. nov. sp. nov.: an obligately anaerobic dissimilatory sulfur-reducing bacterium from soda lakes.” International Journal of Systemic bacteriology, 2010, DOI: 10.1007/s00792-010-0314-7
(15) Yoneda, Y., Yoshida, T., Kawaichi, S., Daifuku, T., Takabe, K., Sako, Y. “Carboxydothermus pertinax sp. nov., a thermophilic, hydrogenogenic, Fe(III)-reducing, sulfur-reducing carboxydotrophic bacterium from an acidic hot spring.” International Journal of Systematic and Evolutionary Microbiology, 2012, DOI: 10.1099/ijs.0.031583-0