Toxoplasma gondii
A Microbial Biorealm page on the genus Toxoplasma gondii
Classification
Higher order taxa
Cellular organisms; Eukaryota; Alveolata; Apicomplexa; Coccidia; Eucoccidiorida; Eimeriorina; Sarcocystidae; Toxoplasma [1]
Species
|
NCBI: Taxonomy |
Toxoplasma gondii [1]
Description and significance
Toxoplasma gondii is a single-celled eukaryotic protozoan parasite. The name Toxoplasma is derived from the shape of the organism, which is crescent-like (toxon is Greek for “arc”). T. gondii holds notoriety as the pathogen that causes the disease toxoplasmosis in humans. Infants (especially via vertical transmission) and immuno-compromised hosts (including cats) are readily infected with the pathogen and show accelerated and concentrated symptoms, but it is interesting to observe that the pathogen is generally docile and useless in healthy individuals with a competent immune system[7,8]. Nearly 1/3rd of the world human population has Toxoplasma infection, though this is the latent form of the disease and is non-fatal[19]. The microbe preferentially encysts itself within the tissues of the brain and the central nervous system of the host, causing encephalopathy, loss of normal cognitive brain function, impaired judgment, peripheral neuropathy, slowed reflexes, pathological lesions and other disorders classically associated with the invasion of brain tissue [4,5].
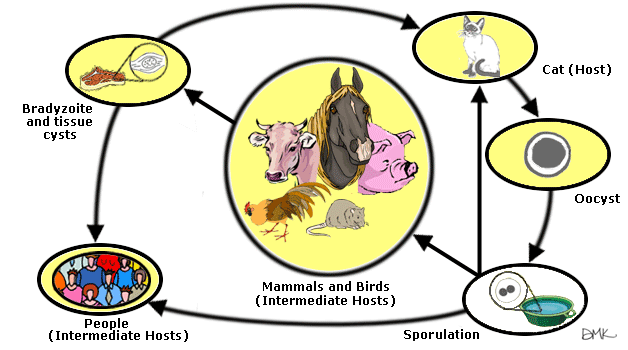
Toxoplasmosis was discovered almost a century ago, in 1908, and the pathogen itself was identified in the North African rodent Ctenodactylus gundi as it was being used for research in the Pasteur Institute in Tunis. The complete lifecycle of T. gondii was not examined in its entirety until 1970. It is shown that cats are definitive hosts to the pathogen, and are excreted as oocytes in their feces, while humans and other mammals, as well as birds, are intermediate hosts. When encysted, the pathogen is very tough and resistant to the low temperature extremes, sustaining viability at temperatures of 0, down to −12˚ C. Harsh agents (corrosives, such as bleach) also have minimal affect on it, as does direct sunlight. High temperatures (approximately 66˚C) can successfully destroy the protozoan, however. [5, 23]
In 1923, the first case of human toxoplasmosis was reported, with the victim being an 11-month-old child[5]. It is universally accepted that this zoonosis occurred much farther back in time, however, and that the parasite originated from animal hosts that were in close contact with humans, most notably cats (even though, as noted above, T. gondii can infect several mammals and birds) [2]. T. gondii mainly spreads via contaminated cat feces, consumption of undercooked meat, tainted water and vertical transmission[5].
Genome structure
The T. gondii haploid genome is approximately 80 Megabases in size, and consists of 11 circular [9] chromosomes, and about 6000 genes, and currently serves as a basis of trying to understand and decipher more complicated areas in the related malarial pathogen, Plasmodium falciparum, among other protozoa. Research is still underway to decipher the functions of all the genes of this microbe, and a variety of techniques have been employed to gather data, notably among them are environmental shotgun sequencing (ESS), expressed sequence tags (EST) and bacterial artificial chromosome (BAC). As of late 2002, the genomic database for T. gondii consists of an excess of 70 million base pairs of nucleotide sequence. As mentioned above, though the function and underpinnings of every gene have yet to be resolved as data is received, the data obtained thus far represents the majority of the coding potential in the T. gondii genome[8]. The primary pool of data accumulation and analysis is neatly assembled into an extensive on-line database made specifically for Toxoplasma gondii [1], and is continually updated as new data is received.
Among the more important plasmids of the pathogen are ones that are associated with lactate dehydrogenase production. This dehydrogenase is expressed by genes LDH1 and LDH2 of T. gondii plasmids, and though the exact function is still under investigation, it was found that –LDH1 and –LDH2 mutants had substantially decreased viability and infectious ability, mostly brought upon by delayed stages of maturation. It was observed that tachyzoites of these mutants failed to form a stable number of tissue cysts in mice hosts, thereby severely diminishing their ability to inflict chronic infection, though this deficiency was not immediately fatal[10].
Like most apicomplexan microbes, Toxoplasma gondii contains a secondary internal symbiotic non-photosynthetic plastid, presumably incorporated by lateral gene transfer from a eukaryotic alga from its phylogenetic evolutionary past. The apicoplast, with 35 kB circular DNA, is a potential target for chemotherapy, hence the interest surrounding it. T. gondii microbes die in the absence of this plastid, and is considered to be vital to the immediate viability of the parasite, since it affects replication of the microbe[11,14].
Though it is not completely understood how T. gondii crosses into the host cell, it is postulated that internal plasmids encode for organelles that provide adhesive protein expression, called micronemes[12] that play a crucial role in host cell-membrane breeching ability. It was discovered that “M2AP-MIC2” complex proteins were responsible for adhesive secretions, and mutants with the respective deficiency had a staggering 80% impairment of host cell entry, as well as severely stunted excretion of the adhesive protein itself that usually occurs when the parasite latches on to the cell in preparation for entry[12, 13].
Cell structure and metabolism
The protozoan has a crescent-shaped cell that is characteristic of it’s species. The conoid end is believed to be central in breeching the host’s cell membrane for entry. The apical site of the microbe is the more pointed part of the cell, and houses the three main secretary organelles whose main function, again, is attachment and infiltration of host cells: the micronemes, rhoptries and dense-granule organelles. It is understood that the micronemes secrete the M2AP-MIC2 complex that not only aid in adhesion and attachment, but also facilitate the motility of the organism, since they have no other detected means of motility (such as cilia, flagella or pili)[15]. The rhoptry proteins are also secreted during the infiltration process, and are observed to reside within a vacuole known as the parasitophorous vacuole. The dense-granule proteins are in charge of altering this vacuole for maximal survival once it enter the host cell, and also assists in the replication process. Lactate dehydrogenase is also thought to be secreted by these organelles, and though the function of this protein has yet to be understood, it has been found to be absolutely vital in the survivability of the parasite once it enter the host [10].
The microbe has a large, central nucleus housing the core of its genome, also contains a single mitochondrion, Golgi body and rough endoplasmic reticulum (ER). T. gondii lacks organelles that produce cholesterol, an essential lipid that the organism cannot live without, and obtains this from the environment of its host by diversion of cholesterol from LDLs (low-density lipoproteins). The ER contains two essential enzymes that participate in this cholesteryl-ester production [18].
Amylopectin, the branched storage polysaccharide common in plants, is present in scattered granules near the apical end of the cell. Due to its branched nature, this carbohydrate is commonly used in plants as an immediate source of energy, as opposed to the harder, straight-chained form, amylose. It is interesting to note that T. gondii harbors all necessary enzymes required for glycolysis within the apicoplast, and this may well be the core metabolic pathway that drives the protozoan’s cellular activity [16].
The pathogen is also thought to make use of the water-soluble form of Vitamin B-9 (folates). Folates have long been understood as essential in the biosynthesis of eukaryotes, and T. gondii has been observed as having very efficient folate transport mechanisms. These active transport processes are a target for new drugs designed to counter the pathogen’s viability [17].
Toxoplasma gondii has three main parts to its complex life cycle. The first two stages, i.e., the tachyzoite (rapidly dividing form upon initial infection) and the bradyzoite (delayed reproduction tissue cysts), have roughly the same cell structure as described above. The sporozoites oocyte stage, however is slightly different in terms of cell structure. This stage can be thought of as a hibernation stage of sorts, with the protozaon now having lost its crescent shape for a softer, more circular and rounder shape. Oocytes form once the parasite has exited its cat host through feces. Usporulated oocytes are undivided, and have a large, central mass known as a sporont. This mass divides into four sporozites once the oocyte becomes sporulated, ready to move on to another host for infection. Sporozoites contain a high number of lipids and amylopectin. This is perhaps directly related to the energy requirement of the microbe in its inactive hibernation state, while bradyozoites have the highest number of micronemes, again, relating directly to organelle requirements during the invasion process.
Ecology
This organism has no known adverse effects on the environment, and does not participate in any biochemical synthesis that actively disrupt or positively contribute to the biosphere. Its main effect is on an individual level as an obligate intracellular a parasite, targeting mainly warm-blooded vertebrates as their hosts. No known symbiosis between T. gondii and other organisms has been observed yet.
Pathology
Toxoplasma gondii causes the disease toxoplasmosis, and is an important human pathogen that is present in up to 1/3rd of the world’s human population [19]. The microbe is usually not a threat to hosts with a competent immune system, and thus a great majority of the microbe is latent within those infected, but nearly 30% of AIDS patients have toxoplasmosis, and is almost always fatal unless continually treated with antibiotics for the rest of their life [15]. It can also be spread via vertical transmission (mother-to-child prenatal transmissions via placenta), and unfortunately, medicines administered to the mother do not assist in protecting or aiding the fetus while still inside the womb.
T. gondii preferentially targets cats, and other members of the feline family, as its definitive hosts, though it has been known to readily invade most warm-blooded vertebrates as "accidental hosts". The parasite forms an extremely tough and resistant oocyte in the intestinal lining of cats, as one of the three stages of its complex life cycle, and exits the host in feces. Via exposure, if the feces is somehow ingested (for e.g., not washing hands after handling cat litter), the protozoan alters its form to a rapidly dividing trachyzoite, and then to a slower dividing bradyzoite that encysts in brain tissue in humans. Since the microbe also encysts in muscle and nervous tissue, another way it can successfully spread is via consumption of undercooked meat. Contaminated drinking water is another possibility for contraction, meaning that the pathogen relies on oral entry via ingestion, and it seems to survive gastric enzymes and very low pH[24].
Short-term effects of toxoplasmosis may include fever, sore throat, headache, and muscle pain, and sometimes, mild forms of lymphadenopathy, and it is easy to confuse these symptoms for the flu, though blindness and retinal infection can also occur. In severely immunocompromised patients, the microbe makes its way to the brain and the nervous tissue. Toxoplasma does not produce any toxins, and its main mode of destruction is rapid trachyzoite multiplication that eventually lyses the host cells. When this happens in the brain and nervous tissue, some more serious and life-threatening symptoms emerge. These include encephalitis, loss of brain function, and in more recent years, schizophrenia has been added to the list.
There are three main strains of T. gondii that have been examined thus far, and have closely approximated lineages not much different from one another. They do, however, differ in virulence. Type I strains are considerably more lethal than Type II or III, and studies on mice have attributed this to a trait locus on chromosome VII, which was responsible for about 50% of the microbe’s virulence.[22] All strains utilize the secretory proteins M2AP-MIC2 complex for attachment and invasion of host cell (as noted in the "Genome" section previously). There are 6 main steps involved in host cell entry: 1) cellular recognition via antigens and surface proteins, 2) motility of parasite by stimulus dependent chemotaxis towards recognized host cell, 3) adhesion and attachment, 4) secretion of rhoptry proteins, 5) penetration of conoid, and finally, 6) parasitophorous vacuole injection[24].
Toxoplasmosis is detected via standard ELISA-based antigen test, blood tests, and occasionally, tissue sampling[21].There is no cure for toxoplasmosis, though treatment is possible. For AIDS patients, lifelong treatments involving antibiotic agents, such as sulfadiazine, clindamycin, spiramycin, and atovaquone have proven to enhance life expectancy significantly[25]. Prevention in the first place is always a better strategy than a cure later on, and Toxoplasma infections can be easily avoided by maintaining a high level of hygiene, especially around cats, regular washing of hands before meals, avoiding contaminated water, thoroughly cooking meat, and limited feline contact during pregnancy.
Application to Biotechnology
It is understood that T. gondii invasion triggers an immune response which prevents reinfection of the parasite, and on this very basis, the protozoan could prove to be a good model for vaccine action against other parasites. Thus far, antigens produced by the ESA (excreted-secreted antigens) from the dense granules of the microbe are of interest to biomedical research, as they provide a high degree of protection, making them ideal for the basis of vaccine construction that could prevented related parasites from infecting humans [26].
Current Research
Single-Most Deadly Gene in T. gondii Isolated:
In a recent report released in December 2006 by the Washington University School of Medicine, scientists believe that they have found a single gene in T. gondii that could may well be responsible for a staggering 90% of the pathogen’s entire virulence. This is a bold statement, considering the protozoan has nearly 6000 genes. The isolated gene is called ROP18, and encodes a kinase protein. Kinases are widely found in the human body, and active in certain cancers and immune malfunctions of the body. ROP18, once having infected the host cell, is thought to block certain important signaling functions of the host cell, as it preps an environment suitable for the pathogen’s survival. To pinpoint a single gene with so much influence on a microbe’s pathogenicity is an exciting find, since the focus on antibiotic development can been narrowed down drastically, and perhaps related pathogens within the same genus and family could also share such a trait[27, 28]
Atomic Structure of Key Invasive Protein of T. gondii Determined:
It is widely accepted that T. gondii uses a variety of proteins to successfully attach and invade its host cells, but scientists from Europe recently determined (2006) the structure of a key microneme protein called TgMIC1. This protein was discovered to specifically target sialylated oligosaccharadies (sialylated sugars contain sialic acid as part of their structure) on the host cell surface for adhesion, removing previous misconceptions of its function [29]. This level of detail in understanding parasite adhesion is ground-breaking, and again, much like the pin-pointing of the ROP18 gene, can help focus antibiotic research in the right direction.
T. gondii Strains Lacking Oral Tranmission Ability Discovered:
Work done by Fux, Nawas et. al. published in 2007 shows that two exotic strains of the microbe called “FOU” and “MAS” were unable to transmit effectively to their hosts through oral means. In culture, these strains were less likely to develop into bradyzoites, and harbored significantly less resistant to the protein-digesting gastric enzyme pepsin. Two other rare strains, “CAST” and “GPHT” also had diminished virility, and had lowered oral transmission as a byproduct of failure to form cysts [30]. These findings are important, since a large portion of Toxoplasma infections are orally induced, investigation of the causative agents of the decreased survival of these strains could be incorporated in biotechnological solutions to weaken the stronger strains prevalent in the population today.
References
((IMG1) Image: [microscopy] http://www.dpd.cdc.gov/dpdx/HTML/ImageLibrary/Toxoplasmosis_il.htm
(IMG2) Image: [life cycle] http://cal.vet.upenn.edu/parasitc/tox_site/index.htm
(1) NCBI: Toxoplasma gondii, Accessed August 15, 2007.
<http://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&id=5811&lvl=3&p=mapview&p=has_linkout&p=blast_url&p=genome_blast&lin=f&keep=1&srchmode=1&unlock>
(2) Toxoplasma in the Norway Rat, The Journal of Parasitology, Vol. 38, No. 3, pp. 226-229 doi:10.2307/3274038Don E. Eyles, June 3rd 1952.
(3) Toxoplasmosis, Sergio Stagno, The American Journal of Nursing, Vol. 80, No. 4. (Apr., 1980), pp. 720-722.
(4) Human Toxoplasmosis: Occurrence in Infants as an Encephalomyelitis Verification by Transmission to Animals , Science, New Series, Abner Wolf; David Cowen; Beryl Paige, Vol. 89, No. 2306. (Mar. 10, 1939), pp. 226-227.
(5) Fetal Encephalomyelitis: Prenatal Inception of Infantile Toxoplasmosis, Abner Wolf; David Cowen; Beryl H. Paige, Science, New Series, Vol. 93, No. 2423. (Jun. 6, 1941), pp. 548-549.
(6) http://www.toxodb.org/toxo/home.jsp (please note: “ToxoDB” is part of an NIH/NIAID funded Bioinformatics Resource Center to provide Apicomplexan Database Resources.)
(7) Primary and Secondary Toxoplasma gondii Infection in Normal and Feline Immunodeficiency Virus-Infected Cats Michael R. Lappin, Jeanne W. George, Niels C. Pedersen, Jeffrey E. Barlough, Christopher J. Murphy, Lawrence S. Morse The Journal of Parasitology, Vol. 82, No. 5 (Oct., 1996), pp. 733-742 doi:10.2307/3283884
(8) ToxoDB: Accessing the Toxoplasma gondii Genome Jessica C. Kissinger, Bindu Gajria, Li Li, Ian T. Paulsen, and David S. Roos Department of Genetics/Center for Tropical and Emerging Global Diseases, University of Georgia, Athens, GA 30602-2606, USA 1Department of Biology, University of Pennsylvania, Philadelphia, PA 19104, USA The Institute for Genomic Research, Rockville, MD 20850, USA http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=165519
(9) The Plastid of Toxoplasma gondii is Divided by Association with the Centrosomes. Striepen B, Crawford MJ, Shaw MK, Tilney LG, Seeber F, Roos DS., The Journal of Cell Biol. 2000 Dec 25;151(7):1423-34Department of Cellular Biology and Center for Tropical and Emerging Global Diseases, University of Georgia, , USA
(10) The Expression of Lactate Dehydrogenase is Important for the Cell Cycle of Toxoplasma gondii, The Journal of Biological Chemistry. 2004 Dec 10;279(50):52300-11. Epub 2004 Sep 30. Al-Anouti F, Tomavo S, Parmley S, Ananvoranich S.
(11) A Plastid Segregation Defect in the Protozoan Parasite Toxoplasma gondii., The EMBO Journal 2001 Feb 1;20(3):330-9. He CY, Shaw MK, Pletcher CH, Striepen B, Tilney LG, Roos DS.
(12) Rapid Invasion of Host Cells by Toxoplasma Requires Secretion of the MIC2–M2AP Adhesive Protein Complex, The EMBO Journal, My-Hang Huynh, Karen E. Rabenau, Jill M. Harper, Wandy L. Beatty, L.David Sibley, and Vern B. Carruthers Received January 10, 2003; Revised March 6, 2003; Accepted March 13, 2003.
(13) Armed and Dangerous: Toxoplasma gondii uses an Arsenal of Secretory Proteins to Infect Host Cells, Parasitology International, PMID: 11269320 [PubMed - indexed for MEDLINE], Carruthers VB.
(14) Ultrastructure of a Toxoplasma gondii Tachyzoite, Expert Reviews in Molecular Medicine, James W. Ajioka, Jennifer M. Fitzpatrick and Christopher P. Reitter, (01)00221-6h.htm (shortcode: fig001jac); 6 January 2001.
(15) Structures of Toxoplasma gondii Tachyzoites, Bradyzoites, and Sporozoites and Biology and Development of Tissue Cysts , Clinical Microbiology Reviews, J. P. Dubey,D. S. Lindsay,and C. A. SpeerApril 1998, p. 267-299, Vol. 11, No. 2 0893-8512/98.
(16) Carbohydrate Metabolism in the Toxoplasma gondii Apicoplast, PUBMED: 17449654, Eukaryot Cell. Aprile 20th 2007.
(17) Toxoplasma gondii is Capable of Eexogenous Folate Ttransport. A Likely Expansion of the BT1 family of Transmembrane Proteins. Mol Biochem Parasitol.;144 (1):44-54 16159678 Kristen M Massimine, Lanxuan T Doan , et al., November 2005.
(18) Host Cell Lipids Control Cholesteryl Ester Synthesis and Storage in Intracellular Toxoplasma, Cell Microbiology, Yoshifumi Nishikawa , Friederike Quittnat , et. al;7 (6):849-67 15888087, June 2005.
(19) "Toxoplasmosis". Lancet 363 (9425):. Montoya J, Liesenfeld O (2004). PMID 15194258, 1965-76.
(20) Toxoplasma gondii Cellular Invasion, Parassitologia. Bonhomme A, Pingret L, Pinon JM.;34(1-3):31-43 PMID: 1339976 [PubMed - indexed for MEDLINE], Dcember 1992.
(21) http://www.cdc.gov/NCIDOD/dpd/parasites/toxoplasmosis/factsht_toxoplasmosis.htm
(22) Identification of Quantitative Trait Loci Controlling, Acute Virulence in Toxoplasma gondii, JSTOR Chunlei Su; Daniel K. Howe; J.P. Dubey; James W. Ajioka;L. David Sibey, Vol. 99, No. 16, pp. 10753-10758.(Aug. 6, 2002).
(23) Survival of Toxoplasma gondii Tissue Cysts in 0.85-6% NaCl Solutions at 4-20 C, The Journal of Parasitology, pp. 946-949, J. P. Dubey, Vol. 83, No. 5.Oct., 1997.
(24) Potential Contamination of Drinking Water with Toxoplasma gondii Oocysts, Epidemiology and Infection, pp. 305-315., J. J. Aramini; C. Stephen; J. P. Dubey; C. Engelstoft; H. Schwantje; C. S. Ribble, Vol. 122, No. 2. (Apr., 1999)
(25) Efficacy of Atovaquone Combined with Clindamycin against Murine Infection with a Cystogenic (Me49) strain of Toxoplasma gondii., J Antimicrob Chemother, 50 (6): 981-7. DOI:10.1093/jac/dkf251. PMID 12461021, Djurković-Djaković O, Milenković V, Nikolić A, Bobić B, Grujić J (2002).
(26) Study of the Secretion of Toxoplasma gondii Dense Granule Antigens, Application in the Production of a Vaccine against Intracellular Parasites, Prof. Andri Capron, nstitut Pasteur de Lille Toxoplasmosis Groupe INSERM Unite 415 RUE DU PROFESSEUR CALMETTE 1, France.
(27) One Gene 90 Percent Responsible for making Common Parasite Dangerous, Washington University School of Medicine in St. Louis, Michael C. Purdy, 14th December 2006.
(28) The Toxoplasma Kinase ROP18: An Active Member of a Degenerate Family, PLoS Pathog. 2007 February; 3(2): e16. Anthony P Sinai,. doi: 10.1371/journal.ppat.0030016, Published online 2007 February 16.
(29) Atomic Resolution Insight into Host Cell Recognition by Toxoplasma gondii, The EMBO Journal (2007), Tharin M A Blumenschein, Nikolas Friedrich, Robert A Childs, Savvas Saouros, Elisabeth P Carpenter, Maria A Campanero-Rhodes, Peter Simpson, Wengang Chai, Theodoros Koutroukides, Michael J Blackman, Ten Feizi, Dominique Soldati-Favre and Stephen Matthews 26, 2808–2820, doi:10.1038/sj.emboj.7601704, Published online 10 May 2007.
(30) Toxoplasma gondii Strains Lacking Oral Transmission are Defective in Stage Differentiation, ,Infection and Immunity, Blima Fux, Julie Nawas, Asis Khan, Darcy Gill, Chuneli Su, and L David Sibley, March 2007.
Edited by Shumyyal Akhtar Malik