Nematocida parisii
Introduction to Microsporidia
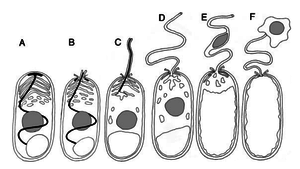
A) Shows a dormant spore containing a polar filament (black), nucleus (gray), polaroplast and posterior vacuole.
B) The posterior vacuole swells with water and ruptures the anchoring disk, allowing the polar filament to emerge through through spore cell wall.
C) Polar filament continues to to outward and evert.
D) Polar filament is fully everted and becomes a polar tube. Sporoplasm is squeezed into the polar tube.
E) Sporoplasm is moving through the polar tube.
F) Entire sporoplasm emerges from the polar tube while bound to the new membrane
Image source: http://www.annualreviews.org/doi/pdf/10.1146/annurev.micro.56.012302.160854 [1]
Microsporidia are a phylum of eukaryotic, intracellular, spore-forming parasites [1]. Fungi are currently considered the closest relative to this phylum, but it is still being debated whether or not they should be cateogorized as Fungi [1,2, 3]. Microsporidia are able to use vertebrates as well as invertebrates as hosts. They are most prevalent in arthropods and fish [1]. The life cycles of microsporidia vary between each species, but all species have at least one intracellular and extracellular spore stage [3]. Microsporidian spores range in size from 1 m to 40 m and also vary in shape from sphericial to rod-shaped to cresant-shaped [1]. However, a majority of the species has an ovoid spore. Microsporidia are able to survive outside of their hosts as spores [6]. Microsporidian spores are characterized by its thick chitinous cell wall surround the cell membrane and the presence of a polar filament inside the spore [3]. Polar filaments are tubes located within the spore, attached to the apex of the spore by an anchoring disk [1]. These polar filaments are an essential component to allow for spore germination and infection of the host cell.
Spore germination in microsporidian spores occurs in several phases: activation, increase in intrasporal osmotic pressure, eversion of the polar tube, and passage of sporoplasm through the polar tube [2]. Microsporidia has to be activated by an environmental trigger such as change in pH, presence of ions, or ultraviolet radiation exposure. Since microsporidia live in a wide range of hosts, each species requires a different activation condition. In response to the activation, all microsporidia respond by increasing flow of water into spore, increasing intrasporal osmotic pressure and swelling the polaroplast [2]. The pressure build-up within the spore becomes the driving force of germination as it forces the eversion of the polar tube [1]. In eversion, the discharging polar filament breaks through the thinnest portion of the spore cell wall (Figure 1B), where it serves as a polar tube (Figure 1D). The polar tube is used as a bridge to siphon the sporoplasm out of the spore and into its host cell [2]. However, it has yet to be determined how exactly the polar tube or sporoplasm interacts with the host cell’s membrane. It is a predicted possibility that the polar tube launches and pierces the cell membrane of a nearby host cell and injects the sporoplasm, allowing it to avoid all extracellular defenses [1,2,7].
Once the sporoplasm of the microsporidia spore is in the host cell, it replicates in a form without a cell wall called meront [6]. Meronts differentiate and develops into the spore form. Once the meronts complete development, they will begin producing more spores, which will be used to infest other host cells [5].
Microsporidia have mitochondria, which are used for their metabolism. However, they do not have electron transfer chains, oxidative phosphorylation, and the tricarboxylic acid (TCA) cycle [1]. Instead of these pathways, microsporidia break down glucose with glycolysis and followed by substrate-level phosphorylation. The resulting pyruvate from glycolysis is decarboxylated by either pyruvate dehydrogenase complex (PDHC) or the enzyme, pyruvate:ferredoxin oxidoreductase in anaerobic microsporidia [1]. Electrons are removed from pyruvate and placed on ferredoxin, which is later transferred to NADH and then to an organic terminal electron acceptor. The lack of TCA cycle induces a requirement for ATP, so the microsporidia also conduct fermentation to synthesis ATP and also had enzymes found in pentose-phosphate pathway (PPP) [1].
Background on Nematocida parisii
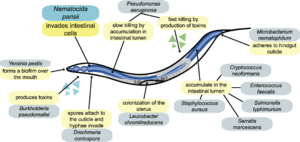
Image source: http://www.plosbiology.org/article/fetchObject.action?uri=info%3Adoi%2F10.1371%2Fjournal.pbio.1000005&representation=PDF [3]
Include some current research in each topic, with at least one figure showing data.
Nematocida parisii Cell Shape, Components, and Metabolism
Cell shape and Metabolism
Include some current research in each topic, with at least one figure showing data.
Current Research on N. parisii
Include some current research in each topic, with at least one figure showing data.
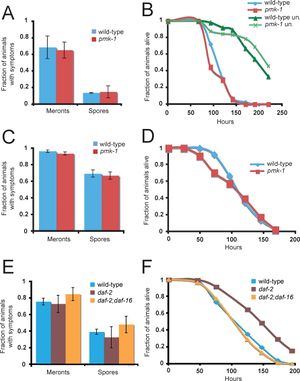
A)
B)
C)
D)
E)
F)
Image Source: http://www.plosbiology.org/article/fetchObject.action?uri=info%3Adoi%2F10.1371%2Fjournal.pbio.0060309&representation=PDF[4]
Conclusion
Overall paper length should be 3,000 words, with at least 3 figures.
References
[8] Moretto, M.M., Khan, I.A., and Weiss, L.M. "Gastrointestinal Cell Mediated Immunity and the Microsporidia". "PLoS Pathogens". 2012. Volume 8 Issue 7. p. 1-4.
Edited by student of Joan Slonczewski for BIOL 238 Microbiology, 2014, Kenyon College.
