Methicillin Resistant Staphylococcus Aureus (MRSA)

Etiology/Bacteriology
Taxonomy
| Domain = Bacteria | Phylum = Firmicutes | Class = Bacilli | Order = Bacillales | Family = Staphylococcaceae | Genus = Staphylococcus | species = S. aureus
|
NCBI: Taxonomy Genome: Staphylococcus aureus |
Description
Staphylococcus aureus is a Gram-positive, non-spore forming, nonmotile, cocci bacterium that colonizes in yellow clusters [8]. This facultative anaerobe is considered natural flora in 20-30% of humans, living in the anterior nares and on the skin and was first isolated in the 1890’s from the pus from a surgical abscess in a knee joint [3] . S. aureus is the most common type of staphylococci to cause infections because of its ability to evade the immune system and many antibiotics. These “Staph infections” can cause bacteremia, endocarditis, soft tissue infections, pneumonia, bone and joint infections, CNS, toxic shock syndrome, and even food poisoning [4]. The most notorious strain of Staphylococcus aureus was identified in the 1960's and is known as the methicillin resistant strain, commonly known for causing mild to severe skin infections resulting in death if not treated promptly [5]. The most common place to contract Methicillin Resistant Staphylococcus aureus (MRSA) infection is typically in a hospital, but community outbreaks during the past decade have been widely observed, leading scientists to distinguish between Hospital-acquired Methicillin Resistant Staphylococcus Aureus (MRSA) and Community-Acquired Methicillin-Resistant Staphylococcus Aureus (CA-MRSA).
Pathogenesis
Transmission
Since S. aureus are a part of the natural flora in many humans’ nose and mouths, especially humans working in hospitals, the transmission of these bacteria can be difficult to prevent. Because the bacterium is nonmotile, S. aureus is most commonly spread through human-to-human contact or through contaminated surfaces/foods. MRSA is typically spread from human-to-human by the hands. In a few cases MRSA can be transmitted through the cough of someone infected with MRSA pneumonia. This bacterium is most commonly transmitted horizontally rather than vertically [4].
Infectious dose, incubation, and colonization
S. aureus naturally colonizes in many parts of the body such as the nose, mouth, mammary glands, hair, upper respiratory tract and more. In most cases, S. aureus colonize these parts of the body harmlessly. These bacteria can survive in a wide range of temperatures ranging from 15-45 degrees Celsius and can also colonize in NaCl concentrations up to 15% [8]. When ingested, the infectious dose of S. aureus is greater than 100,000 organisms, and the incubation period ranges from 30 minutes to 8 hours. The infectious dose and incubation period for other types of S. aureus infections can be hard to determine, as the bacteria can be carried for unknown amounts of time before causing infection, sometimes months [4]. S. aureus can also be carried both chronically and intermittently.
Epidemiology
S. aureus is distributed worldwide and is most often found in hospitals and nursing homes, but community settings are becoming increasingly contaminated. Scientists typically distinguish between CA-MRSA and HA-MRSA when studying epidemiology. Staphylococcus aureus was first isolated by a surgeon in the 1890’s. The first strain of MRSA was observed in 1961, and now approximately 35% of hospital strains of S. aureus are methicillin resistant (isca) In 2002 a new strain of vancomycin-resistant S. aureus was reported in Japan. The first outbreak of CA-MRSA in the United States occurred in 1982 among intravenous drug users. Roughly 24 drug users and 16 nonusers were infected. Since then, CA-MRSA has become a common infection in the United States. Numbers of CA-MRSA infections have spiked in the 2000’s, while HA-MRSA is slightly on the decline [1] [5].
Virulence factors
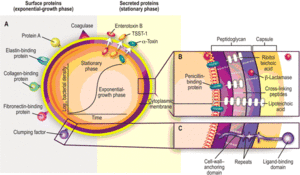
Adherence
S. aureus expresses certain surface proteins that are necessary for binding throughout the body. These surface proteins typically promote attachment to laminin and fibronectin. Most strains also express a clumping factor, coagulase, that promotes attachment to blood clots and traumatized tissue. Fibronectin and fibrinogen-binding proteins are also produced by S. aureus as virulence factors. Mutations of these proteins that have been studied drastically decrease the bacteria's virulence. Adhesins that bind to collagen are significant in infections that cause osteomyelitis. Once the bacteria have adhered, they can secrete a biofilm that make them difficult to eradicate [8].
Invasion
Invasins help to promote bacterial spread within the tissues of the body. Alpha toxin is the most characterized and potent membrane-damaging toxin secreted by S. aureus. It is originally expressed as a monomer that binds to the surface of susceptible cells before becoming oligamerized into a heptomeric ring that causes a pore in the membrane of the attacked cell, which causes the contents of the cell to leak out. Platelets and monocytes are especially susceptible to this toxin. ß-toxin is a sphingomyelinase toxin that damages lipid membranes that are rich in sphingomyelin. It is not often expressed in human isolated strains of the bacteria [8].
A mere 2% of S. aureus isolates express leukocidin, a multicomponent protein that acts to severely damage cell membranes, but 90% of S. aureus strains isolated from severe dermonecrotic lesions are found to express this toxin. This correlation suggests that the toxin is a large component of necrotizing skin infections. Leukocidin forms a hetero-oligameric transmembrane pore made from four LukF and four LukS subunits. This toxin is hemolytic, but not as hemolytic as alpha toxin.
S. aureus also produces a host of proteases, lipases, and DNase, and FAME (fatty acid modifying enzyme). FAME may be important for virulence in abscesses, where it could prolong bacterial survival by modifying anti-bacterial lipids in the cell [8].
Avoidance
To avoid the immune system, S. aureus produces a microcapsule composed of surface polysaccharide that can only be detected by electron microscopy. Strains of the bacteria isolated from infections have been found to produce this capsule in high amounts. This capsule helps the bacteria evade phagocytosis in the absence of complement. Protein A is an Fc receptor on the surface of the pathogen that specifically binds IgG antibody in the wrong orientation. This incorrect orientation of the antibody disrupts opsonization and phagocytosis of the bacteria, allowing it to evade the immune system. Leukocidin also helps in avoidance of the immune system, as acts specifically on phagocytic cells called polymorphonuclear leukocytes [8].
Toxins
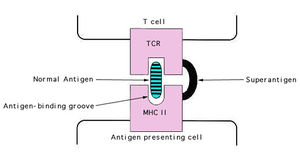
This bacterium produces several toxins. A few are listed above, such as invasion toxins. S. aureus also releases other exotoxins and enterotoxins. Exfoliatoxin comes in two distinct forms (ETA and ETB) and causes scalded skin syndrome in infants. These bacteria secrete two different types of toxins that are considered to have "Superantigen activity," enterotoxins and Toxic Shock Syndrome Toxin (TSST-1). There are six types of enterotoxins (SE-A, B, C, D, E, and G) which cause food poisoning in the host. Enterotoxins B and C are also responsible for 50% of non-menstrual related cases of TSS.
S. aureus enterotoxins and TSS toxins work by stimulating T-cells nonspecifically without normal antigen recognition, causing them to release cytokines in large amounts, which cause the symptoms of TSS. These super antigens bind directly to MHC II receptors rather than in the antigen binding groove of the MHC, as shown on the right [8].
Resistance
Through many generations, Staphylococcus aureus has developed a wide variety of antibiotic resistance. The most commonly known resistance is that of methicillin like antimicrobials. These antimicrobials typically prevent the synthesis of the bacteria's cell wall. The mecA resistance gene in the bacteria's DNA keeps these β-lactam antibiotics from inhibiting the bacteria's transpeptidase enzymes that are critical for cell wall construction. This gene is carried on a mobile Staphylococcal cassette chromosome (SCC). The SCC carries a variety of genes that lend themselves to antibiotic resistance and are usually transmitted both vertically and horizontally across S. aureus strains. Instead of producing normal PBP-2, the enzyme responsible for catalyzing peptidoglycan cross-linking, the bacteria begin to produce PBP-2A. Non-mutated PBP-2 has a affinity for β-lactam rings that PBP-2A does not. Therefore, PBP-2A is not inhibited by the antibiotic [5].

Clinical features
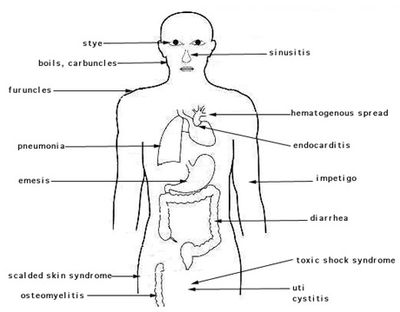
Symptoms and Diagnosis
S. aureus can cause a whole host of different diseases and symptoms depending on where the infection is occurring in the body. The most well known Staph infection is that of the skin, but many other diseases are also caused by Staph infections as well. Diagnosis of the infection depends on where the bacteria has caused infection.
Bacteremia
Bacteremia, also known as "blood poisoning" can occur when bacteria enter into the blood stream and begin to colonize. In some cases, bacteremia is self-limiting and asymptomatic but in other cases is known to lead to septic shock, which is fatal. Bacteremia may be characterized by one of more of the following symptoms: fever, chills, malaise, abdominal pain, nausea, vomiting, diarrhea, anxiety, shortness of breath, and confusion. Bacteremia is diagnosed by culturing blood for bacteria. The blood might also reveal elevated white blood cell levels. [10]
Endocarditis
Endocarditis occurs when the endocardium of the heart muscle is damaged, allowing bacteria in the blood stream to become lodged onto the heart valves or lining and cause infection. Endocarditis is not characterized by a single symptom, but can be detected by many symptoms such as a mild fever, chills, weakness, cough, trouble breathing, headaches, aching joints, and loss of appetite. It also causes heart murmurs and regurgitation in the heart valves. This disease usually affects people between the ages of 15-60. A risk that accompanies endocarditis is the formation of emboli. This occurs when bacteria break off from the site of colonization in clumps and become lodged in blood vessels. This can lead to organ failure as nutrients are blocked from accessing certain areas of the blood stream. Diagnosis of this is usually done by taking a sample of blood from the patient to test it for bacteria. Another way to diagnose Endocarditis is echocardiography, which uses ultrasound waves to make an image of the heart. This allows doctors to check for abnormalities like bacterial vegetation [10].
Soft tissue infections
Staph infections lead to several different kinds of soft tissue infections. Impetigo, abscesses, Necrotizing Fasciitis, erysipelas, and cellulitis are all examples of soft tissue infections caused by S. aureus. Each of these infections are caused by a bacterial invasion of the skin, usually through an open wound. Once invasion has occurred, the staph bacteria colonize the area and produce mild to severe infections. These skin infections can be very contagious and spread quickly to other parts of the body through contact. These infections also spread easily to others as the bacteria are often transferred by the hands to other surfaces. The symptoms of these skin infections vary from type to type, but nearly all of them are characterized by a lesion or bump in the skin that contains pus. The bump then develops a scab like crust that may become itchy. Skin infections can also be characterized by redness, warmth, pain, and swelling (typical signs of inflammation). Though typically not severe, even simple infections can cause major problems in the bones and blood stream if left untreated. Severe infections such as necrotizing fasciitis are fatal if not treated. Most skin infections are acquired through CA-MRSA as opposed to HA-MRSA. Diagnosis of these diseases are done by an observation of symptoms and sometimes laboratory tests [7] [10].
Pneumonia
When S. aureus infects the lungs, the resulting sickness is pneumonia. Pneumonia is the most common fatal infection contracted by those in the hospital. Symptoms of this infection include fever, cough, chest pain, shortness of breath, and increased respirations. Chills accompanying fevers are especially suspicious. Many patients with Pneumonia will also cough up sputum produced by the alveoli. Pneumonia is diagnosed through symptoms and a chest examination. Cultures of sputum and x-ray examinations may also be used to detect infection [10].
Osteomyelitis
Infections of the bones and joints are called Osteomyelitis and are most often caused by bacteria, specifically S. aureus. Symptoms of osteomyelitis are pain in the affected bone, tenderness, fever, and chills. If the infection is caused by the spread of the bacteria from an area of soft tissue infection, the only symptom of note may be slow healing of the wounded area. To diagnose this staph infection, blood samples are collected to check white blood cell levels and then cultured to allow for the growth of bacteria. Bone inflammation is then tested using radioactive elements that show up in X-rays. An MRI may also be used. If a pocket of pus is available, samples are taken and cultured to detect the infectious bacteria [10].
Meningitis
The meninges is the thin, membranous covering of the brain and spinal chord. Infection of this membrane is called meningitis and can be caused by bacteria, including S. aureus but it is rare. Meningitis caused by bacteria is typically the most serious and hard to treat. In many cases, meningitis can be fatal. Common symptoms of bacterial meningitis are fever, headache, vomiting, sensitivity to light, irritability, severe fatigue, stiff neck, and a reddish purple rash on the skin. Seizures, confusion, and an eventual coma are all possibilities if the infection remains untreated. The most reliable way to diagnose meningitis is a lumbar tap, where a sample of spinal fluid is taken from the lower spinal chord. The fluid is then examined under a microscope for signs of bacteria [10].
Toxic Shock Syndrome
Toxic Shock Syndrome is typically caused by a characteristic toxin of a strain of S. aureus. When produced in large quantities, this toxin can enter the blood stream and cause high fever, vomiting, diarrhea, headache, rash, sore throat, and body aches. A drop in blood pressure, kidney failure, and skin peeling on hands and feet may also accompany the later stages of TSS. While menstruating women are most commonly the victims of TSS, the disease can affect anyone of any age, race, and sex, especially those whose immune systems are weakened at any time. TSS can be fatal if not diagnosed. A variety of blood tests and physical examinations are used to diagnose this infection [10]
Morbidity and Mortality
MRSA has been on the decline for many years as health care professionals become more adept at preventing and treating the infections. From 1997 to 2007, MRSA blood infections dropped 50% and invasive infections dropped 28% from 2005-2008. In the United States alone 94,000 cases of MRSA were reported in 2007. Of those 94,000 cases, approximately 18,000 cases were fatal, or 19%. Statistics for MRSA infections can be difficult to acquire, as they can often go undetected or clear up on their own [6] [2].
Treatment
Depending on the severity of the infection, Staph infections, even those caused by MRSA, can be treated outpatient with antibiotics. Severe infections such as TSS and sepsis, however, may require hospitalization, surgery, and antimicrobials. Vancomycin has been the go-to antimicrobial prescribed in the past because of the MRSA's resistance to drugs falling in the methicillin family. Unfortunately, strains of S. aureus resistant to Vancomycin have begun to spread as well. Other barriers to treating MRSA include the bacteria's ability to form biofilms on infected surfaces such as catheters, endotracheal tubes, and other foreign devices. Antimicrobials may be given in topical, oral, and IV form depending upon the severity of the infection. Some soft tissue infections do not require antibiotics, but can simply be drained and packed until the body's immune response is able to kill off the infection [6] [5].
Prevention
Because it naturally colonizes certain areas of the body, preventing Staphylococcus aureus infections can be difficult, and those who serve as a reservoir for the bacteria often have minor reoccurring infections. Regardless, the first step to preventing Staph infection is good hygiene. Washing one's hands properly and often is one way to prevent spreading of the bacteria. Not sharing razors, towels, and other personal items also contribute to keeping the bacteria from spreading. Any wounds should be kept covered and surfaces/equipment that may be infected, especially in places like locker rooms and day care centers, should be cleaned with a disinfectant often [9]
Immune Response
The innate immune response is the leading defense against MRSA infections. Neutrophils are recruited to the site of infection by Pathogen Recognition Receptors binding to the bacteria, causing them to release interleukin-8 (IL-8, CXCL8), GROα (CXCL1), granulocyte chemotactic protein 2 (GCP2, CXCL6), and complement component C5a. These chemokines help to recruit neutrophils to the site of infection in order to assist macrophages in phagocytosis of the bacteria. The triggering of the innate immune response and neutrophil recruitment also serve to trigger complement activation and inflammation, which contribute to the destruction of the pathogens [11] [12]
References
1 Batabyal, B., Kundu, G. and Biswas, S. (2012). Methicillin-Resistant Staphylococcus Aureus: A Brief Review. International Research Journal of Biological Sciences, 1 (7), pp. 65-71.
2 Cdc.gov (2006). CDC - MRSA Treatment | MRSA Infections.
3 Hopkinsguides.com (n.d.). Johns Hopkins Guides: Staphylococcus aureus.
4 Phac-aspc.gc.ca (2011). Staphylococcus aureus - Pathogen Safety Data Sheets - Public Health Agency of Canada.
5 Rybak, M. and Laplante, K. (2005). Community-Associated Methicillin-Resistant Staphylococcus aureus: A Review. Pharmacotherapy, 25 (1), pp. 74-85.
6 Snider, J. and Rivard, B. (2012). Methicillin-resistant Staphylococcus aureus: A Review of Current Antibiotic Therapy. Available through: Michigan Pharmacists
7 Stevens Et. Al. (2011). Practice Guidelines for the Diagnosis and Management of Skin and Soft-Tissue Infections. IDSA guidelines.
8 Textbookofbacteriology.net (2008). Staphylococcus aureus.
9 Unmc.edu (n.d.). Prevention | UNMC.
10 Medical-dictionary.thefreedictionary.com (2013). Medical Dictionary.
11 Deleo, F., Diep, B. and Otto, M. (2010). Host Defense and Pathogenesis in Staphylococcus aureus Infections. National Institute of Health.
12 Gordon, R. and Lowy, F. (2008). Pathogenesis of Methicillin-Resistant Staphylococcus aureus Infection. Infectious Disease Society of America.
Created by Dehra McGuire, student of Tyrrell Conway at the University of Oklahoma.

