Racial Disparities in MRSA Infections: Difference between revisions
| Line 19: | Line 19: | ||
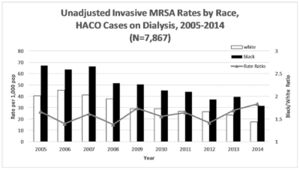
[[Image:MRSADIABETES.jpeg|thumb|300px|left| Figure 3. Unadjusted healthcare-acquired community-onset cases of invasive methicillin-resistant Staphylococcus aureus rates by race for cases among dialysis patients. Figure credits belong to the authors of the cited paper. <ref name=gualandiclass> [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6232852/figure/F1/ Gualandi, Nicole et al. “Racial Disparities in Invasive Methicillin-resistant Staphylococcus aureus Infections, 2005-2014.” Clinical infectious diseases: an official publication of the Infectious Diseases Society of America vol. 67,8 2018: 1175-1181. doi:10.1093/cid/ciy277] </ref> ]] | [[Image:MRSADIABETES.jpeg|thumb|300px|left| Figure 3. Unadjusted healthcare-acquired community-onset cases of invasive methicillin-resistant Staphylococcus aureus rates by race for cases among dialysis patients. Figure credits belong to the authors of the cited paper. <ref name=gualandiclass> [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6232852/figure/F1/ Gualandi, Nicole et al. “Racial Disparities in Invasive Methicillin-resistant Staphylococcus aureus Infections, 2005-2014.” Clinical infectious diseases: an official publication of the Infectious Diseases Society of America vol. 67,8 2018: 1175-1181. doi:10.1093/cid/ciy277] </ref> ]] | ||
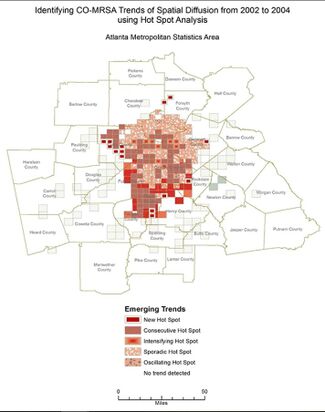
[[Image:atlmrsa.jpeg|thumb| | [[Image:atlmrsa.jpeg|thumb|325px|right| Figure 4. Image depicts the areas with a higher concentration of community-associated MRSA cases within Atlanta during the period of 2002–2004. Figure credits belong to the authors of the cited paper. <ref name=ATLANTARACISM>[http://doi.org/10.5334/egems.308 Ali, F., Immergluck, L. C., Leong, T., Waller, L., Malhotra, K., Jerris, R. C., … Rust, G. S. (2019.) A Spatial Analysis of Health Disparities Associated with Antibiotic Resistant Infections in Children Living in Atlanta (2002–2010). Egems (generating Evidence & Methods to Improve Patient Outcomes), 7(1), 50.]</ref> ]] | ||
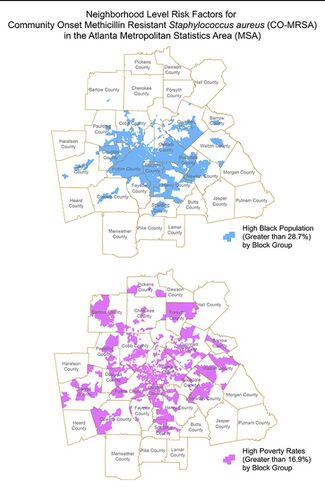
[[Image:atlracism.jpeg|thumb| | [[Image:atlracism.jpeg|thumb|325px|right| Figure 5. Image depicts the areas with a more highly concentrated population of Black people are shown in the map on the top and areas of high poverty concentration during the same time period are shown below. Figure credits belong to the authors of the cited paper. <ref name=ATLANTARACISM/> ]] | ||
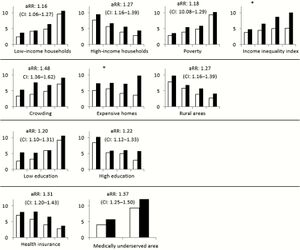
[[Image:sociologicalMRSA.jpeg|thumb|300px|right| Figure 6. MRSA cases by race, stratified by quartiles of US census data pertaining to income, housing, education, and health. Cases are depicted in increasing quartiles of census data, per 100000 persons for white (white bars) and black (black bars) people. Figure credits belong to the authors of the cited paper. <ref name=socio>[https://academic.oup.com/cid/article/64/5/597/3002713?login=true Isaac See, Paul Wesson, Nicole Gualandi, Ghinwa Dumyati, Lee H. Harrison, Lindsey Lesher, Joelle Nadle, Susan Petit, Claire Reisenauer, William Schaffner, Amy Tunali, Yi Mu, Jennifer Ahern "Socioeconomic Factors Explain Racial Disparities in Invasive Community-Associated Methicillin-Resistant Staphylococcus aureus Disease Rates" 2017. Clinical Infectious Diseases, Volume 64, Issue 5, 1 March 2017, Pages 597–604.]</ref> ]] | [[Image:sociologicalMRSA.jpeg|thumb|300px|right| Figure 6. MRSA cases by race, stratified by quartiles of US census data pertaining to income, housing, education, and health. Cases are depicted in increasing quartiles of census data, per 100000 persons for white (white bars) and black (black bars) people. Figure credits belong to the authors of the cited paper. <ref name=socio>[https://academic.oup.com/cid/article/64/5/597/3002713?login=true Isaac See, Paul Wesson, Nicole Gualandi, Ghinwa Dumyati, Lee H. Harrison, Lindsey Lesher, Joelle Nadle, Susan Petit, Claire Reisenauer, William Schaffner, Amy Tunali, Yi Mu, Jennifer Ahern "Socioeconomic Factors Explain Racial Disparities in Invasive Community-Associated Methicillin-Resistant Staphylococcus aureus Disease Rates" 2017. Clinical Infectious Diseases, Volume 64, Issue 5, 1 March 2017, Pages 597–604.]</ref> ]] | ||
Revision as of 19:32, 16 April 2021
MRSA Overview
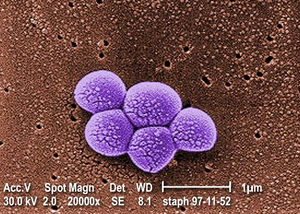
Methicillin-Resistant Staphylococcus aureus, or MRSA for short, is a Gram-positive cocci-shaped (spherical) bacterium that measures approximately 1μm in diameter and forms clusters that are popularly described as being grape-like. [1] S. aureus is present on and within the bodies of many individuals asymptomatically, and as a result of this it often remains unnoticed. According to studies, around 20% of people are persistent nasal carriers of S. aureus and around 30% are intermittent carriers, with the remaining 50% not carrying the bacterium. [1] Other than within the nose, S. aureus can be commonly seen present on the skin, skin glands, guts, and a variety of mucous membranes. This presence within the body is referred to as colonization, and it significantly increases the chances of acquiring an infection by providing a reservoir of the pathogen. [1]
In most cases, the previously asymptomatic, commensal S. aureus that previously colonized different sections of individuals' microbiomes are responsible for their infection. [2] Within the world of public health and medicine, a hugely important factor associated with S. aureus is its significant level of acquisition of resistance against multiple antibiotic classes, which greatly complicates efforts to treat it clinically. [3] Methicilin is an antibiotic class of particular interest with regard to resistance acquisition among S. aureus. According to the latest CDC data released in 2019, Methicillin-Resistant Staphylococcus aureus and other, less prominent strains of S. aureus accounted for an estimated 119,247 bloodstream infections within the United States in 2017, while causing 19,832 of those infected to pass away from complications associated with infection. [4]
As shown in the number of cases within 2017 alone given above, infections due to MRSA are a widespread phenomenon, however, there are stark differences in the incidence rates between different races in the US, particularly amongst Black and White people. [5] Data analysis suggests that these disparities are the result of the combined effects of socioeconomic inequalities, socio-environmental inequalities, and broader disparities in the overall prevalence of certain health factors between races, for instance, diabetes. [5] [6]
Health Consequences of MRSA
MRSA is responsible for causing a wide array of negative health outcomes. Among these outcomes are the likes of sepsis syndrome, toxic shock, skin and soft tissue infections (SSTIs),
bacteremia and endocarditis, pneumonia, bone and joint infections, CNS disease, and bacteremia. [8] Of note, bacteremia is present in roughly 75% of invasive MRSA cases. [8] These varied infections can result in health outcomes ranging from rashes to organ failure and, of course, death. [1] When considering the often dire health consequences associated with invasive MRSA infections, it becomes clear to see why the stark disparities in the rate of infection between races are a topic of burgeoning importance within the medical community. [9]
Hospital and Healthcare Acquired MRSA Disparities
Hospital Acquired MRSA Disparities
Methicillin-Resistant Staphylococcus aureus has been one of the most well-known and clinically relevant pathogens within hospitals and the broader healthcare industry for decades, and it has remained a constant source of infection within them, though its frequency has oscillated somewhat over time. [10] Hospital-acquired MRSA is typically acquired via nosocomial infection, often as a result of a surgical or invasive medical procedure during a hospital stay. [11]
MRSA's immense clinical relevance is why the study of its racial health disparities are a topic of interest amongst both sociologists and scientists alike, and given such there have been studies and analyses conducted to sparse out just how significant these racial disparities with regard to MRSA infection are, and to what degree said racial disparities exist in broader medical settings. According to a ten-year study conducted in the states of California, Colorado, Connecticut, Georgia, Maryland, Minnesota, New York, Oregon, and Tennessee, incident rates for hospital-associated MRSA infection decreased by over 65% for Black and White people from 2005 through 2013, however, the rate of decrease varied between Black and White patients. [5] Amongst Black people, incidence rates declined from 18.16 cases per 100,000 Black people to 6.21 cases per 100,000 Black people. In White people, there was a decline from 8.46 cases per 100,000 to 2.94 per 100,000. [5] These changes represent a small shift in the unadjusted rate ratio of the Black community's MRSA prevalence of infection from 2.15 in 2005 to 2.11 in 2013. This adjusted rate ratio conveys that in both 2005 and 2013 Black people were a little more than twice as likely to be infected by Hospital Acquired MRSA compared to White people. [5] When adjusting the rate ratio to account for age, sex, and year, this rate ratio climbs to 3.2, meaning that when observing Black and White people of the same age and sex within the same year, Black people were 3.2x more likely to suffer infection. [5] These results showcase that even though the raw number of cases throughout these 9 states decreased in excess of 65% over the course of the 8-year long observational period, the rate of infection amongst Black people proved to remain significantly higher than that of White people. [5]
When considering the wide array of negative health consequences that can manifest as a result of MRSA infection, incidence rates being three times higher for Black people than those of white people demonstrates a clear racial disparity. A commonly proposed reason for this observed disparity is the fact that on average, Black people within the US face heavily burdening socioeconomic challenges to a degree that White people do not. Within the United States, it has been discerned that Black people who are socioeconomically challenged, particularly those below the poverty line, as well as those living in urban areas, face many persistent challenges in gaining access to quality health care centers where they will not be discriminated against. [12]
Healthcare Acquired MRSA Disparities
Healthcare-acquired MRSA develops outside of hospitals while former patients are in the community and have recently been involved with a health care facility, be it receiving treatment at one or simply being inside of one for a prolonged period of time. [11] For reference, health care facilities include the likes of inpatient hospitals, ambulatory surgical centers, dialysis centers, and nursing facilities. [11] In the same 9 state study conducted to measure the rate change in Hospital-acquired MRSA along racial lines Black people were found to possess a rate ratio of 3.84 when accounting for age, sex, and year. [5] This adjusted rate ratio once again demonstrates a rate of MRSA acquisition among Black people that failed to fall in proportion to the smaller number of overall cases of MRSA acquisition over time, remaining roughly the same instead. This also plainly demonstrates once again that Black people have higher incidence rates of MRSA infection than White people, 3.84x higher in this case. [5] Similarly to instances of Hospital-acquired MRSA, both socioeconomic factors and the issue of inadequate access to quality healthcare are the reasoning for such disparities. [12]
These overall disparities in access to adequate healthcare are made more severe by the fact that those with preexisting medical conditions are at higher risk to contract MRSA, and also more likely to die from it. [6] Diabetes and kidney disease are examples of this trend that are well suited to demonstrate the ways with which medical conditions can heighten incidence rates of MRSA through racial lines. According to the CDC's 2020 National Diabetes Statistics Report, the prevalence of diagnosed diabetes is 56% greater in Black people than in White people. [13] Similarly, the prevalence of undiagnosed diabetes is 32.5% greater in Black people than White people. [13] Diabetes necessitates more numerous hospital and healthcare facility visits, particularly visits to dialysis centers among those with severe diabetes and/or kidney disease. [5] As can be seen in Figure 3, the rates of MRSA incidence amongst Black and White people are clearly disparate, with Black people being infected at a significantly higher rate [5] This example of diabetic dialysis patients showcases the ways with which pre-existing medical conditions contribute to racial disparities in the rate of MRSA infection, however, it also highlights the effects of disparities in access to high-quality healthcare facilities, in this case, dialysis centers. [14] These disparities in access to high-quality healthcare facilities are well documented, with those living in predominantly Black neighborhoods being 53% less likely to receive treatment at high-quality dialysis centers, despite often living closer to them than White people. [14] This is the result of both socioeconomic and institutional barriers faced by Black people, such as not being able to afford high-quality insurance/healthcare providers and dialysis centers possessing the ability to outright deny Black patients at their own discretion. [14] Poverty and limited access to transport are additional factors, which all go to show the interconnectedness of these contributors to MRSA-related health disparities. [14] [5] Socioeconomic and institutional factors contribute to Black people's predisposition towards acquiring certain medical conditions at higher rates, which in turn places them in situations where they are at risk of MRSA infection more frequently. [14] [5] Both because of this heightened visitation and limited access to high-quality care, Black people are then more likely to suffer MRSA infections and to die from them. [14] [5]
Community-Acquired MRSA Disparities
For the majority of its history, MRSA was considered to be a nosocomial pathogen, however, in the late 90s MRSA appeared within communities independent of any connections to particular hospitals or health care settings. [6] This was the birth of community-acquired MRSA in the United States, which now has incidence rates exceeding those seen in hospital-acquisition cases. [15] [6] A large proportion of community-acquired MRSA cases are invasive in nature, meaning that the MRSA infection happens beyond the skin (i.e. within the bloodstream or organs). According to estimates from 2014, there are roughly 15,000 instances of invasive community-acquired MRSA per year, in addition to roughly 1000 deaths as a result. [15] [6] These numbers show how detrimental invasive community-acquired MRSA is to people's health, as it kills a sizable amount of those who are infected by it each year. Incidence rates for invasive community-acquired MRSA infections are significantly higher amongst Black people than they are White people. [6] Furthermore, when adjusting for age, sex, and year, Black people are 2.78 times more likely to have an incident of community-acquired MRSA infection. [5] This data plainly demonstrates that there are clear disparities in the number of incidents of community-acquired MRSA infection for Black and White people, with Black people once again suffering from higher rates of infection, hospitalization, and death. [6] [5]
An Atlanta study ranging from 2002 to 2010 reveals further details about the nature of these disparities in community-acquired MRSA infection rates. The similarities in the highlighted areas in Figures 4 and 5 of Atlanta should be noted, as they visually demonstrate the fact that Black people are poor at disproportionate rates, while also living in more densely packed, crowded communities as a result of this poverty and urbanicity.[7] Due to these factors, Black people are exposed to community-acquired MRSA at disproportionately high rates, which then leads to higher rates of MRSA infection. [7] When compared to White people, Black people were 87% more likely to suffer from a community-acquired MRSA infection in this study. [7] Both this data and the previous data are in line with the trends seen in hospital and healthcare-acquired MRSA, demonstrating that there are racial disparities in incidence rates of MRSA infection across the board.
Conclusion
According to numerous studies, as well as data from organizations such as the CDC, there are clearly defined racial disparities in the incidence rates of MRSA infection in America. [15] [6] [7] In the cases of Hospital and Healthcare-acquired MRSA, these disparities have persisted despite overall declines in the number of MRSA cases nationally. [9] [7] [5] This signals that while efforts are being made to combat MRSA's prevalence in healthcare settings, nothing is being done to address the factors contributing to the disparities faced by Black Americans. [6] [7] In Figure 6, census data showing MRSA incidence of Black and White people of similar socioeconomic status (for specific categories) shows that when socioeconomic conditions are the same, the disparities between Black and White MRSA incidence rates are generally less drastic. [6] This lends credence to the notion that socioeconomic factors associated with race are a driving force between these inequities. [6] [16] Despite this, though, Black incidence rates for MRSA remain higher than those of White people even with similar socioeconomic factors at play. [6] A reason for this is that within the US, Black people that lack societal power and influence, live in urban areas and/or are below the poverty level in the USA are constantly challenged to access quality health care—free of discrimination. [16] Furthermore, these socioeconomic disadvantages and issues of inadequate healthcare access contribute to Black people having certain diseases and sicknesses at higher rates, which we now know places them at higher risk of MRSA infection as well. <ref name=ballukarirace> [13] In order for these racial disparities in incidence rates of MRSA infection to be quelled, there must be serious work done to ameliorate the pre-existing socio-economic disadvantages faced by Black people, as well as comprehensive reform to healthcare facilities such to eliminate the racial discrimination against that contributes to lower access to high-quality healthcare.
References
- ↑ 1.0 1.1 1.2 1.3 Lakhundi, S., & Zhang, K. https://doi.org/10.1128/CMR.00020-18 "Methicillin-Resistant Staphylococcus aureus: Molecular Characterization, Evolution, and Epidemiology." 2018. Clinical microbiology reviews, 31(4), e00020-18.
- ↑ Rhonda Griffiths, Ritin Fernandez, Elizabeth Halcomb "Reservoirs of MRSA in the acute hospital setting: A systematic review" 2002. Contemporary Nurse, 13:1, 38-49
- ↑ Ventola, C Lee. “The antibiotic resistance crisis: part 1: causes and threats.” 2015 P & T : a peer-reviewed journal for formulary management vol. 40,4: 277-83.
- ↑ Kourtis AP, Hatfield K, Baggs J, et al. "Vital Signs: Epidemiology and Recent Trends in Methicillin-Resistant and in Methicillin-Susceptible Staphylococcus aureus Bloodstream Infections — United States." 2019 MMWR Morb Mortal Wkly Rep 2019;68:214–219.
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 Gualandi, Nicole et al. “Racial Disparities in Invasive Methicillin-resistant Staphylococcus aureus Infections, 2005-2014.” Clinical infectious diseases: an official publication of the Infectious Diseases Society of America vol. 67,8 2018: 1175-1181. doi:10.1093/cid/ciy277
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 Isaac See, Paul Wesson, Nicole Gualandi, Ghinwa Dumyati, Lee H. Harrison, Lindsey Lesher, Joelle Nadle, Susan Petit, Claire Reisenauer, William Schaffner, Amy Tunali, Yi Mu, Jennifer Ahern "Socioeconomic Factors Explain Racial Disparities in Invasive Community-Associated Methicillin-Resistant Staphylococcus aureus Disease Rates" 2017. Clinical Infectious Diseases, Volume 64, Issue 5, 1 March 2017, Pages 597–604.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 7.7 Ali, F., Immergluck, L. C., Leong, T., Waller, L., Malhotra, K., Jerris, R. C., … Rust, G. S. (2019.) A Spatial Analysis of Health Disparities Associated with Antibiotic Resistant Infections in Children Living in Atlanta (2002–2010). Egems (generating Evidence & Methods to Improve Patient Outcomes), 7(1), 50.
- ↑ 8.0 8.1 Liu C, Bayer A, Cosgrove SE, Daum RS, Fridkin SK, Gorwitz RJ, Kaplan SL, Karchmer AW, Levine DP, Murray BE, J Rybak M, Talan DA, Chambers HF; Infectious Diseases Society of America. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011 Feb 1;52(3):e18-55.
- ↑ 9.0 9.1 Bakullari A, Metersky ML, Wang Y, Eldridge N, Eckenrode S, Pandolfi MM, Jaser L, Galusha D, Moy E. Racial and ethnic disparities in healthcare-associated infections in the United States, 2009-2011. Infect Control Hosp Epidemiol. 2014 Oct;35 Suppl 3:S10-6.
- ↑ Dantes R, Mu Y, Belflower R, et al. National Burden of Invasive Methicillin-Resistant Staphylococcus aureus Infections, United States, 2011. JAMA Intern Med. 2013;173(21):1970–1978.
- ↑ 11.0 11.1 11.2 Sutton JP, Steiner CA. Hospital-, Health Care-, and Community-Acquired MRSA: Estimates From California Hospitals, 2013: Statistical Brief #212. 2016 Oct. In: Healthcare Cost and Utilization Project (HCUP) Statistical Briefs [Internet. Rockville (MD): Agency for Healthcare Research and Quality (US)]
- ↑ 12.0 12.1 D.‐L., Commodore‐Mensah, Y., Alexander, K.A., Jacques, K., Wilson, P.R., Akomah, J., Sharps, P. and Cooper, L.A. (2020), COVID‐19: Shedding light on racial and health inequities in the USA. J Clin Nurs, 29: 2734-2736.
- ↑ 13.0 13.1 13.2 Centers for Disease Control and Prevention. "National diabetes statistics report, 2020." Atlanta, GA: Centers for Disease Control and Prevention, US Department of Health and Human Services (2020): 12-15.
- ↑ 14.0 14.1 14.2 14.3 14.4 14.5 Saunders, M. R., Lee, H., Maene, C., Schuble, T., & Cagney, K. A. (2014). Proximity Does Not Equal Access: Racial Disparities in Access to High Quality Dialysis Facilities. Journal of racial and ethnic health disparities, 1(4), 291–299.
- ↑ 15.0 15.1 15.2 Centers for Disease Control and Prevention. ABCs report: methicillin-resistant Staphylococcus aureus, 2014.
- ↑ 16.0 16.1 D.‐L., Commodore‐Mensah, Y., Alexander, K.A., Jacques, K., Wilson, P.R., Akomah, J., Sharps, P. and Cooper, L.A. (2020), COVID‐19: Shedding light on racial and health inequities in the USA. J Clin Nurs, 29: 2734-2736.
