Bordetella pertussis and the Importance of Vaccination: Difference between revisions
| (21 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
<!-- Do not edit this line-->{{Curated}} | <!-- Do not edit this line-->{{Curated}} | ||
==Introduction and History== | ==Introduction and History== | ||
[[Image:disease_pertussis.jpg|thumb| | [[Image:disease_pertussis.jpg|thumb|315px|right|This image is a photomicrograph image of <i>B. pertussis</i>. This image belongs to the CDC (PHIL Photo ID# 2121) [https://www.cdc.gov/pertussis/about/photos.html]] | ||
By Alexandra White<br> | |||
Vaccination is a widely used practice to help prevent infectious disease and commonly spread illnesses. A widely known and common vaccine is used to prevent infection of <i>Bordetella pertussis</i>, the causative agent of Whooping cough. Whooping cough is known as one of the most common death causing infectious disease in the world<ref name=Kerr>[https://link.springer.com/article/10.1007/s100960050435/ Kerr, J.R. and Matthews, R.C."<i>Bordetella pertussis</i> Infection: Pathogenesis, Diagnosis, Management, and the Role of Protective Immunity." 2000. European Journal of Clinical Microbiology and Infectious Disease 19:77-88.]</ref>. The disease results in over 50 million cases worldwide per year, with the majority being unvaccinated individuals present in Third World Countries<ref name=Kerr></ref>. | |||
<i>B. pertussis</i> is spread through coughing and sneezing and symptoms first appear seven to ten days after infection<ref name=WHO>[https://www.who.int/health-topics/pertussis#tab=tab_1/ World Health Organization: Pertussis 2018]</ref>. These symptoms include: fever, runny nose, coughing which develops into a whooping cough, and phenomena<ref name=WHO></ref>. Those infected with <i>B. pertussis</i> are contagious for around three weeks once coughing is displayed as a symptom yet symptoms can last up to eight weeks<ref name=WHO></ref>. Most commonly infants under two months are the most vulnerable and have the highest mortality rate<ref name=Kapil>[https://www.sciencedirect.com/science/article/pii/S0952791518301237#bib0025/ Kapil, P. Merkel, T.J. "Pertussis vaccines and protective immunity" Current Opinion in Immunology. 59:72-78]</ref>. In infants symptoms include: gagging, gasping, bradycardia, cyanosis, and vomiting<ref name=Cherry2>[https://academic.oup.com/cid/article/63/suppl_4/S119/2526416?login=true/ "Pertussis in Young Infants Throughout the World " Clinical Infectious Diseases. 63:S119-S122]</ref>. Mortality in infants most commonly is from leukocytosis, pulmonary hypertension, and pneumonia<ref name=Cherry2></ref>. Treatment for whooping cough consists of antibiotic therapy and intubation with ventilation and oxygen<ref name=Cherry2></ref>. | <i>B. pertussis</i> is spread through coughing and sneezing and symptoms first appear seven to ten days after infection<ref name=WHO>[https://www.who.int/health-topics/pertussis#tab=tab_1/ World Health Organization: Pertussis 2018]</ref>. These symptoms include: fever, runny nose, coughing which develops into a whooping cough, and phenomena<ref name=WHO></ref>. Those infected with <i>B. pertussis</i> are contagious for around three weeks once coughing is displayed as a symptom yet symptoms can last up to eight weeks<ref name=WHO></ref>. Most commonly infants under two months are the most vulnerable and have the highest mortality rate<ref name=Kapil>[https://www.sciencedirect.com/science/article/pii/S0952791518301237#bib0025/ Kapil, P. Merkel, T.J. "Pertussis vaccines and protective immunity" Current Opinion in Immunology. 59:72-78]</ref>. In infants symptoms include: gagging, gasping, bradycardia, cyanosis, and vomiting<ref name=Cherry2>[https://academic.oup.com/cid/article/63/suppl_4/S119/2526416?login=true/ "Pertussis in Young Infants Throughout the World " Clinical Infectious Diseases. 63:S119-S122]</ref>. Mortality in infants most commonly is from leukocytosis, pulmonary hypertension, and pneumonia<ref name=Cherry2></ref>. Treatment for whooping cough consists of antibiotic therapy and intubation with ventilation and oxygen<ref name=Cherry2></ref>. | ||
| Line 10: | Line 9: | ||
==<i>Bordetella pertussis</i> and Infection Stages== | ==<i>Bordetella pertussis</i> and Infection Stages== | ||
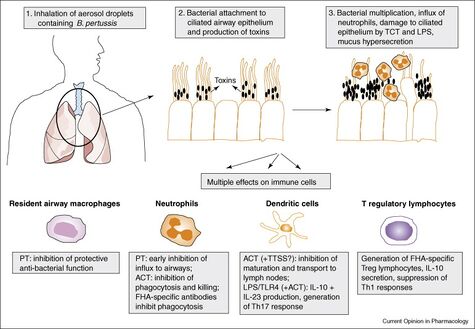
[[Image:1-s2.0-S1471489207000598-gr1.jpg|thumb| | [[Image:1-s2.0-S1471489207000598-gr1.jpg|thumb|475px|right|This diagram depict the infection of <i> B. pertussis</i> and effects on the Immune System. The image credit belongs to Nicholas H Carbonetti. [https://www.sciencedirect.com/science/article/pii/S1471489207000598.]] | ||
<i>B. pertussis</i> is a gram-negative, aerobic coccobacillus bacteria a part of the genus Bordetella<ref name=Guiso></ref>. <i>B. pertussis</i> produces antigenic and biologically active products: pertussis toxin, filamentous hemagglutinin, agglutinogens, adenylate cyclase, pertactin, and tracheal cytotoxin<ref name= Havers></ref>. An immune response to one of these products will result in immunity after infection<ref name= Havers></ref>.<i>Pertussis</i> is a human-specific pathogen causing severe respiratory disease that cannot survive outside of its host<ref name=Guiso></ref>. Infection occurs with <i>B. pertussis</i> entering through the host airway via respiratory droplets from an already infected individual, usually from coughing <ref name=Smith> [https://academic.oup.com/femsre/article/25/3/309/624335/ Smith, A.M et al. "The virulence factors of <i>Bordetella pertussis</i>: a matter of control" 2001. FEMS Microbiology Reviews 25:309-333.]</ref>. <i>B. pertussis</i> then proceeds down the respiratory tract and adheres to ciliated epithelial cells in the trachea and nasopharynx <ref name=Smith></ref>. Once the bacteria is attached it begins to replicate and colonies with-in the host cells <ref name=Smith></ref>. Toxins are secreted by micro-organism which inflict damage to the epithelial lining, resulting in loss of ciliated cells which induces the coughing most commonly associated with whooping cough <ref name=Smith></ref>. The pertussis toxins released also allow the bacteria to avoid host immune response by interfering with clearance mechanisms <ref name=Smith></ref>. The damage caused by the toxins released by the replicating bacteria also halts ciliary function, short-circuiting host G proteins signaling apparatus, and also inhibits immune cell functions by up regulating cAMP levels <ref name=Smith></ref>. | <i>B. pertussis</i> is a gram-negative, aerobic coccobacillus bacteria a part of the genus Bordetella<ref name=Guiso></ref>. <i>B. pertussis</i> produces antigenic and biologically active products: pertussis toxin, filamentous hemagglutinin, agglutinogens, adenylate cyclase, pertactin, and tracheal cytotoxin<ref name= Havers></ref>. An immune response to one of these products will result in immunity after infection<ref name= Havers></ref>.<i>Pertussis</i> is a human-specific pathogen causing severe respiratory disease that cannot survive outside of its host<ref name=Guiso></ref>. Infection occurs with <i>B. pertussis</i> entering through the host airway via respiratory droplets from an already infected individual, usually from coughing <ref name=Smith> [https://academic.oup.com/femsre/article/25/3/309/624335/ Smith, A.M et al. "The virulence factors of <i>Bordetella pertussis</i>: a matter of control" 2001. FEMS Microbiology Reviews 25:309-333.]</ref>. <i>B. pertussis</i> then proceeds down the respiratory tract and adheres to ciliated epithelial cells in the trachea and nasopharynx <ref name=Smith></ref>. Once the bacteria is attached it begins to replicate and colonies with-in the host cells <ref name=Smith></ref>. Toxins are secreted by micro-organism which inflict damage to the epithelial lining, resulting in loss of ciliated cells which induces the coughing most commonly associated with whooping cough <ref name=Smith></ref>. The pertussis toxins released also allow the bacteria to avoid host immune response by interfering with clearance mechanisms <ref name=Smith></ref>. The damage caused by the toxins released by the replicating bacteria also halts ciliary function, short-circuiting host G proteins signaling apparatus, and also inhibits immune cell functions by up regulating cAMP levels <ref name=Smith></ref>. | ||
| Line 16: | Line 15: | ||
==<i>B. Pertussis</i> Vaccine History== | ==<i>B. Pertussis</i> Vaccine History== | ||
[[Image:pertussis-vacc-all.jpeg|thumb| | [[Image:pertussis-vacc-all.jpeg|thumb|400px|left|This figure displays when individual age groups should receive a <i>B. pertussis </i>vaccine and what vaccine they should get.[https://www.cdc.gov/pertussis/images/pertussis-vacc-all.jpg]] | ||
Soon after <i>B. pertussis</i> was isolated as the causative agent of whooping cough, Pertussis whole-cell (Pw) vaccines were implemented using heat-killed bacteria <ref name=Guiso></ref>. Starting in 1914 the first vaccine was licensed in the United States using whole killed bacteria but was used more for treatment than prevention <ref name=Klein>[https://www.publications.aap.org/pediatrics/article-split/131/6/e1716/31044/Comparative-Effectiveness-of-Acellular-Versus/ Klein, N.P et al. "Comparative effectiveness of acellular versus whole-cell pertussis vaccines in teenagers" 2013. Pediatrics]</ref>. The problem with early Pw vaccines is they killed whole cell organisms which contained endotoxins that could cause serious side effects<ref name=Cherry>[https://publications.aap.org/pediatrics/article/115/5/1422/67631/The-Epidemiology-of-Pertussis-A-Comparison-of-the/ Kerr, Cherry J.D."The Epidemiology of Pertussis: A Comparison of the Epidemiology of the Disease pertussis With the Epidemiology of <i>Bordetella pertussis</i> Infection" 2005. American Academy of Pediatrics" 1422-1427.]</ref>. Soon after the Pw vaccine was first discovered, new research and diagnostic tools were used to continue researching for more effective and safe vaccines. Through 1934-1935 Pearl Kendrick and Grace Eldering ran clinical trials on children using a whole-cell vaccine that was generated from chemically inactive bacteria instead of just heat killed bacteria <ref name=Kuchar></ref>. The vaccine showed an efficacy rate of 89% and was widely distributed <ref name=Kuchar></ref>. Pw vaccines became used for wholesale distribution starting in the 1940’s <ref name=Mattoo> [https://journals.asm.org/doi/full/10.1128/CMR.18.2.326-382.2005/ Mattoo, S. Cherry, J.D. "Molecular Pathogenesis, Epidemiology, and Clinical Manifestations of Respiratory Infections Due to <i>Bordetella pertussis</i> and Other <i>Bordetella</i> Subspecies" 2005. Clinical Microbiology Reviews]</ref>. Since <i>B. pertussis</i> in human host restrictive, there is a very low display of genetic diversity <ref name=Guiso></ref>. Recent analyses have concluded that there were several different types of isolates circulating in the pre-vaccine era that produce various proteins <ref name=Guiso></ref>. Only one or two types predominated with no significant difference in an associated pattern to which was the dominant isolate <ref name=Guiso></ref>. This allowed for the Pw vaccine to be composed of 1-3 strains selected from the predominant isolate types <ref name=Guiso></ref>. Pw vaccines were given to infants and toddlers extensively due to the majority of infections occurring in younger children <ref name=Guiso></ref>. Where vaccine distribution was effective, vaccine strains of <i>B. pertussis</i> were low and in areas with low vaccination rates the pre-vaccine era isolated were still circulating <ref name=Guiso></ref>. Pw vaccines were shown to create immunity for some isolates of <i>B. pertussis</i> but not all isolates <ref name=Guiso></ref>. | Soon after <i>B. pertussis</i> was isolated as the causative agent of whooping cough, Pertussis whole-cell (Pw) vaccines were implemented using heat-killed bacteria <ref name=Guiso></ref>. Starting in 1914 the first vaccine was licensed in the United States using whole killed bacteria but was used more for treatment than prevention <ref name=Klein>[https://www.publications.aap.org/pediatrics/article-split/131/6/e1716/31044/Comparative-Effectiveness-of-Acellular-Versus/ Klein, N.P et al. "Comparative effectiveness of acellular versus whole-cell pertussis vaccines in teenagers" 2013. Pediatrics]</ref>. The problem with early Pw vaccines is they killed whole cell organisms which contained endotoxins that could cause serious side effects<ref name=Cherry>[https://publications.aap.org/pediatrics/article/115/5/1422/67631/The-Epidemiology-of-Pertussis-A-Comparison-of-the/ Kerr, Cherry J.D."The Epidemiology of Pertussis: A Comparison of the Epidemiology of the Disease pertussis With the Epidemiology of <i>Bordetella pertussis</i> Infection" 2005. American Academy of Pediatrics" 1422-1427.]</ref>. Soon after the Pw vaccine was first discovered, new research and diagnostic tools were used to continue researching for more effective and safe vaccines. Through 1934-1935 Pearl Kendrick and Grace Eldering ran clinical trials on children using a whole-cell vaccine that was generated from chemically inactive bacteria instead of just heat killed bacteria <ref name=Kuchar></ref>. The vaccine showed an efficacy rate of 89% and was widely distributed <ref name=Kuchar></ref>. Pw vaccines became used for wholesale distribution starting in the 1940’s <ref name=Mattoo> [https://journals.asm.org/doi/full/10.1128/CMR.18.2.326-382.2005/ Mattoo, S. Cherry, J.D. "Molecular Pathogenesis, Epidemiology, and Clinical Manifestations of Respiratory Infections Due to <i>Bordetella pertussis</i> and Other <i>Bordetella</i> Subspecies" 2005. Clinical Microbiology Reviews]</ref>. Since <i>B. pertussis</i> in human host restrictive, there is a very low display of genetic diversity <ref name=Guiso></ref>. Recent analyses have concluded that there were several different types of isolates circulating in the pre-vaccine era that produce various proteins <ref name=Guiso></ref>. Only one or two types predominated with no significant difference in an associated pattern to which was the dominant isolate <ref name=Guiso></ref>. This allowed for the Pw vaccine to be composed of 1-3 strains selected from the predominant isolate types <ref name=Guiso></ref>. Pw vaccines were given to infants and toddlers extensively due to the majority of infections occurring in younger children <ref name=Guiso></ref>. Where vaccine distribution was effective, vaccine strains of <i>B. pertussis</i> were low and in areas with low vaccination rates the pre-vaccine era isolated were still circulating <ref name=Guiso></ref>. Pw vaccines were shown to create immunity for some isolates of <i>B. pertussis</i> but not all isolates <ref name=Guiso></ref>. | ||
<br> By 1943 the American Academy of Pediatrics (ACIP) allowed for routine use in all children <ref name=Kuchar></ref>. In 1948 the DTP vaccine, which contained diphtheria, tetanus, and whole-cell pertussis vaccine) was created as used effectively<ref name=Kuchar></ref>. By the 1980s <i>B. pertussis</i> infections had fallen 99% and the fear of infection was replaced into the fear of the vaccine’s side effects<ref name=Kuchar></ref>. Common side effects included: fever, crying, febrile seizures, limb redness and swelling, and hypo-responsive episodes<ref name=Kuchar></ref>. The side effects did not cause extreme harm but the public began linking the vaccine to sudden infant death syndrome (SIDS), asthma, and encephalopathies<ref name=Kuchar></ref>. Through the 1970-8os there was a decrease in vaccination rates due to countries recalling the vaccine due to safety reasons or lack of parental approval <ref name=Klein></ref>. | <br> By 1943 the American Academy of Pediatrics (ACIP) allowed for routine use in all children <ref name=Kuchar></ref>. In 1948 the DTP vaccine, which contained diphtheria, tetanus, and whole-cell pertussis vaccine) was created as used effectively<ref name=Kuchar></ref>. By the 1980s <i>B. pertussis</i> infections had fallen 99% and the fear of infection was replaced into the fear of the vaccine’s side effects<ref name=Kuchar></ref>. Common side effects included: fever, crying, febrile seizures, limb redness and swelling, and hypo-responsive episodes<ref name=Kuchar></ref>. The side effects did not cause extreme harm but the public began linking the vaccine to sudden infant death syndrome (SIDS), asthma, and encephalopathies<ref name=Kuchar></ref>. Through the 1970-8os there was a decrease in vaccination rates due to countries recalling the vaccine due to safety reasons or lack of parental approval <ref name=Klein></ref>. Another problem with wP vaccines was the amount of biologically active components of the vaccine were unclear and made researching the efficacy and effectiveness of the vaccine difficult<ref name= Chen>[https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5404361/#cit0020/ Chen, Z. He, Q. "Immune persistence after pertussis vaccination" 2017. Human Vaccin Immunother. 13:744-756.]</ref>. | ||
<br> With the decrease of vaccination and the increase of cases, research began developing an effective acellular vaccine containing B. pertussis antigens rather than heat killed bacteria <ref name=Kuchar></ref>. This vaccine contained formaldehyde-treated filamentous hemagglutinin and formalin-inactivated pertussis toxin which resulted in a 69% efficacy rate<ref name=Klein></ref>. By the 1990s the use of a combined diphtheria, tetanus, and acellular <i>pertussis</i> vaccine (DTaP) became the most common form of the vaccine in the US<ref name=Kuchar></ref>. To date, the vaccine has continued to be successful but the length of protection has been ambiguous<ref name=Kuchar></ref>. Most commonly, research has shown additional doses of the vaccine may be necessary to have immunity against the disease <ref name=Gustafsson>[https://www.publications.aap.org/pediatrics/article-split/118/3/978/69421/Long-term-Follow-up-of-Swedish-Children-Vaccinated/ Gustafsson, L. et al. "Long-term Follow-up Swedish Children Vaccinated With Acellular Pertussis Vaccines at 3, 5, and 12 Months of Age Indicates the Need for a Booster Dose at 5 to 7 Years of Age" 2006. American Academy of Pediatrics. 978-984]</ref>. The vaccine showed an efficacy rate of 89% and was widely distributed <ref name=Kuchar></ref>. The ACIP and the CDC currently recommends a four to five doses of the DTaP with three during infancy and a booster in pre-school age children or in adolescence<ref name=Guiso></ref>. It is also recommended that pregnant women receive an additional dose during pregnancy<ref name=Liang>[https://www.cdc.gov/mmwr/volumes/67/rr/rr6702a1.htm/Comparative-Effectiveness-of-Acellular-Versus/ Liang, J.L et al. "Prevention of Pertussis, Tetanus, and Diphtheria with Vaccines in the United States: Recommendations of the Advisory Committtee Immunization Practices (ACIP)" 2018.Recommendations and Reports]</ref>. Receiving a dose during the third trimester of pregnancy protects the unborn child from contracting whooping cough during the first few months of life<ref name= Cleveland>[https://my.clevelandclinic.org/health/diseases/15661-whooping-cough-pertussis/Cleveland Clinic. "Pertussis(Whooping Cough)Vaccine" 2022]</ref>. | <br> With the decrease of vaccination and the increase of cases, research began developing an effective acellular vaccine containing B. pertussis antigens rather than heat killed bacteria <ref name=Kuchar></ref>. This vaccine contained formaldehyde-treated filamentous hemagglutinin and formalin-inactivated pertussis toxin which resulted in a 69% efficacy rate<ref name=Klein></ref>. By the 1990s the use of a combined diphtheria, tetanus, and acellular <i>pertussis</i> vaccine (DTaP) became the most common form of the vaccine in the US<ref name=Kuchar></ref>. To date, the vaccine has continued to be successful but the length of protection has been ambiguous<ref name=Kuchar></ref>. Most commonly, research has shown additional doses of the vaccine may be necessary to have immunity against the disease <ref name=Gustafsson>[https://www.publications.aap.org/pediatrics/article-split/118/3/978/69421/Long-term-Follow-up-of-Swedish-Children-Vaccinated/ Gustafsson, L. et al. "Long-term Follow-up Swedish Children Vaccinated With Acellular Pertussis Vaccines at 3, 5, and 12 Months of Age Indicates the Need for a Booster Dose at 5 to 7 Years of Age" 2006. American Academy of Pediatrics. 978-984]</ref>. The vaccine showed an efficacy rate of 89% and was widely distributed <ref name=Kuchar></ref>. The ACIP and the CDC currently recommends a four to five doses of the DTaP with three during infancy and a booster in pre-school age children or in adolescence<ref name=Guiso></ref>. It is also recommended that pregnant women receive an additional dose during pregnancy<ref name=Liang>[https://www.cdc.gov/mmwr/volumes/67/rr/rr6702a1.htm/Comparative-Effectiveness-of-Acellular-Versus/ Liang, J.L et al. "Prevention of Pertussis, Tetanus, and Diphtheria with Vaccines in the United States: Recommendations of the Advisory Committtee Immunization Practices (ACIP)" 2018.Recommendations and Reports]</ref>. Receiving a dose during the third trimester of pregnancy protects the unborn child from contracting whooping cough during the first few months of life<ref name= Cleveland>[https://my.clevelandclinic.org/health/diseases/15661-whooping-cough-pertussis/Cleveland Clinic. "Pertussis(Whooping Cough)Vaccine" 2022]</ref>. | ||
==Vaccine Virulence== | ==Vaccine Virulence== | ||
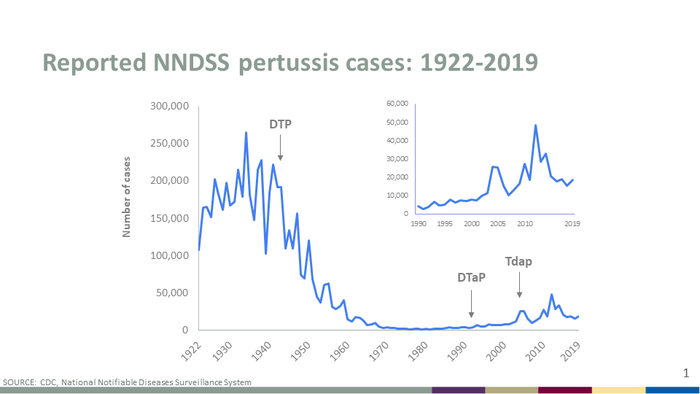
[[Image:Incidence-graph-2019.png|thumb| | [[Image:Incidence-graph-2019.png|thumb|700px|left|This diagram depict the number of incidences of <i> B. pertussis</i> that were reported to the CDC from 1922 to 2019. The image credit belongs to the CDC. [https://www.cdc.gov/pertussis/surv-reporting.html]] | ||
While <i>B. pertussis</i> vaccines have decreased overall mortality rates by over 90% there is still a current resurgence in whooping cough cases <ref name=Dorji>[https://link.springer.com/article/10.1007/s00430-017-0524-z#ref-CR38/ Dorji, D. et al. "<i>Bordetella pertussis</i> virulence factors in the continuing evolution of whooping cough vaccines for improved performance." 2018. Medical Microbiology and Immunology 207:3-26.]</ref>. The two most common reasons for the resurgence of whooping cough is that the aP (acellular whole cell) vaccine has a relative short-term protection rate and the evolution of the <i>B. pertussis</i> pathogen <ref name=Dorji></ref>. Current evolution has included the expansion of B. pertussis strains that contain an allele for the <i>pertussis</i> toxin (ptx) promotor <i>ptxP3</i> that replaces the <i>ptxP1</i> strains <ref name= King>[https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0066150/ King A.J. et al. "Genome-Wide Gene Expression Analysis of <i>Bordetella pertussis</i> Isolates Associated with a Resurgence in Pertussis: Elucidation of Factors Involved in the Increased Fitness of Epidemic Strains." 2013. PLOS Computational Biology.]</ref>. <i>PtxP3</i> strains produce more <i>ptx</i> than the <i>ptxP1</i> stains which increase the virulence factors of <i>B. pertussis</i> that increases the pathogens overall fitness<ref name= King></ref>. <i>PtxP3</i> causing <i>B. pertussis</i> produces more pertussis toxins and suppress the host immune system more effectively than <i>ptxP1</i><ref name= King></ref>. Sarfarchi et al (2016) found that the <i>ptxP3</i> strains colonized the respiratory tract in vaccinated and unvaccinated mice better than <i>ptxP1</i> <ref name=Safarchi>[https://www.sciencedirect.com/science/article/pii/S0264410X16304765/ Safarchi, A. et al. "Better colonisation of newly emerged <i>Bordetella pertussis</i> in the co-infection mouse model study" 2016. Vaccine 34:3967-3971.]</ref>. Along with the <i>ptxP3</i> strain evolving, new <i>B. pertussis</i> strains can increase expression of a regulatory molecule PD-LI which may produce vaccine-induced protective responses<ref name=Dorji></ref>. Recently, there has been an increase in mutations of <i>B. pertussis</i> that are not producing pertactin which is a <i>pertussis</i> acellular vaccine immunogen that is a part of the bacteria adhering to the upper respiratory epithelium <ref name=Bodilis>[https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3647673/#R8/ Bodilis, H and Guiso, N."Virulence of Pertactin-Negative <i>Bordetella pertussis</i> Isolates from Infants, France" 2013. Emerging Infectious Disease 19:471-474.]</ref>. The strains of <i>B. pertussis</i> that do not express pertactin have been increasing in regions where acellular <i>pertussis</i> vaccines have been used for over seven years <ref name=Bodilis></ref>. | While <i>B. pertussis</i> vaccines have decreased overall mortality rates by over 90% there is still a current resurgence in whooping cough cases <ref name=Dorji>[https://link.springer.com/article/10.1007/s00430-017-0524-z#ref-CR38/ Dorji, D. et al. "<i>Bordetella pertussis</i> virulence factors in the continuing evolution of whooping cough vaccines for improved performance." 2018. Medical Microbiology and Immunology 207:3-26.]</ref>. The two most common reasons for the resurgence of whooping cough is that the aP (acellular whole cell) vaccine has a relative short-term protection rate and the evolution of the <i>B. pertussis</i> pathogen <ref name=Dorji></ref>. Current evolution has included the expansion of B. pertussis strains that contain an allele for the <i>pertussis</i> toxin (ptx) promotor <i>ptxP3</i> that replaces the <i>ptxP1</i> strains <ref name= King>[https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0066150/ King A.J. et al. "Genome-Wide Gene Expression Analysis of <i>Bordetella pertussis</i> Isolates Associated with a Resurgence in Pertussis: Elucidation of Factors Involved in the Increased Fitness of Epidemic Strains." 2013. PLOS Computational Biology.]</ref>. <i>PtxP3</i> strains produce more <i>ptx</i> than the <i>ptxP1</i> stains which increase the virulence factors of <i>B. pertussis</i> that increases the pathogens overall fitness<ref name= King></ref>. <i>PtxP3</i> causing <i>B. pertussis</i> produces more pertussis toxins and suppress the host immune system more effectively than <i>ptxP1</i><ref name= King></ref>. Sarfarchi et al (2016) found that the <i>ptxP3</i> strains colonized the respiratory tract in vaccinated and unvaccinated mice better than <i>ptxP1</i> <ref name=Safarchi>[https://www.sciencedirect.com/science/article/pii/S0264410X16304765/ Safarchi, A. et al. "Better colonisation of newly emerged <i>Bordetella pertussis</i> in the co-infection mouse model study" 2016. Vaccine 34:3967-3971.]</ref>. Along with the <i>ptxP3</i> strain evolving, new <i>B. pertussis</i> strains can increase expression of a regulatory molecule PD-LI which may produce vaccine-induced protective responses<ref name=Dorji></ref>. Recently, there has been an increase in mutations of <i>B. pertussis</i> that are not producing pertactin which is a <i>pertussis</i> acellular vaccine immunogen that is a part of the bacteria adhering to the upper respiratory epithelium <ref name=Bodilis>[https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3647673/#R8/ Bodilis, H and Guiso, N."Virulence of Pertactin-Negative <i>Bordetella pertussis</i> Isolates from Infants, France" 2013. Emerging Infectious Disease 19:471-474.]</ref>. The strains of <i>B. pertussis</i> that do not express pertactin have been increasing in regions where acellular <i>pertussis</i> vaccines have been used for over seven years <ref name=Bodilis></ref>. | ||
<br> As the number of pseudo-genes decreases, B. pertussis seems to adapt to survive their human host and continue to lose genes not | <br> As the number of pseudo-genes decreases, <i>B. pertussis</i> seems to adapt to survive their human host and continue to lose genes not | ||
not necessary for human infection<ref name=Guiso></ref>. Pw vaccination did not control the circulation of other isolates and did not control virulence of isolates<ref name=Guiso></ref>. The increase of aP vaccine induced immunity in the population and new isolates would lack the vaccine antigens due to deletion or inactivation of their structural genes<ref name=Guiso></ref>. Work by Hegerle et al. showed that wP vaccines induced immunity led to having one form of B. pertussis whereas aP vaccine-induced immunity lead to increase in the number of B. pertussis isolates not expressing virulence factors <ref name=Hegerle>[https://www.sciencedirect.com/science/article/pii/S1198743X14610508/ Hegerle, N. et al. "Evolution of French <i>Bordetella pertussis</i> and <i>Bordetella parapertussis</i> isolates: increase of <i>Bordetellae</i> not expressing pertactin" 2012. Clinical Microbiology and Infection 18:E340-E346.]</ref>. The expression of virulence factors comes from the regulation of the Bordetella virulence gene (bvg) that encodes for two proteins that sense the environmental conditions and act according to the conditions<ref name=Smith></ref>. Spontaneous mutation during DNA replication has also shown to produce avirulent variants of <i>B. pertussis</i><ref name=Guiso></ref>. This form of bacteria does not have the ability to invade and colonise the host<ref name=Smith></ref>.Overall the circulation of B. pertussis likely to decrease but isolates that do not produce vaccine antigens may persist<ref name=Guiso></ref>. | not necessary for human infection<ref name=Guiso></ref>. Pw vaccination did not control the circulation of other isolates and did not control virulence of isolates<ref name=Guiso></ref>. The increase of aP vaccine induced immunity in the population and new isolates would lack the vaccine antigens due to deletion or inactivation of their structural genes<ref name=Guiso></ref>. Work by Hegerle et al. showed that wP vaccines induced immunity led to having one form of <i>B. pertussis</i> whereas aP vaccine-induced immunity lead to increase in the number of <i>B. pertussis</i> isolates not expressing virulence factors <ref name=Hegerle>[https://www.sciencedirect.com/science/article/pii/S1198743X14610508/ Hegerle, N. et al. "Evolution of French <i>Bordetella pertussis</i> and <i>Bordetella parapertussis</i> isolates: increase of <i>Bordetellae</i> not expressing pertactin" 2012. Clinical Microbiology and Infection 18:E340-E346.]</ref>. The expression of virulence factors comes from the regulation of the <i>Bordetella</i> virulence gene (bvg) that encodes for two proteins that sense the environmental conditions and act according to the conditions<ref name=Smith></ref>. Spontaneous mutation during DNA replication has also shown to produce avirulent variants of <i>B. pertussis</i><ref name=Guiso></ref>. This form of bacteria does not have the ability to invade and colonise the host<ref name=Smith></ref>.Overall the circulation of <i>B. pertussis</i> is likely to decrease but isolates that do not produce vaccine antigens may persist<ref name=Guiso></ref>. | ||
<br>Another problem with B. pertussis vaccines is the length of time immunity is provided. With the invention of the acellular vaccine patients had less side effects and was overall tolerated but follow-up research indicates a decrease in protection against pertussis<ref name= Kuchar></ref>.The switch from whole-cell vaccines to the acellular vaccine reversed the trend of decreasing infections to an increase of overall B. pertussis infections <ref name=Tan>[https://journals.lww.com/pidj/fulltext/2015/09000/Pertussis_Across_the_Globe__Recent_Epidemiologic.23.aspx/ Tan, T. et al. "Pertussis Across the Globe | <br>Another problem with <i>B. pertussis</i> vaccines is the length of time immunity is provided. With the invention of the acellular vaccine patients had less side effects and was overall tolerated but follow-up research indicates a decrease in protection against <i>pertussis</i><ref name= Kuchar></ref>.The switch from whole-cell vaccines to the acellular vaccine reversed the trend of decreasing infections to an increase of overall <i>B. pertussis</i> infections <ref name=Tan>[https://journals.lww.com/pidj/fulltext/2015/09000/Pertussis_Across_the_Globe__Recent_Epidemiologic.23.aspx/ Tan, T. et al. "Pertussis Across the Globe | ||
Recent Epidemiologic Trends From 2000 to 2013" 2015. The Pediatric Infectious Disease Journal 34:e222-e232.]</ref>. | Recent Epidemiologic Trends From 2000 to 2013" 2015. The Pediatric Infectious Disease Journal 34:e222-e232.]</ref>. When vaccine protection is compared to naturally gained immunity vaccine protection has a shorter length of protection<ref name= Chen></ref>. Along with natural immunity, antibody responses have also supported that immunity decreases after vaccination with the acellular <i>B. pertussis</i> vaccine<ref name= Chen></ref>. | ||
<br>The past strategy of reducing cases of whooping cough most likely needs to be re-evaluated. During cyclic outbreaks of | <br>The past strategy of reducing cases of whooping cough most likely needs to be re-evaluated to current epidemiological trends. During cyclic outbreaks of <i>B. pertussis</i> older children, adolescents, and adults have made up the majority of cases <ref name= Kuchar></ref>. The current immunization programs target infants, which has caused a significant decrease in mortality in a vulnerable population<ref name= Kuchar></ref>. Tdap vaccination was primarily used to reduce the burden of <i>B. pertussis</i> among older populations while also protecting infants from the transmission of the disease<ref name= Chen></ref>. Protection is currently seen as short term and results in a population of other kids/adults that is no longer immune<ref name= Kuchar></ref>. There is also a switch in family dynamics where families are having a smaller number of children and spending less time with others due to the rising age of technology<ref name= Kuchar></ref>. This can result in fewer populations experiencing natural immunity of <i>B. pertussis</i><ref name= Kuchar></ref>. | ||
==Why Vaccination?== | ==Why Vaccination?== | ||
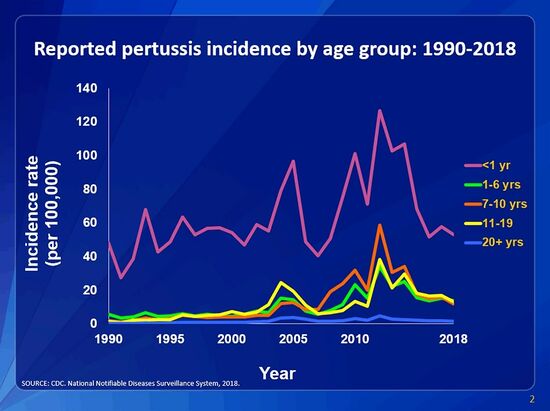
[[Image:incidence-graph-age-2018.jpg|thumb| | [[Image:incidence-graph-age-2018.jpg|thumb|550px|right|This graph shows the reported B. pertussis incidences for 1990-2018 by age group (per 100,00 persons). The graph shows infants less than one year old have the greatest risk for disease compared to all other age groups. [https://www.cdc.gov/pertussis/surv-reporting.html]] | ||
With widespread use of vaccination there are still close to 48.5 million yearly cases and 250,000 deaths <ref name=Mattoo></ref>. This partially surfaces from Incomplete immunity in infants who have received fewer than three doses and have relatively short-lived immunity <ref name=Mattoo></ref>. The biggest worries towards <i>B. pertussis</i> to become epidemiological is due to adults and young adults being a major source of transmission and a reservoir<ref name=Mattoo></ref>. Vaccination coverage with DTaP remains at a 95% rate for children 19-35 months high infants younger than 6 month have the highest rate of infection at 72.3% per 100,000 <i>pertussis</i> cases (Havers). This high number of cases is the result of infants being too young to be completely vaccinated for B. pertussis <ref name=Greef>[https://academic.oup.com/cid/article/50/10/1339/375948?login=true/ Greef S.C de et al."Pertussis Disease Burden in the Household: How to Protect Young Infants" 2010. Clinical Infectious Disease. 50:1339-1345]</ref>. Studies by Greef et al (2010) showed that infants hospitalized with whooping cough were primarily infected by their siblings or their mother<ref name=Greef></ref>. Adults, vaccinated and unvaccinated, can spread it to infants which causes a more life-threatening disease<ref name=Greef></ref>. Most commonly infants get whooping cough from an adult individual with a cough illness that is not recognized as pertussis <ref name=Cherry></ref>. Infants that contract B. pertussis may develop pneumonia, uncontrollable shaking, and brain damage whereas young adults and adults may only develop cold-like symptoms<ref name=Cleveland></ref>. It is recommended that anyone who is planning to be around a newborn child in the first few months of life be vaccinated for <i>B. pertussis</i> <ref name=Cleveland></ref> | |||
With widespread use of vaccination there are still close to 48.5 million yearly cases and 250,000 deaths <ref name=Mattoo></ref>. This partially surfaces from Incomplete immunity in infants who have received fewer than three doses and have relatively short-lived immunity <ref name=Mattoo></ref>. The biggest worries towards <i>B. pertussis</i> to become epidemiological is due to adults and young adults being a major source of transmission and a reservoir<ref name=Mattoo></ref>. Vaccination coverage with DTaP remains at a 95% rate for children 19-35 months high infants younger than 6 month have the highest rate of infection at 72.3% per 100,000 <i>pertussis</i> cases (Havers). This high number of cases is the result of infants being too young to be completely vaccinated for B. pertussis <ref name=Greef>[https://academic.oup.com/cid/article/50/10/1339/375948?login=true/ Greef S.C de et al."Pertussis Disease Burden in the Household: How to Protect Young Infants" 2010. Clinical Infectious Disease. 50:1339-1345]</ref>. Studies by Greef et al (2010) showed that infants hospitalized with whooping cough were primarily infected by their siblings or their mother<ref name=Greef></ref>. Adults, vaccinated and unvaccinated, can spread it to infants which causes a more life-threatening disease<ref name=Greef></ref>. Most commonly infants get whooping cough from an adult individual with a cough illness that is not recognized as pertussis <ref name=Cherry></ref>. Infants that contract B. pertussis may develop pneumonia, uncontrollable shaking, and brain damage whereas young adults and adults may only develop cold-like symptoms<ref name=Cleveland></ref>. It is recommended that anyone who is planning to be around a newborn child in the first few months of life be vaccinated for B. pertussis <ref name=Cleveland></ref> | |||
<br><i>B. pertussis</i> has shown to follow a cyclic pattern with peaks of infection every 2 to 5 years regardless of vaccination <ref name=Cherry></ref>. This cyclic pattern occurred during whole-cell <i>pertussis</i> vaccination era and with the present acellular vaccine era<ref name=Cherry2></ref>. Along with a pattern of infection, the increase of pertussis cases coincides with the transition to acellular vaccines in the United States<ref name=Havers></ref>. The newer vaccine displays less side effects than the Pw vaccine but has a lack of long term immunity<ref name=Kuchar></ref>. While vaccines are highly successful, immunity is not lasting<ref name=Cherry2></ref>. | <br><i>B. pertussis</i> has shown to follow a cyclic pattern with peaks of infection every 2 to 5 years regardless of vaccination <ref name=Cherry></ref>. This cyclic pattern occurred during whole-cell <i>pertussis</i> vaccination era and with the present acellular vaccine era<ref name=Cherry2></ref>. Along with a pattern of infection, the increase of pertussis cases coincides with the transition to acellular vaccines in the United States<ref name=Havers></ref>. The newer vaccine displays less side effects than the Pw vaccine but has a lack of long term immunity<ref name=Kuchar></ref>. While vaccines are highly successful, immunity is not lasting<ref name=Cherry2></ref>. | ||
| Line 46: | Line 43: | ||
==Conclusion== | ==Conclusion== | ||
Overall <i>B. pertussis</i> infections have decreased significantly since the discovery of the pathogen and the invention of effective and safe vaccines. Whooping cough is still a significant disease affecting mainly infants who are too young to receive a vaccine. Vaccines have been shown to give direct protection to vaccinated individuals while also displaying an indirect effect of protection for unvaccinated individuals in a population<ref name=Doherty>[https://www.sciencedirect.com/science/article/pii/S0264410X16309434?via%3Dihub/ Doherty et al. "Vaccine impact: Benefits for human health" 2016. Vaccine 34: 6707-6714]</ref>. In the case of <i>B. pertussis</i>, individuals getting vaccinated, especially other age groups, can create an indirect effect of protection on infants and younger children who are unable to be vaccinated. Most industrialized countries have been able to control B. pertussis infections but due to whooping cough being seen as a minor problem and pressure from anti-immunization groups have caused fluctuations in vaccination rates<ref name= Galazka></ref> | |||
<br>More research is needed on the true effectiveness of the DTaP vaccine in terms of lasting immunity for <i>B. pertussis</i>. Research could include finding a way to incorporate the effectiveness of immunity of <i>pertussis</i> whole-cell vaccines with the safety of acellular <i>pertussis</i> vaccines to create a safe and long lasting vaccine. Research could be conducted on why infants and young children have more severe symptoms to <i> B. pertussis</i> infection than young adults and adults. This would create a better understanding of rates of infection and how the disease works as a pathogen in the presence of vaccines. More research could be completed on why immunity is not long term after vaccination or natural immunity from already having <i>B. pertussis</i>. This would allow for more understanding on how populations can protect themselves even after being vaccinated. A potential benefit that comes from more research on effectiveness and lasting immunity is to continue education on vaccination and the causative effects of the disease. Public influence is a large part of how individuals make decisions so providing accurate resources that education concisely would be a benefit towards controlling new infections. The primary fear being parents not vaccinating their children for <i>B. pertussis</i> is due to misconceptions about side effects of the vaccine. <i>B. pertussis</i> is a preventable disease where spread can be limited by vaccination from a young age and would decrease the amount of lives lost each year. The main goal of any person should be to protect others who are more susceptible to <i>B. pertussis</i>. Infants and small children with zero doses or one to two doses are extremely susceptible and can experience extreme symptoms. Protecting the susceptible allows for a decrease in mortality rates and infections. | |||
<br>More research is needed on the true effectiveness of the DTaP vaccine in terms of lasting immunity for <i>B. pertussis</i>. Research could include finding a way to incorporate the effectiveness of immunity of <i>pertussis</i> whole-cell vaccines with the safety of acellular <i>pertussis</i> vaccines to create a safe and long lasting vaccine. More research could be completed on why immunity is not long term after vaccination or natural immunity from already having <i>B. pertussis</i>. This would allow for more understanding on how populations can protect themselves even after being vaccinated. A potential benefit that comes from more research on effectiveness and lasting immunity is to continue education on vaccination and the causative effects of the disease. Public influence is a large part of how individuals make decisions so providing accurate resources that education concisely would be a benefit towards controlling new infections. The primary fear being parents not vaccinating their children for <i>B. pertussis</i> is due to misconceptions about side effects of the vaccine. <i>B. pertussis</i> is a preventable disease where spread can be limited by vaccination from a young age and would decrease the amount of lives lost each year. The main goal of any person should be to protect others who are more susceptible to <i>B. pertussis</i>. Infants and small children with zero doses or one to two doses are extremely susceptible and can experience extreme symptoms. Protecting the susceptible allows for a decrease in mortality rates and infections. | |||
==References== | ==References== | ||
<references /> | <references /> | ||
<br><br>Authored for BIOL 238 Microbiology, taught by [mailto:slonczewski@kenyon.edu Joan Slonczewski], 2022, [http://www.kenyon.edu/index.xml Kenyon College] | <br><br>Authored for BIOL 238 Microbiology, taught by [mailto:slonczewski@kenyon.edu Joan Slonczewski], 2022, [http://www.kenyon.edu/index.xml Kenyon College] | ||
Latest revision as of 22:51, 17 April 2022
Introduction and History
By Alexandra White
Vaccination is a widely used practice to help prevent infectious disease and commonly spread illnesses. A widely known and common vaccine is used to prevent infection of Bordetella pertussis, the causative agent of Whooping cough. Whooping cough is known as one of the most common death causing infectious disease in the world[1]. The disease results in over 50 million cases worldwide per year, with the majority being unvaccinated individuals present in Third World Countries[1].
B. pertussis is spread through coughing and sneezing and symptoms first appear seven to ten days after infection[2]. These symptoms include: fever, runny nose, coughing which develops into a whooping cough, and phenomena[2]. Those infected with B. pertussis are contagious for around three weeks once coughing is displayed as a symptom yet symptoms can last up to eight weeks[2]. Most commonly infants under two months are the most vulnerable and have the highest mortality rate[3]. In infants symptoms include: gagging, gasping, bradycardia, cyanosis, and vomiting[4]. Mortality in infants most commonly is from leukocytosis, pulmonary hypertension, and pneumonia[4]. Treatment for whooping cough consists of antibiotic therapy and intubation with ventilation and oxygen[4].
B. pertussis was first discovered and isolated to be the cause of whooping cough in 1906 [1]. Compared to human infectious diseases, B. pertussis is a relatively recent human infectious disease that first appeared in France in 1414[5]. The disease was identified in Paris, France in 1900 by Jules Bordet and Octave Gengou who obtained the sample from a 5-month-old child[5]. The bacteria was finally isolated in 1906 from Jules Bordet’s own son[1]. A vaccine for B. pertussis was developed in the early 1940s for widespread population use [6]. Before the development of the vaccine there were over 200,000 cases in the United States reported annually and with widespread use incidence has decreased 75% compared to the pre-vaccination era[6]. Currently the vaccine is 80% effective in preventing whooping cough infections and preventing severe disease and deaths [7]. There are two vaccines that currently protect children and infants from whooping cough: DTaP and Tdap but they do not offer lifetime protection [8]. Whooping cough is still seen as a growing epidemic with a decrease in vaccination rates and the seriousness of the disease[6]. Currently, there are 24 million new pertussis cases globally in children under the age of 5 resulting in 160,700 deaths[6]. Vaccines and natural acquired immunity are the two ways to prevent infection against B. pertussis [8]. Being vaccinated for B. pertussis can lead to less infections and a smaller mortality rate.
Bordetella pertussis and Infection Stages
B. pertussis is a gram-negative, aerobic coccobacillus bacteria a part of the genus Bordetella[5]. B. pertussis produces antigenic and biologically active products: pertussis toxin, filamentous hemagglutinin, agglutinogens, adenylate cyclase, pertactin, and tracheal cytotoxin[6]. An immune response to one of these products will result in immunity after infection[6].Pertussis is a human-specific pathogen causing severe respiratory disease that cannot survive outside of its host[5]. Infection occurs with B. pertussis entering through the host airway via respiratory droplets from an already infected individual, usually from coughing [9]. B. pertussis then proceeds down the respiratory tract and adheres to ciliated epithelial cells in the trachea and nasopharynx [9]. Once the bacteria is attached it begins to replicate and colonies with-in the host cells [9]. Toxins are secreted by micro-organism which inflict damage to the epithelial lining, resulting in loss of ciliated cells which induces the coughing most commonly associated with whooping cough [9]. The pertussis toxins released also allow the bacteria to avoid host immune response by interfering with clearance mechanisms [9]. The damage caused by the toxins released by the replicating bacteria also halts ciliary function, short-circuiting host G proteins signaling apparatus, and also inhibits immune cell functions by up regulating cAMP levels [9].
The infection of B. pertussis has an incubation period of seven through ten days but a range of four through 21 days [6]. Infection consist of three stages: catarrhal, paroxysmal, and convalescent[6]. Catarrhal stage starts the onset of symptoms including runny nose, sneezing, and a minor cough [6]. One to two weeks later the cough becomes more severe and the disease progresses into the paroxysmal stage [6]. A fever is either not present or has minimal presence during infection at any stage [6]. The paroxysmal stage is commonly when whooping cough is diagnosed due to numerous, rapid coughs due to thick mucus building up [6]. The end of the paroxysmal stage begins with a high-pitched whoop and difficulty breathing where the infected individual may become cyanotic (blue or purple discoloration due to lack of oxygen)[6]. These symptoms usually occur as attacks rather than continuous symptoms [6]. They occur more often during the night and may reach 15 attacks in 24 hours[6]. This stage may last anywhere from one week to 10 weeks [6]. The convalescent stage consists of a gradual recovery yet paroxysmal symptoms may reoccur and cause respiratory infections for months after diagnosis [6].
B. Pertussis Vaccine History

Soon after B. pertussis was isolated as the causative agent of whooping cough, Pertussis whole-cell (Pw) vaccines were implemented using heat-killed bacteria [5]. Starting in 1914 the first vaccine was licensed in the United States using whole killed bacteria but was used more for treatment than prevention [10]. The problem with early Pw vaccines is they killed whole cell organisms which contained endotoxins that could cause serious side effects[11]. Soon after the Pw vaccine was first discovered, new research and diagnostic tools were used to continue researching for more effective and safe vaccines. Through 1934-1935 Pearl Kendrick and Grace Eldering ran clinical trials on children using a whole-cell vaccine that was generated from chemically inactive bacteria instead of just heat killed bacteria [7]. The vaccine showed an efficacy rate of 89% and was widely distributed [7]. Pw vaccines became used for wholesale distribution starting in the 1940’s [12]. Since B. pertussis in human host restrictive, there is a very low display of genetic diversity [5]. Recent analyses have concluded that there were several different types of isolates circulating in the pre-vaccine era that produce various proteins [5]. Only one or two types predominated with no significant difference in an associated pattern to which was the dominant isolate [5]. This allowed for the Pw vaccine to be composed of 1-3 strains selected from the predominant isolate types [5]. Pw vaccines were given to infants and toddlers extensively due to the majority of infections occurring in younger children [5]. Where vaccine distribution was effective, vaccine strains of B. pertussis were low and in areas with low vaccination rates the pre-vaccine era isolated were still circulating [5]. Pw vaccines were shown to create immunity for some isolates of B. pertussis but not all isolates [5].
By 1943 the American Academy of Pediatrics (ACIP) allowed for routine use in all children [7]. In 1948 the DTP vaccine, which contained diphtheria, tetanus, and whole-cell pertussis vaccine) was created as used effectively[7]. By the 1980s B. pertussis infections had fallen 99% and the fear of infection was replaced into the fear of the vaccine’s side effects[7]. Common side effects included: fever, crying, febrile seizures, limb redness and swelling, and hypo-responsive episodes[7]. The side effects did not cause extreme harm but the public began linking the vaccine to sudden infant death syndrome (SIDS), asthma, and encephalopathies[7]. Through the 1970-8os there was a decrease in vaccination rates due to countries recalling the vaccine due to safety reasons or lack of parental approval [10]. Another problem with wP vaccines was the amount of biologically active components of the vaccine were unclear and made researching the efficacy and effectiveness of the vaccine difficult[13].
With the decrease of vaccination and the increase of cases, research began developing an effective acellular vaccine containing B. pertussis antigens rather than heat killed bacteria [7]. This vaccine contained formaldehyde-treated filamentous hemagglutinin and formalin-inactivated pertussis toxin which resulted in a 69% efficacy rate[10]. By the 1990s the use of a combined diphtheria, tetanus, and acellular pertussis vaccine (DTaP) became the most common form of the vaccine in the US[7]. To date, the vaccine has continued to be successful but the length of protection has been ambiguous[7]. Most commonly, research has shown additional doses of the vaccine may be necessary to have immunity against the disease [14]. The vaccine showed an efficacy rate of 89% and was widely distributed [7]. The ACIP and the CDC currently recommends a four to five doses of the DTaP with three during infancy and a booster in pre-school age children or in adolescence[5]. It is also recommended that pregnant women receive an additional dose during pregnancy[15]. Receiving a dose during the third trimester of pregnancy protects the unborn child from contracting whooping cough during the first few months of life[16].
Vaccine Virulence
While B. pertussis vaccines have decreased overall mortality rates by over 90% there is still a current resurgence in whooping cough cases [17]. The two most common reasons for the resurgence of whooping cough is that the aP (acellular whole cell) vaccine has a relative short-term protection rate and the evolution of the B. pertussis pathogen [17]. Current evolution has included the expansion of B. pertussis strains that contain an allele for the pertussis toxin (ptx) promotor ptxP3 that replaces the ptxP1 strains [18]. PtxP3 strains produce more ptx than the ptxP1 stains which increase the virulence factors of B. pertussis that increases the pathogens overall fitness[18]. PtxP3 causing B. pertussis produces more pertussis toxins and suppress the host immune system more effectively than ptxP1[18]. Sarfarchi et al (2016) found that the ptxP3 strains colonized the respiratory tract in vaccinated and unvaccinated mice better than ptxP1 [19]. Along with the ptxP3 strain evolving, new B. pertussis strains can increase expression of a regulatory molecule PD-LI which may produce vaccine-induced protective responses[17]. Recently, there has been an increase in mutations of B. pertussis that are not producing pertactin which is a pertussis acellular vaccine immunogen that is a part of the bacteria adhering to the upper respiratory epithelium [20]. The strains of B. pertussis that do not express pertactin have been increasing in regions where acellular pertussis vaccines have been used for over seven years [20].
As the number of pseudo-genes decreases, B. pertussis seems to adapt to survive their human host and continue to lose genes not
not necessary for human infection[5]. Pw vaccination did not control the circulation of other isolates and did not control virulence of isolates[5]. The increase of aP vaccine induced immunity in the population and new isolates would lack the vaccine antigens due to deletion or inactivation of their structural genes[5]. Work by Hegerle et al. showed that wP vaccines induced immunity led to having one form of B. pertussis whereas aP vaccine-induced immunity lead to increase in the number of B. pertussis isolates not expressing virulence factors [21]. The expression of virulence factors comes from the regulation of the Bordetella virulence gene (bvg) that encodes for two proteins that sense the environmental conditions and act according to the conditions[9]. Spontaneous mutation during DNA replication has also shown to produce avirulent variants of B. pertussis[5]. This form of bacteria does not have the ability to invade and colonise the host[9].Overall the circulation of B. pertussis is likely to decrease but isolates that do not produce vaccine antigens may persist[5].
Another problem with B. pertussis vaccines is the length of time immunity is provided. With the invention of the acellular vaccine patients had less side effects and was overall tolerated but follow-up research indicates a decrease in protection against pertussis[7].The switch from whole-cell vaccines to the acellular vaccine reversed the trend of decreasing infections to an increase of overall B. pertussis infections [22]. When vaccine protection is compared to naturally gained immunity vaccine protection has a shorter length of protection[13]. Along with natural immunity, antibody responses have also supported that immunity decreases after vaccination with the acellular B. pertussis vaccine[13].
The past strategy of reducing cases of whooping cough most likely needs to be re-evaluated to current epidemiological trends. During cyclic outbreaks of B. pertussis older children, adolescents, and adults have made up the majority of cases [7]. The current immunization programs target infants, which has caused a significant decrease in mortality in a vulnerable population[7]. Tdap vaccination was primarily used to reduce the burden of B. pertussis among older populations while also protecting infants from the transmission of the disease[13]. Protection is currently seen as short term and results in a population of other kids/adults that is no longer immune[7]. There is also a switch in family dynamics where families are having a smaller number of children and spending less time with others due to the rising age of technology[7]. This can result in fewer populations experiencing natural immunity of B. pertussis[7].
Why Vaccination?
With widespread use of vaccination there are still close to 48.5 million yearly cases and 250,000 deaths [12]. This partially surfaces from Incomplete immunity in infants who have received fewer than three doses and have relatively short-lived immunity [12]. The biggest worries towards B. pertussis to become epidemiological is due to adults and young adults being a major source of transmission and a reservoir[12]. Vaccination coverage with DTaP remains at a 95% rate for children 19-35 months high infants younger than 6 month have the highest rate of infection at 72.3% per 100,000 pertussis cases (Havers). This high number of cases is the result of infants being too young to be completely vaccinated for B. pertussis [23]. Studies by Greef et al (2010) showed that infants hospitalized with whooping cough were primarily infected by their siblings or their mother[23]. Adults, vaccinated and unvaccinated, can spread it to infants which causes a more life-threatening disease[23]. Most commonly infants get whooping cough from an adult individual with a cough illness that is not recognized as pertussis [11]. Infants that contract B. pertussis may develop pneumonia, uncontrollable shaking, and brain damage whereas young adults and adults may only develop cold-like symptoms[16]. It is recommended that anyone who is planning to be around a newborn child in the first few months of life be vaccinated for B. pertussis [16]
B. pertussis has shown to follow a cyclic pattern with peaks of infection every 2 to 5 years regardless of vaccination [11]. This cyclic pattern occurred during whole-cell pertussis vaccination era and with the present acellular vaccine era[4]. Along with a pattern of infection, the increase of pertussis cases coincides with the transition to acellular vaccines in the United States[6]. The newer vaccine displays less side effects than the Pw vaccine but has a lack of long term immunity[7]. While vaccines are highly successful, immunity is not lasting[4].
Widespread vaccinations have mainly occurred in developed first world countries. Mortality rates are still commonly reported from underdeveloped countries where mass immunization programs are not readily available[24]. Immunization coverage for infants in developing countries reached 80% of infants having at least three doses of the DPT vaccine[25]. The reason for the lack of strong vaccination rates in developing countries has been placed on weak management of immunization services and missing opportunities to immunize eligible children[25]. There is also ineffective communication and information given to mothers to complete the immunization series due to a lack of motivation[25].
Conclusion
Overall B. pertussis infections have decreased significantly since the discovery of the pathogen and the invention of effective and safe vaccines. Whooping cough is still a significant disease affecting mainly infants who are too young to receive a vaccine. Vaccines have been shown to give direct protection to vaccinated individuals while also displaying an indirect effect of protection for unvaccinated individuals in a population[26]. In the case of B. pertussis, individuals getting vaccinated, especially other age groups, can create an indirect effect of protection on infants and younger children who are unable to be vaccinated. Most industrialized countries have been able to control B. pertussis infections but due to whooping cough being seen as a minor problem and pressure from anti-immunization groups have caused fluctuations in vaccination rates[25]
More research is needed on the true effectiveness of the DTaP vaccine in terms of lasting immunity for B. pertussis. Research could include finding a way to incorporate the effectiveness of immunity of pertussis whole-cell vaccines with the safety of acellular pertussis vaccines to create a safe and long lasting vaccine. Research could be conducted on why infants and young children have more severe symptoms to B. pertussis infection than young adults and adults. This would create a better understanding of rates of infection and how the disease works as a pathogen in the presence of vaccines. More research could be completed on why immunity is not long term after vaccination or natural immunity from already having B. pertussis. This would allow for more understanding on how populations can protect themselves even after being vaccinated. A potential benefit that comes from more research on effectiveness and lasting immunity is to continue education on vaccination and the causative effects of the disease. Public influence is a large part of how individuals make decisions so providing accurate resources that education concisely would be a benefit towards controlling new infections. The primary fear being parents not vaccinating their children for B. pertussis is due to misconceptions about side effects of the vaccine. B. pertussis is a preventable disease where spread can be limited by vaccination from a young age and would decrease the amount of lives lost each year. The main goal of any person should be to protect others who are more susceptible to B. pertussis. Infants and small children with zero doses or one to two doses are extremely susceptible and can experience extreme symptoms. Protecting the susceptible allows for a decrease in mortality rates and infections.
References
- ↑ 1.0 1.1 1.2 1.3 Kerr, J.R. and Matthews, R.C."Bordetella pertussis Infection: Pathogenesis, Diagnosis, Management, and the Role of Protective Immunity." 2000. European Journal of Clinical Microbiology and Infectious Disease 19:77-88.
- ↑ 2.0 2.1 2.2 World Health Organization: Pertussis 2018
- ↑ Kapil, P. Merkel, T.J. "Pertussis vaccines and protective immunity" Current Opinion in Immunology. 59:72-78
- ↑ 4.0 4.1 4.2 4.3 4.4 "Pertussis in Young Infants Throughout the World " Clinical Infectious Diseases. 63:S119-S122
- ↑ 5.00 5.01 5.02 5.03 5.04 5.05 5.06 5.07 5.08 5.09 5.10 5.11 5.12 5.13 5.14 5.15 5.16 5.17 Guiso, N. "Bordetella pertussis and Pertussis Vaccines." 2009. Clinical infectious Diseases 49:1565-1569.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 6.13 6.14 6.15 6.16 6.17 Havers et al. "Pertussis." 2021. Center for Disease Control and Prevention. 1-16.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 7.13 7.14 7.15 7.16 7.17 7.18 Kuchar, E. et al. "Pertussis: History of the Disease and Current Prevention Failure" 2016. Advances in Exerimental Medicine and Biology
- ↑ 8.0 8.1 CDC. "Vaccines for your Children" 2022.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 9.7 Smith, A.M et al. "The virulence factors of Bordetella pertussis: a matter of control" 2001. FEMS Microbiology Reviews 25:309-333.
- ↑ 10.0 10.1 10.2 Klein, N.P et al. "Comparative effectiveness of acellular versus whole-cell pertussis vaccines in teenagers" 2013. Pediatrics
- ↑ 11.0 11.1 11.2 Kerr, Cherry J.D."The Epidemiology of Pertussis: A Comparison of the Epidemiology of the Disease pertussis With the Epidemiology of Bordetella pertussis Infection" 2005. American Academy of Pediatrics" 1422-1427.
- ↑ 12.0 12.1 12.2 12.3 Mattoo, S. Cherry, J.D. "Molecular Pathogenesis, Epidemiology, and Clinical Manifestations of Respiratory Infections Due to Bordetella pertussis and Other Bordetella Subspecies" 2005. Clinical Microbiology Reviews
- ↑ 13.0 13.1 13.2 13.3 Chen, Z. He, Q. "Immune persistence after pertussis vaccination" 2017. Human Vaccin Immunother. 13:744-756.
- ↑ Gustafsson, L. et al. "Long-term Follow-up Swedish Children Vaccinated With Acellular Pertussis Vaccines at 3, 5, and 12 Months of Age Indicates the Need for a Booster Dose at 5 to 7 Years of Age" 2006. American Academy of Pediatrics. 978-984
- ↑ Liang, J.L et al. "Prevention of Pertussis, Tetanus, and Diphtheria with Vaccines in the United States: Recommendations of the Advisory Committtee Immunization Practices (ACIP)" 2018.Recommendations and Reports
- ↑ 16.0 16.1 16.2 Clinic. "Pertussis(Whooping Cough)Vaccine" 2022
- ↑ 17.0 17.1 17.2 Dorji, D. et al. "Bordetella pertussis virulence factors in the continuing evolution of whooping cough vaccines for improved performance." 2018. Medical Microbiology and Immunology 207:3-26.
- ↑ 18.0 18.1 18.2 King A.J. et al. "Genome-Wide Gene Expression Analysis of Bordetella pertussis Isolates Associated with a Resurgence in Pertussis: Elucidation of Factors Involved in the Increased Fitness of Epidemic Strains." 2013. PLOS Computational Biology.
- ↑ Safarchi, A. et al. "Better colonisation of newly emerged Bordetella pertussis in the co-infection mouse model study" 2016. Vaccine 34:3967-3971.
- ↑ 20.0 20.1 Bodilis, H and Guiso, N."Virulence of Pertactin-Negative Bordetella pertussis Isolates from Infants, France" 2013. Emerging Infectious Disease 19:471-474.
- ↑ Hegerle, N. et al. "Evolution of French Bordetella pertussis and Bordetella parapertussis isolates: increase of Bordetellae not expressing pertactin" 2012. Clinical Microbiology and Infection 18:E340-E346.
- ↑ [https://journals.lww.com/pidj/fulltext/2015/09000/Pertussis_Across_the_Globe__Recent_Epidemiologic.23.aspx/ Tan, T. et al. "Pertussis Across the Globe Recent Epidemiologic Trends From 2000 to 2013" 2015. The Pediatric Infectious Disease Journal 34:e222-e232.]
- ↑ 23.0 23.1 23.2 Greef S.C de et al."Pertussis Disease Burden in the Household: How to Protect Young Infants" 2010. Clinical Infectious Disease. 50:1339-1345
- ↑ M."Pertussis vaccines: present status" 1990. Advances in Biotechnological Processes. 13:169-199
- ↑ 25.0 25.1 25.2 25.3 Galazka, A."Control of pertussis in the world" 1992. World Heath Stat Q.. 45:238-247
- ↑ Doherty et al. "Vaccine impact: Benefits for human health" 2016. Vaccine 34: 6707-6714
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski, 2022, Kenyon College
