Methanosarcina barkeri: Difference between revisions
| Line 79: | Line 79: | ||
[http://www.sciencemag.org/cgi/content/abstract/315/5814/1003 Sato, T, Atomi, H, and Imanaka, T. "Archaeal Type III RuBisCOs Function in a Pathway for AMP Metabolism". Science. 2007. 315-5817, p. 1003.]<br /> | [http://www.sciencemag.org/cgi/content/abstract/315/5814/1003 Sato, T, Atomi, H, and Imanaka, T. "Archaeal Type III RuBisCOs Function in a Pathway for AMP Metabolism". Science. 2007. 315-5817, p. 1003.]<br /> | ||
Types I and II RuBisCO are found in all phototrophic organisms and are essential enzymes for the carbon fixation of the Calvin cycle. Type IV RuBisCO is found in ''Bacillus subtilis'' and is part of the Met salvage pathway. This paper describes the characterization of Type III RuBisCO, which is found only in select archaea, including ''Methanosarcina barkeri''. It was dtermined that Type III RuBisCO catalyzes the reaction of ribulose-1,5-bisphosphate to two molecules 3-phosphoglycerate, which can then enter either glycolysis or gluconeogenesis. This is mediated via two other enzymes which break down AMP into the ribulose-1,5-bisphosphate.<br /><br /> | |||
[http://doi.wiley.com/10.1002/prot.21262 Chu, HM and Andrew, HJW. "Enzyme-Substrate Interactions Revealed by the Crystal Structures of the Archaeal Sulfolobus PTP-Fold Phosphatase and its Phosphopeptide Complexes". PROTEINS: Structure, Function, and Bioinformatics. 2007. 66, p. 996-1003.]<br /> | [http://doi.wiley.com/10.1002/prot.21262 Chu, HM and Andrew, HJW. "Enzyme-Substrate Interactions Revealed by the Crystal Structures of the Archaeal Sulfolobus PTP-Fold Phosphatase and its Phosphopeptide Complexes". PROTEINS: Structure, Function, and Bioinformatics. 2007. 66, p. 996-1003.]<br /> | ||
Revision as of 05:37, 5 June 2007
A Microbial Biorealm page on the genus Methanosarcina barkeri
Classification
Higher order taxa
|
NCBI: Archaea : Euryarchaeota : Methanomicrobia : Methanosarcinales : Methanosarcinaceae : Methanosarcina |
Species
|
NCBI: Methanosarcina barkeri |
Strains
Methanosarcina barkeri str. MS
Methanosarcina barkeri str. W
Methanosarcina barkeri str. 227
Description and significance
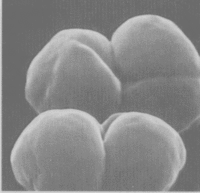
Methansoarcina barkeri is a large, spherical gram-positive anaerobe that forms packets of varying size. The cells have no flagella and are not mobile. The species have been isolated from lake mud samples, sewage sludge, and is suspected to live in the rumen of the cow. It makes its energy by fermenting small carbon sources, involving the oxidation of H2 and reducing CO2 to methane. The CO2 is produced by pathways starting with methanol, methylamaines, or acetate.
M. barkeri has a thick cell wall (500 nm) and is composed of an acid heteropolysaccharide.
Genome structure
Methanosarcina barkeri fusaro has one circular chromosome and one circular plasmid.
Chromosome
GenBank id: CP000099 Refseq id: NC_007355
Size: 4,837,408 bp (69% coding) 39% GC
3811 genes: 3606 proteins, 73 structural RNAs, 132 pseudogenes
Plasmid
GenBank id: CP000098 Refseq id: NC_007349
Size: 36,358 bp (58% coding) 33% GC
20 genes: 18 proteins, 2 pseudogenes
Cell structure and metabolism
Methanosarcina barkeri is a methanogen and produces methane from a variety of carbon sources, including CO2, acetate, and methanol. It is also an anaerobe and is very oxygen sensitive, so all of its energy production pathways involve some sort of fermentation.
Ecology
Methanosarcina barkeri is an anaerobe has been isolated from mud samples in lakes and bogs. M. barkeri also lives in the rumen of cows where it helps digest organic matter for the cow. A USA Today article has reported that up to 17% of the world's atmospheric methane comes from cows, a large majority of which would come from M. barkeri. Because methane is a green house gas and can interfere with the ozone layer, this small organism may be partially responsible for two of the major environmental crises that we have face: thinning of the ozone layer and global warming.
Pathology
Methanosarcina barkeri is an archaea and therefore causes no known diseases.
Application to Biotechnology
As a methanogen, Methanosarcina barkeri has been looked at as a source of methane (natural gas) for use as an energy source.
Current Research
Longstaff, DG and Blight, SK and Zhang, L and Green-Church, KB and Krzycki, JA. "In vivo contextual requirements for UAG translation as pyrrolysine". Molecular Microbiology. 2007. 63-1, p. 229-241.
Pyrrolysine and selenocysteine are coded by a nonstandard translation of normal stop codons. For selenocysteine, it is known that th insertion of the amino acid instead of message termination is dependent on an insertion sequence that causes the formation of secondary structure in the mRNA that favors insertion of selenocysteine. In this paper, the researchers demonstrate experimentally the requirement of a similar mechanism for pyrrolysine. This sequence, which they call the PYrroLysine Insertion Sequence (PYLIS), is located downstream and is not translated. The experiment consisted of the transfer of a pyrrolysine-containing gene from Methanosarcina barkeri to Methanosarcina acetivorans. This is not a definitive study, as other mechanisms may contribute to the insertion of pyrrolysine or termination.
Sato, T, Atomi, H, and Imanaka, T. "Archaeal Type III RuBisCOs Function in a Pathway for AMP Metabolism". Science. 2007. 315-5817, p. 1003.
Types I and II RuBisCO are found in all phototrophic organisms and are essential enzymes for the carbon fixation of the Calvin cycle. Type IV RuBisCO is found in Bacillus subtilis and is part of the Met salvage pathway. This paper describes the characterization of Type III RuBisCO, which is found only in select archaea, including Methanosarcina barkeri. It was dtermined that Type III RuBisCO catalyzes the reaction of ribulose-1,5-bisphosphate to two molecules 3-phosphoglycerate, which can then enter either glycolysis or gluconeogenesis. This is mediated via two other enzymes which break down AMP into the ribulose-1,5-bisphosphate.
Chu, HM and Andrew, HJW. "Enzyme-Substrate Interactions Revealed by the Crystal Structures of the Archaeal Sulfolobus PTP-Fold Phosphatase and its Phosphopeptide Complexes". PROTEINS: Structure, Function, and Bioinformatics. 2007. 66, p. 996-1003.
"The P-loop-containing protein phos-phatases are important regulators in signal transduction. These enzymes have structural and functional similarity with a conserved sequence of Dx(25-41)HCxxGxxR(T/S) essential for catalysis. The singular protein tyrosine phosphatase (PTP) from archaeal Sulfolobus solfataricus is one of the smallest known PTPs with extreme thermostability. Here, we report the crystal structure of this phosphatase and its complexes with two tyrosyl phosphopeptides A-(p)Y-R and N-K-(p)Y-G-N. The structure suggests the minimal structural motif of the PTP family, having two variable sequences inserted between the 2-3 and 3-4 strands, respectively. The phosphate of both phosphopeptide substrates is bound to the P-loop through several hydrogen bonds. Comparison of several phosphatase-substrate complexes revealed that Gln135 on the Q-loop has different modes of recognition toward phosphopeptides. The substrate specificity of SsoPTP is primarily localized at the phosphotyrosine, suggesting that this phosphatase may be a prototypical PTP."
Ambrogelly, A, Gundllapalli, S, Herring, S, Polycarpo, C, Frauer, C, and Soll, D. "Pyrrolysine is not hardwired for cotranslational insertion at UAG codons". Proceeds of the National Academy of Sciences. 2007. 104-9, p. 3141-3146.
"Pyrrolysine (Pyl), the 22nd naturally encoded amino acid, gets acylated to its distinctive UAG suppressor tRNA(Pyl) by the cognate pyrrolysyl-tRNA synthetase (PylRS). Here we determine the RNA elements required for recognition and aminoacylation of tRNA(Pyl) in vivo by using the Pyl analog N-epsilon-cyclopentyloxycarbonyl-l-lysine. Forty-two Methanosarcina barkeri tRNA(Pyl) variants were tested in Escherichia coli for suppression of the lac amber A24 mutation; then relevant tRNA(Pyl) mutants were selected to determine in vivo binding to M. barkeri PylRS in a yeast three-hybrid system and to measure in vitro tRNA(Pyl) aminoacylation. tRNA(Pyl) identity elements include the discriminator base, the first base pair of the acceptor stem, the T-stem base pair G51:C63, and the anticodon flanking nucleotides U33 and A37. Transplantation of the tRNA(Pyl) identity elements into the mitochondrial bovine tRNA(Ser) scaffold yielded chimeric tRNAs active both in vitro and in vivo. Because the anticodon is not important for PylRS recognition, a tRNA(Pyl) variant could be constructed that efficiently suppressed the lac opal U4 mutation in E. coli. These data suggest that tRNA(Pyl) variants may decode numerous codons and that tRNA(Pyl):PylRS is a fine orthogonal tRNA:synthetase pair that facilitated the late addition of Pyl to the genetic code."
Feist, AM, Scholten, JCM, Palsson, BO, Brockman, FJ, and Ideker, T. "Modeling methanogenesis with a genome-scale metabolic reconstruction of Methanosarcina barkeri". Molecular Systems Biology. 2006. vol. 2-1.
"Methanogenesis is a unique way of life for a group of archaea (methanogens) that generate energy by converting simple substrates such as acetate, methanol or H2/CO2 to methane. Because of this, methanogens serve as a key component of the carbon cycle by degrading low carbon molecules in a number of anaerobic environments. The methane they produce contributes to the greenhouse effect and is a potential source of renewable energy. In addition, some methanogens can form syntrophic relationships with other microorganisms, making them an interesting target for the study of interactions between different organisms. Although many pieces of methanogenic metabolism are understood, there are still many questions to be answered about the biochemistry of methanogenesis and how these pieces work together in the context of the whole organism. To address these questions, we reconstructed a genome-scale metabolic network for one of the most versatile methanogens, Methanosarcina barkeri, and analyzed the network to determine biochemical properties of key components and methanogenic metabolism as a whole."
References
Balch, WE, Fox, GE, Magrum, LJ, Woese, CR, and Wolfe, RS. "Methanogens: Reevaluation of a Unique Biological Group". Microbiological Reviews. June 1979. p. 260-296.
Maeder DL, Anderson I, Brettin TS, Bruce DC, Gilna P, Han CS, Lapidus A, Metcalf WW, Saunders E, Tapia R, Sowers KR. "The Methanosarcina barkeri Genome: Comparative Analysis with ...". Journal of Bacteriology. 2006. 188-22, p. 7922-7931.
Stadtman, TC and Barker, HA. "STUDIES ON THE METHANE FERMENTATION IX. The Origin of Methane in the Acetate and Methanol Fermentations by Methanosarcina". Journal of Bacteriology. 1951. 61-1, p. 81-86.
"Cutting cattle's methane emissions". USA Today (Magazine). June 01, 2002.
Atkins, JF and Gesteland, R. "The 22nd Amino Acid". Science. 2002. 296-5572, p. 1409-1410.
TIGR Comprehensive Microbial Resource: Methanosarcina barkeri
US Department of Energy Joint Genome Institute Organism Detail page for Methanosarcina barkeri
TIGR Comprehensive Microbial Resource: Methanosarcina barkeri fusaro Genome Page
Edited by Ian Kerman, a student of Rachel Larsen at UCSD.