Shewanella woodyi NEU Spring 2012: Difference between revisions
| (54 intermediate revisions by the same user not shown) | |||
| Line 7: | Line 7: | ||
==Description and significance== | ==Description and significance== | ||
[[File:shewa.jpg|200px|thumb|left|Figure 1: Polar | [[File:shewa.jpg|200px|thumb|left|Figure 1: Polar Flagellum [1]]] | ||
''Shewanella woodyi'' is a gram negative bacilli shaped bacterium between | Named after J. Woodland Hastings, a famous American biologist recognized for his work about bacterial bioluminescence, ''Shewanella woodyi'' is a gram negative bacterium found to have some unique characteristics and play a couple of important environmental roles [1,4]. It is a bacilli shaped bacterium and is generally between one to two micrometers in length [1,4]. ''S. woodyi'' is entirely marine and can be found in deep-sea environments therefore making it barophilic (tolerant to high pressure) [1,2]. Although other species within the Shewanella genus are psychrophilic, ''S. woodyi'' is instead mesophilic growing at an optimal temperature around 25°C [1,2]. Each ''S. woodyi'' bacterial cell possesses a single polar flagellum making it motile, as seen in Figure 1 [1]. This bacterium is also luminescent, meaning it can emit light[1]. | ||
It is well known that the ocean acts as a sink for harmful compounds from both terrestrial and aquatic environments. ''S. woodyi'' is significant in that it helps reduce the levels of select toxic compounds that collect in the sediment on the ocean floor [2,6]. Additionally, this bacterium also plays a role in carbon and nitrogen cycling through its anaerobic capabilities [2,6,7]. | It is well known that the ocean acts as a sink for harmful compounds from both terrestrial and aquatic environments. ''S. woodyi'' is significant in that it helps reduce the levels of select toxic compounds that collect in the sediment on the ocean floor [2,6]. Additionally, this bacterium also plays a role in carbon and nitrogen cycling through its anaerobic capabilities [2,6,7]. | ||
| Line 17: | Line 17: | ||
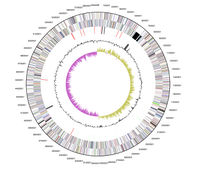
[[File:ChromosomalMap.jpg|200px|thumb|right|Figure 2: Chromosomal Map [3]]] | [[File:ChromosomalMap.jpg|200px|thumb|right|Figure 2: Chromosomal Map [3]]] | ||
<i> S. woodyi</i> has a circular DNA chromosome of 5,935,403 bases, of which 86.02% are coding bases. This chromosome has a G+C content of 43.7%. The entire chromosome can be seen in Figure 2. These base pairs code for 5,085 genes in total, with 96.81%, or 4,923 genes, coding for proteins. The remaining 3.19%, or 162 genes, code for RNA. | <i> S. woodyi</i> has a circular DNA chromosome of 5,935,403 bases, of which 86.02% are coding bases. This chromosome has a G+C content of 43.7%. The entire chromosome can be seen in Figure 2. These base pairs code for 5,085 genes in total, with 96.81%, or 4,923 genes, coding for proteins. The remaining 3.19%, or 162 genes, code for RNA. [3] | ||
==Cell structure and metabolism== | ==Cell structure and metabolism== | ||
'' | ''S. woodyi'' is a gram negative, luminescent, nonsporulating rod (0.4-1.0 by 1.4-2.0 pm) with an unsheathed flagellum as was mentioned above. For this bacteria's metabolism, it is catalase and oxidase positive and most strains produce amylase. All strains do produce gelatinase but are unable to make lipase, chtinase, and agarase. ''S. woodyi'' is able to metabolize D-galactose, cellobiose, D-glucuronic acid, acetate, a-ketoglutarate,propionate, succinate, L-alanine, L-threonine, L-leucine, L-serine, and putrescine. Oxygen and nitrite are used by these cells as electron acceptors for their metabolic processes. Also, ''S. woodyi'' is a respiratory bacteria, which means that it is non-fermentative. This bacteria is involved in carbon and nitrogen fixation in its marine environment and contributes to the biodegradation of pollutants. [1] | ||
==Ecology== | ==Ecology== | ||
| Line 36: | Line 36: | ||
==Current Research== | ==Current Research== | ||
An article posted to Decoded Science on January 21, 2012 showed how ''S.woodyi'' was used in a study about how bacteria go about making biofilms. This involves quorum sensing, how the cells communicate with each other, and biofilm formation and bioluminescence are used for this research. It is agreed upon that a signaling molecule is involved called bis-(3’-5’)-cyclic dimeric guanosine monophosphate (c-di-GMP), but this study showed how nitric oxide, which is a signaling molecule in mammals, is also a regulator in bacteria impacting biofilms. ''S. woodyi'' has a gene called H-NOX near the c-di-GMP gene, and they directly interact. The H-NOX gene is heme-nitic oxide/oxygen binding, so when there is nitric oxide, c-di-GMP is degraded. The absence of nitric oxide up-regulates the c-di-GMP activity. When the concentration of the c-di-GMP goes up, bacteria enter biofilms. This was the first study to show how nitric oxide plays a role in c-di-GMP metabolism. [9] | |||
Currently, there is little research available about this bacteria, but a study was done in 2008 about the horizontal gene transfer of the lux gene, which encodes the proteins involved in luminescence. Horizontal gene transfer plays a huge role in the evolution of bacteria, and genera in the family, ''Vibrionaceae'' (Gammaproteobacteria), were studied including ''Shewanella'' (''Shewanella woodyi'' and ''Shewanella hanedai'') to show a pattern of incidence of the lux gene and how it must have been started in an ancestor of the ''Vibrionaceae'' and then was horizonatally transfered to members of ''Shewanellaceae'' and ''Enterobacteriaceae''. Since most of the species of these families lack the lux gene, the scattered incidence most likely is from horizontal transfer, but this study tried to show some evidence since there has been no phylogenetic tests performed. An evolutionary approach was used to test for the horizontal gene transfer of the lux gene from ''Vibrionaceae''. They examined the incongruence of phylogenies of 300 strains of luminous bacteria based on housekeeping genes and multiple lux genes, and the results showed that horizonatal gene transfer of the lux gene is actually very rare. Surprisingly, the findings from this research lacked evidence for horizontal gene transfer, so it supports the other idea that the scattered incdence of the lux gene may have been due to vertical inheritance with a loss of this gene from multiple lineages. [10] | |||
==Cool Factor== | ==Cool Factor== | ||
| Line 44: | Line 44: | ||
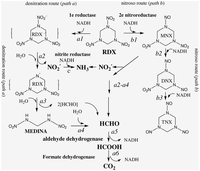
[[File:Rdx pathway 2.png|200px|thumb|left|Figure 3: RDX degradation pathways [6]]] | [[File:Rdx pathway 2.png|200px|thumb|left|Figure 3: RDX degradation pathways [6]]] | ||
As previously mentioned, ''S. woodyi'' plays a role in alleviating the environment of toxic compounds. There are several organisms which also complete similar tasks and remove harmful components from the air or soil, however, not many are capable of degrading compounds such as RDX (hexahydro-1,3,5-trinitro-1,3,5-triazine) along with other explosives [6]. ''S. woodyi'' contains cytochrome c genes and nitroreductases which allow for the break down of RDX into nitric oxide and carbon dioxide [6]. This pathway is shown on the right side of | As previously mentioned, ''S. woodyi'' plays a role in alleviating the environment of toxic compounds. There are several organisms which also complete similar tasks and remove harmful components from the air or soil, however, not many are capable of degrading compounds such as RDX (hexahydro-1,3,5-trinitro-1,3,5-triazine) along with other explosives [6]. ''S. woodyi'' contains cytochrome c genes and nitroreductases which allow for the break down of RDX into nitric oxide and carbon dioxide [6]. This pathway is shown on the right side of Figure 3 [6]. Although ''Shewanella halifaxensis'' and ''Shewanella sediminis'' are more efficient at breaking down RDX than ''S. woodyi'' and can use both degradation pathways shown in Figure 3, ''S. woodyi'' is still capable of this activity making it unique from many other microorganisms [6]. This factor also makes it even more important to the countless animals that depend on non-polluted waters for better survival. Recent research has suggested that in the presence of explosive compounds, such as RDX, bacteria in the ''Shewanella'' genus, including ''S. woodyi'', actually thrive [6]. | ||
==References== | ==References== | ||
[1] Makemson, J. C., Fulayfil, N. R., Landry, W., Van Ert, L. M., Wimpee, C. F., Widder, E. A. & Case, J. F. (1997). Shewanella woodyi sp. nov., an exclusively respiratory luminous bacterium isolated from the Alboran Sea. Int J Syst Bacteriol 47, 1034-1039. | |||
[2] Kato, C., and Nogi, Y. (2001). Correlation between phylogenetic structure and function: examples from deep-sea Shewanella. FEMS Microbiology Ecology 35, 223-230. | |||
[3] | [1] Makemson, J. C., Fulayfil, N. R., Landry, W., Van Ert, L. M., Wimpee, C. F., Widder, E. A. & Case, J. F. (1997). Shewanella woodyi sp. nov., an exclusively respiratory luminous bacterium isolated from the Alboran Sea. ''Int J Syst Bacteriol'' 47, 1034-1039. | ||
[4] http://www.ncbi.nlm.nih.gov/sites/entrez?db=bioproject&cmd=ShowDetailView&TermToSearch=17455 | |||
[5] Wang, F., Xiao, X., Ou, H., Gai, Y., and Wang, F. (2009). Role and regulation of fatty acid biosynthesis in the response of S. pizotolerans WP3 to different temperatures and pressures. Journal of Bacteriology 191, 2574-2584. | [2] Kato, C., and Nogi, Y. (2001). Correlation between phylogenetic structure and function: examples from deep-sea Shewanella. ''FEMS Microbiology Ecology'' 35, 223-230. | ||
[3] Integrated Microbial Genomes. U.S. Department of Energy Joint Genome Institute. (2008). Shewanella Woodyi MS32, ATCC 51908. From: | |||
http://img.jgi.doe.gov/cgi-bin/w/main.cgi?section=TaxonDetail&page=taxonDetail&taxon_oid=641522649 | |||
[4] National Center for Biotechnology Information. (2008). Shewanella woodyi ATCC 51908: A luminous marine bacteria isolated from squid ink. From: | |||
http://www.ncbi.nlm.nih.gov/sites/entrez?db=bioproject&cmd=ShowDetailView&TermToSearch=17455 | |||
[5] Wang, F., Xiao, X., Ou, H., Gai, Y., and Wang, F. (2009). Role and regulation of fatty acid biosynthesis in the response of S. pizotolerans WP3 to different temperatures and pressures. ''Journal of Bacteriology'' 191, 2574-2584. | |||
[6] Zhao, J., Deng, Y., Manno, D., and Hawari, J. (2010). Genomic evolution for a cold marine lifestyle and in-situ explosive biodegration. PLoS ONE 5, online. | [6] Zhao, J., Deng, Y., Manno, D., and Hawari, J. (2010). Genomic evolution for a cold marine lifestyle and in-situ explosive biodegration. PLoS ONE 5, online. | ||
[ | [7] Nealson, K.H., and Scott, J. Ecophysiology of the Genus Shewanella. (2006). ''Prokaryotes'' 6, 1133-1151. | ||
[8] Li, H., Qiao, G., Li, Q., Zhou, W., Won, K. M., Xu, D.-H. and Park, S.-I. (2010), Biological characteristics and pathogenicity of a highly pathogenic <i>Shewanella marisflavi</i> infecting sea cucumber, <i>Apostichopus japonicus</i>. ''Journal of Fish Diseases'', 33: 865–877. | |||
[9] Stone, Marcia. (2012) Nitric Oxide: A Newly Discovered Potential Target for Biofilm Control. Published on Decoded Science on January 21, 2012. From: http://www.decodedscience.com/nitric-oxide-a-newly-discovered-potential-target-for-biofilm-control/9730 | |||
[10] Urbanczyk, H., Ast, J.C., Kaeding, A. J., Oliver, J. D., and Dunlap, P. D. (2008). Phylogenetic Analysis of the Incidence of lux Gene Horizontal Transfer in Vibrionaceae. ''J. Bacteriol.''190:10, 3494-350. From: doi: 10.1128/JB.00101-08 | |||
Latest revision as of 02:32, 28 February 2012
A Microbial Biorealm page on the genus Shewanella woodyi NEU Spring 2012
Classification
Bacteria; Proteobacteria; Gammaproteobacteria; Alteromonadales; Shewanellaceae; Shewanella; woodyi [3]
Description and significance
Named after J. Woodland Hastings, a famous American biologist recognized for his work about bacterial bioluminescence, Shewanella woodyi is a gram negative bacterium found to have some unique characteristics and play a couple of important environmental roles [1,4]. It is a bacilli shaped bacterium and is generally between one to two micrometers in length [1,4]. S. woodyi is entirely marine and can be found in deep-sea environments therefore making it barophilic (tolerant to high pressure) [1,2]. Although other species within the Shewanella genus are psychrophilic, S. woodyi is instead mesophilic growing at an optimal temperature around 25°C [1,2]. Each S. woodyi bacterial cell possesses a single polar flagellum making it motile, as seen in Figure 1 [1]. This bacterium is also luminescent, meaning it can emit light[1].
It is well known that the ocean acts as a sink for harmful compounds from both terrestrial and aquatic environments. S. woodyi is significant in that it helps reduce the levels of select toxic compounds that collect in the sediment on the ocean floor [2,6]. Additionally, this bacterium also plays a role in carbon and nitrogen cycling through its anaerobic capabilities [2,6,7].
Genome structure
S. woodyi has a circular DNA chromosome of 5,935,403 bases, of which 86.02% are coding bases. This chromosome has a G+C content of 43.7%. The entire chromosome can be seen in Figure 2. These base pairs code for 5,085 genes in total, with 96.81%, or 4,923 genes, coding for proteins. The remaining 3.19%, or 162 genes, code for RNA. [3]
Cell structure and metabolism
S. woodyi is a gram negative, luminescent, nonsporulating rod (0.4-1.0 by 1.4-2.0 pm) with an unsheathed flagellum as was mentioned above. For this bacteria's metabolism, it is catalase and oxidase positive and most strains produce amylase. All strains do produce gelatinase but are unable to make lipase, chtinase, and agarase. S. woodyi is able to metabolize D-galactose, cellobiose, D-glucuronic acid, acetate, a-ketoglutarate,propionate, succinate, L-alanine, L-threonine, L-leucine, L-serine, and putrescine. Oxygen and nitrite are used by these cells as electron acceptors for their metabolic processes. Also, S. woodyi is a respiratory bacteria, which means that it is non-fermentative. This bacteria is involved in carbon and nitrogen fixation in its marine environment and contributes to the biodegradation of pollutants. [1]
Ecology
Originally found in squid ink, ocean floor sediment, and among detritus from the Alboran Sea, this bacterium can be found in a wide range of areas [1]. It thrives at 25°C in a deep-sea environment where it can use detritus and other compounds present for energy [1,2,7]. Research in the past few years has suggested that these bacteria can use almost any electron acceptor for anaerobic respiration [7]. The only criteria seems to be that the electron acceptor must be more electronegative than sulfate [7].
High pressure and cold temperatures are two major factors that S. woodyi must overcome in its environment. Usually under these conditions, cell membranes become very rigid and stiff. S. woodyi avoids this problem by producing large amounts of polyunsaturated fatty acids in order to create a very lipid rich membrane [2,5]. This characteristic allows for a more fluid membrane even under high pressures and lower temperatures.
Many of these characteristics that S. woodyi have acquired to live in their environment are a result of horizontal gene transfer from other species [6]. For example, S. woodyi inherited its Na+/H+ antiporter pump from other bacteria that were already expressing that particular gene [6]. As a result, this bacteria is able to regulate the concentration of sodium in the cell in order to live in high salinity waters.
Pathology
While other species of Shewanella are known pathogens (ie. Shewanella marisflavi isolate AP629 is pathogenic to sea cucumber), S. woodyi has not proven to be a pathogenic organism. [8]
Current Research
An article posted to Decoded Science on January 21, 2012 showed how S.woodyi was used in a study about how bacteria go about making biofilms. This involves quorum sensing, how the cells communicate with each other, and biofilm formation and bioluminescence are used for this research. It is agreed upon that a signaling molecule is involved called bis-(3’-5’)-cyclic dimeric guanosine monophosphate (c-di-GMP), but this study showed how nitric oxide, which is a signaling molecule in mammals, is also a regulator in bacteria impacting biofilms. S. woodyi has a gene called H-NOX near the c-di-GMP gene, and they directly interact. The H-NOX gene is heme-nitic oxide/oxygen binding, so when there is nitric oxide, c-di-GMP is degraded. The absence of nitric oxide up-regulates the c-di-GMP activity. When the concentration of the c-di-GMP goes up, bacteria enter biofilms. This was the first study to show how nitric oxide plays a role in c-di-GMP metabolism. [9]
Currently, there is little research available about this bacteria, but a study was done in 2008 about the horizontal gene transfer of the lux gene, which encodes the proteins involved in luminescence. Horizontal gene transfer plays a huge role in the evolution of bacteria, and genera in the family, Vibrionaceae (Gammaproteobacteria), were studied including Shewanella (Shewanella woodyi and Shewanella hanedai) to show a pattern of incidence of the lux gene and how it must have been started in an ancestor of the Vibrionaceae and then was horizonatally transfered to members of Shewanellaceae and Enterobacteriaceae. Since most of the species of these families lack the lux gene, the scattered incidence most likely is from horizontal transfer, but this study tried to show some evidence since there has been no phylogenetic tests performed. An evolutionary approach was used to test for the horizontal gene transfer of the lux gene from Vibrionaceae. They examined the incongruence of phylogenies of 300 strains of luminous bacteria based on housekeeping genes and multiple lux genes, and the results showed that horizonatal gene transfer of the lux gene is actually very rare. Surprisingly, the findings from this research lacked evidence for horizontal gene transfer, so it supports the other idea that the scattered incdence of the lux gene may have been due to vertical inheritance with a loss of this gene from multiple lineages. [10]
Cool Factor
As previously mentioned, S. woodyi plays a role in alleviating the environment of toxic compounds. There are several organisms which also complete similar tasks and remove harmful components from the air or soil, however, not many are capable of degrading compounds such as RDX (hexahydro-1,3,5-trinitro-1,3,5-triazine) along with other explosives [6]. S. woodyi contains cytochrome c genes and nitroreductases which allow for the break down of RDX into nitric oxide and carbon dioxide [6]. This pathway is shown on the right side of Figure 3 [6]. Although Shewanella halifaxensis and Shewanella sediminis are more efficient at breaking down RDX than S. woodyi and can use both degradation pathways shown in Figure 3, S. woodyi is still capable of this activity making it unique from many other microorganisms [6]. This factor also makes it even more important to the countless animals that depend on non-polluted waters for better survival. Recent research has suggested that in the presence of explosive compounds, such as RDX, bacteria in the Shewanella genus, including S. woodyi, actually thrive [6].
References
[1] Makemson, J. C., Fulayfil, N. R., Landry, W., Van Ert, L. M., Wimpee, C. F., Widder, E. A. & Case, J. F. (1997). Shewanella woodyi sp. nov., an exclusively respiratory luminous bacterium isolated from the Alboran Sea. Int J Syst Bacteriol 47, 1034-1039.
[2] Kato, C., and Nogi, Y. (2001). Correlation between phylogenetic structure and function: examples from deep-sea Shewanella. FEMS Microbiology Ecology 35, 223-230.
[3] Integrated Microbial Genomes. U.S. Department of Energy Joint Genome Institute. (2008). Shewanella Woodyi MS32, ATCC 51908. From:
http://img.jgi.doe.gov/cgi-bin/w/main.cgi?section=TaxonDetail&page=taxonDetail&taxon_oid=641522649
[4] National Center for Biotechnology Information. (2008). Shewanella woodyi ATCC 51908: A luminous marine bacteria isolated from squid ink. From: http://www.ncbi.nlm.nih.gov/sites/entrez?db=bioproject&cmd=ShowDetailView&TermToSearch=17455
[5] Wang, F., Xiao, X., Ou, H., Gai, Y., and Wang, F. (2009). Role and regulation of fatty acid biosynthesis in the response of S. pizotolerans WP3 to different temperatures and pressures. Journal of Bacteriology 191, 2574-2584.
[6] Zhao, J., Deng, Y., Manno, D., and Hawari, J. (2010). Genomic evolution for a cold marine lifestyle and in-situ explosive biodegration. PLoS ONE 5, online.
[7] Nealson, K.H., and Scott, J. Ecophysiology of the Genus Shewanella. (2006). Prokaryotes 6, 1133-1151.
[8] Li, H., Qiao, G., Li, Q., Zhou, W., Won, K. M., Xu, D.-H. and Park, S.-I. (2010), Biological characteristics and pathogenicity of a highly pathogenic Shewanella marisflavi infecting sea cucumber, Apostichopus japonicus. Journal of Fish Diseases, 33: 865–877.
[9] Stone, Marcia. (2012) Nitric Oxide: A Newly Discovered Potential Target for Biofilm Control. Published on Decoded Science on January 21, 2012. From: http://www.decodedscience.com/nitric-oxide-a-newly-discovered-potential-target-for-biofilm-control/9730
[10] Urbanczyk, H., Ast, J.C., Kaeding, A. J., Oliver, J. D., and Dunlap, P. D. (2008). Phylogenetic Analysis of the Incidence of lux Gene Horizontal Transfer in Vibrionaceae. J. Bacteriol.190:10, 3494-350. From: doi: 10.1128/JB.00101-08