Lentiviral Vectors in Gene Therapy: Difference between revisions
No edit summary |
No edit summary |
||
| Line 55: | Line 55: | ||
[] A.D.A.M. Medical Encyclopedia [Internet]. Atlanta (GA): A.D.A.M., Inc.; c1997-2011. Adrenoleukodystrophy [updated 2011 Dec; cited 2012 Dec] Available from: <http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0002165/><br> | [] A.D.A.M. Medical Encyclopedia [Internet]. Atlanta (GA): A.D.A.M., Inc.; c1997-2011. Adrenoleukodystrophy [updated 2011 Dec; cited 2012 Dec] Available from: <http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0002165/><br> | ||
[] Brady, T., S.L. Roth, N. Malani, <i>et al</i>. 2011. "A method to sequence and quantify DNA integration for monitoring outcome in gene therapy." <i>Nucleic Acids Research</i> 1-8. <br> | [] Brady, T., S.L. Roth, N. Malani, <i>et al</i>. 2011. "A method to sequence and quantify DNA integration for monitoring outcome in gene therapy." <i>Nucleic Acids Research</i> 1-8. <br> | ||
[] Breckpot, K., J.L. Aerts, K. Thielemans. 2007. "Lentiviral vectors for cancer immunotherapy: transforming infectious particles into therapeutics." </i>Gene Therapy</i> 14: 847-862. <br> | |||
[] Cartier, N., S. Hacein-Bey-Abina, C.C. Bartholomae, <i>et al</i>. 2009. "Hematopoietic stem cell gene therapy with a lentiviral vector in X-Linked Adrenoleukodystrophy." <i>Science</i> 326: 818-823. <br> | [] Cartier, N., S. Hacein-Bey-Abina, C.C. Bartholomae, <i>et al</i>. 2009. "Hematopoietic stem cell gene therapy with a lentiviral vector in X-Linked Adrenoleukodystrophy." <i>Science</i> 326: 818-823. <br> | ||
[] Connolly, J.B. 2003. "Conditionally replicating viruses in cancer therapy." </i>Gene Therapy</i> 10: 712-715. <br> | [] Connolly, J.B. 2003. "Conditionally replicating viruses in cancer therapy." </i>Gene Therapy</i> 10: 712-715. <br> | ||
Revision as of 21:10, 13 December 2012
A Viral Biorealm page on the family Lentiviral Vectors in Gene Therapy
Lentiviruses are some of the most infamous–and life threatening–vertebrate viruses. Viruses in this genus include human immunodeficiency virus (HIV), simian immunodeficiency virus (SIV), and feline immunodeficiency virus (FIV). Their exogenous characteristics make them incredibly ideal for use in gene therapy.
Gene therapy requires delivery vehicles to transfer a gene into an organism's chromosome. Lentiviral vectors are highly successful in permanently changing the target cell and thus increase the efficiency of therapeutic treatment. Hemophilia and other blood-related diseases, AIDS, and various cancers are current targets of lentiviral vectors and they can be used in conjunction with other antiviral and anticancer drugs.
Gene Therapy
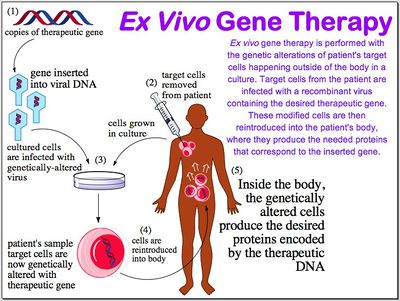
Genetic disorders result from mutated or missing genes. The goal of gene therapy is to repair the flawed DNA by completely repairing it-in most cases the expectation is a total replacement of the gene. Reparation at the source of the disorder impacts subsequent cells and tissues down the line instead of simply treating the symptoms of the disorder. In order to repair the disorder at its source, there must be an efficient way to gain entry into the cells and permanently incorporate the new gene into the target cell's genome. Various methods exist for this delivery and success depends on the nature of the disorder. What genes are involved, how many genes are involved, how the patient's immune system will react, and the physical effects of the mutated or missing gene has on a person must all be taken into account when considering gene therapy as a treatment option. If a disorder can be rectified by replacing the mutated gene, and the target cells are fairly accessible, then a suitable method of gene delivery will be devised.
"Vectors" refer to the method of gene insertion. Non-viral vectors are plasmids cultivated in bacteria and naked plasmids that are inserted directly into the cell. Non-viral vectors are not cell specific but are generally less effective in their integration into the host cell [x]. Viral vectors refer to modified viruses used to deliver DNA into target cells. Viral vectors are usually more specific to certain cell types but are limited in the size of the genome they can fit inside their capsid. Viral vectors may cause an immune response in the patient, or if the patient is inherently immune to the specific type of virus, then the success of DNA integration is diminished.
There are also various ways in which target cells can be isolated for integration. Ex vivo requires a removal of target cells from the patient, growing them in a culture, and introducing the vector into the culture. After the target cells have integrated the DNA, they are replaced into the patient. In vivo treatment simply requires the injection of the vector that is specific to target cells into the patient. Since viral vectors are commonly regarded as the most effective delivery systems, many methods have been researched and utilized various types of viruses: retroviruses, adenoviruses, adeno-associated viruses, and the herpes simplex virus are common. Viruses can also be modified to express fewer capsid proteins and to express certain proteins that are specific to target cells.
Choosing the type of virus vector depends on the nature of the genetic reparation needed. For example, retroviruses only infect dividing cells but subsequent cell lineages will contain the inserted DNA. DNA carried by adenoviruses and the herpes simplex virus will not incorporate into the host cell's DNA and may be discarded or degraded by the host cell after a period of time. For this reason, focus has shifted into retroviral vectors, specifically lentiviruses.
About Lentiviruses
Retroviruses are characterized by their use of the host cell's reverse transcriptase to create DNA from viral RNA. The DNA is then incorporated into the host's DNA genome. Viruses in the genus Lentivirus have slow incubation periods and are categorized into five different serotypes based on their host: primates, sheep/goats, horses, cats, and cattle. RELIK was found to be the first endogenous lentivirus, infecting rabbits, dating millions of years [x]. Present lentiviruses are exogenous, incorporating their DNA into a host's upon infection, and they can infect non-dividing cells, unlike most other retroviruses.
After a long incubation period and subsequent cell divisions and virus gene proliferation, lentiviruses cause prolonged illness, such as AIDS in humans. Much is known about the method of infection of lentiviruses. Glycoproteins (gp) on the envelope of the virus recognize antibody receptors on host cells and attach to them, prompting membrane fusion of the two. Viral RNA is released from the capsid in the cytoplasm of the cell and host cell reverse transcriptase beings to synthesize complementary DNA. The double-stranded DNA is taken into the nucleus and integrated into the host's genome. It can lay dormant until necessary transcription factors are present to begin transcription of the viral DNA. As long as the viral DNA lays dormant within the host's genome, the host will duplicate it during cell division and all lineages thereafter will contain the viral DNA. When the viral DNA undergoes transcription, it will leave the nucleus and the cell's mechanisms will translate the RNA into viral proteins, eventually assembling new virions that erupt from the cell, killing it.
Over time, viral DNA that has incorporated itself into the host's germline becomes endogenous to the host; human DNa is famously made up of at least 8% of endogenous retroviral DNA (that seem to have no known function)[x]. Retroviruses are also notorious for their high mutation rates during reverse transcription and this may cause complications when attempting to directly interfere with drug therapies targeted at specific viruses.
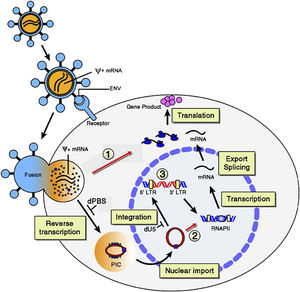
Lentiviral Vectors
Successful gene therapy requires permanent expression of the gene of interest in the target cell. The efficiency of the mode of gene delivery obviously correlates to the success of DNA integration into the host. Lentiviruses have been of interest for many years [x] because of their notoriously successful infection rate. The long terminal repeat (LTR) region of HIV-1 is a target area for modification; the sequence is a mediator for virus DNA integration into host cells. Env proteins can be modified with ligands and antibodies to target a more specific cell type [x]. In vivo cell targeting is certainly the most complicated of the delivery methods but can produce successful noninvasive results more quickly if honed.
One method involves specific antibodies on the envelope of the virus recognizing target cell surface molecules (such as αCD20 on the viral envelope recognizing CD20 on the target cell; CD20 is an antibody on a B-cell), and a separate, modified, fusion glycoprotein attaches to the host cell [x]. **FIGURE 5 FROM YANG** The virus is taken into the cell via endocytoses. The virus is within an endosome and fuses to its membrane, prompting the viral and endosome membranes to open and the capsid is released into the cell. From there, the virus 'infects' the host cell as per usual and the therapeutic gene is incorporated into the target cell.
Current Lentiviral Treatments
HIV
In one study, patients who were resistant to several combinations of drug therapies were given one dose of an ex vivo treatment of a lentiviral vector named VRX496 containing an anti-sense gene against the HIV envelope protein and intact LTRs [Levine] and monitored for several months. When the vector gene is integrated into the patient’s T-cells, RNA created from the antisense gene will block mRNA created by infectious HIV, blocking the mRNA and preventing the cell to continue making envelope proteins for the virus. Viral load, CD4 count, and other immunological factors were tested. Four out of the five patients showed overall improvement in the reduction of their viral load, had increased CD4+ T-cell counts, and increased immune response to HIV antigens [Levine]. VRX496 is a conditionally replicating vector, meaning that transcription is amplified in the presence of wilt-type HIV. As a result, conditionally replicating vectors are less inclined to transfect non-target cells and are “self-propagating” [Connolly]. These patients were treated in 2006 and are entered in Phase 2 of clinical trials, pending results for a second infusion of VRX496 T-cells [virxsys].
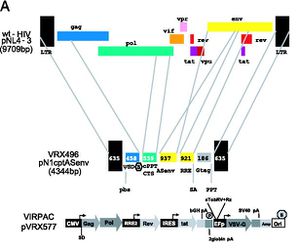
Patients who are undergoing successful HAART (highly active antiretroviral therapy) were also given a dose of VRX496 and monitored for CD4+ counts and viral load. Phase 2 of the study requires the patients to cease HAART and will be monitored for five years [UPenn]. Final data will be available in 2020.
Other RNA-interfering vectors being used in HIV patients to suppress wild-type HIV replication are being tweaked to target either the HIV genome or the mRNAs which the host cell creates [ter Brake].
X-Linked Disease
Adrenoleukodystrophy (ALD) affects males in that they cannot metabolize very long chained fatty acids. The accumulation of these fatty acids can lead to nervous system deterioration (as severe as entering into an untreatable coma), paralysis, and progressively weakening muscles [ADAM]. The ABCD1 gene encodes the ALD proteins that are transporters in the membrane of peroxisomes which normally metabolize the very long chain fatty acids and a mutation of or deletions in ABCD1 disrupts this process. Cell transplantation is the current treatment, but it must be administered at an early age. Lentiviral vectors aim to transduce hematopoietic stem cells that results in long-term expression of the corrected ALD gene in resulting hematopoietic cell lines [cartier]. Two children who had no matching donors for cord blood or human leukocyte antigen were given ex vivo doses of an HIV-derived lentivirus containing wild-type ABCD1. Many transduced CD4+ T-cells expressed ALD proteins and could successfully metabolize very long chained fatty acids. After time, expression decreased but stabilized in blood cell lines [cartier].
Potential Side Effects/Safety
Using foreign vectors in a patient always runs the risk of a severe adverse immune reaction. The "randomness" of the insertion of the lentiviral vector's DNA can potentially disrupt existing normal genes. Virus proteins on its capsid can be removed, leaving only a few and reducing the immune response of the patient. Once in the cell, "vector mobilization" may occur and infect other tissues besides the target.
Future Directions of Lentiviral Vectors
Various pharmaceutical and research companies focus solely on lentiviral gene therapy
see http://www.lentigen.com/
stem cells
vaccines
T cell therapies
proteins?
References
[] A.D.A.M. Medical Encyclopedia [Internet]. Atlanta (GA): A.D.A.M., Inc.; c1997-2011. Adrenoleukodystrophy [updated 2011 Dec; cited 2012 Dec] Available from: <http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0002165/>
[] Brady, T., S.L. Roth, N. Malani, et al. 2011. "A method to sequence and quantify DNA integration for monitoring outcome in gene therapy." Nucleic Acids Research 1-8.
[] Breckpot, K., J.L. Aerts, K. Thielemans. 2007. "Lentiviral vectors for cancer immunotherapy: transforming infectious particles into therapeutics." Gene Therapy 14: 847-862.
[] Cartier, N., S. Hacein-Bey-Abina, C.C. Bartholomae, et al. 2009. "Hematopoietic stem cell gene therapy with a lentiviral vector in X-Linked Adrenoleukodystrophy." Science 326: 818-823.
[] Connolly, J.B. 2003. "Conditionally replicating viruses in cancer therapy." Gene Therapy 10: 712-715.
[] Katzourakis, A., M. Tristem, O. G. Pybus, R. J. Gifford. 2007. "Discovery and analysis of the first endogenous lentivirus." Proceedings of the National Academy of Sciences of the United States of America. 104: 6261-6265.
[] Levine, B.L., L.M. Humeau, J. Boyer. et al. 2006. "Gene transfer in humans using a conditionally replicating lentiviral vector." PNAS 103: 17372-17377.
[] Somia N. V. , Zoppe M. , Verma I. M. 1995. "Generation of targeted retroviral vectors by using single-chain variable fragment: an approach to in vivo gene delivery." Proc. Natl. Acad. Sci. USA 92:7570–7574.
[] ter Brake, O., J.T. Westerink, B. Berkhout. 2010. "Lentiviral vector engineering for anti-HIV RNAi gene therapy." Lentivirus Gene Engineering Protocols, Methods in Molecular Biology 614:201-213.
[] "Tools of the Trade." Learn Genetics. Genetic Science Center, University of Utah, n.d. Web. Nov. 2012. <http://learn.genetics.utah.edu/content/tech/genetherapy/gttools/>.
[] Naldini, L. 1998. "Lentiviruses as gene transfer agents for delivery to non-dividing cells." Current Opinion in Biotechnology. 9: 457-463.
[] University of Pennsylvania, Pennsylvania. Evaluate the Tolerability and Therapeutic Effects of Repeated Doses of Autologous T-Cells with VRX496 in HIV. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2012 Dec]. Available from: <http://www.clinicaltrials.gov/ct2/show/study/NCT00295477?term=VRX496&rank=2>
[] VIRxSYS Corporation. A Rollover Study for Subjects Who Completed Participation in the VRX496-USA-05-002 Trial. In: ClinicalTrials.gov [Internet]. Bethesda (MD): National Library of Medicine (US). 2000- [cited 2012 Dec]. Available from: <http://www.clinicaltrials.gov/ct2/show/NCT00622232?term=VRX496&rank=1>
[] Yang, L., L. Bailey, D. Baltimore, P. Wang. 2006. "Targeting lentiviral vectors to specific cell types in vivo." Proc. Natl. Acad. Sci. USA 103:11479-11484.
Page authored by Irene McIntosh for BIOL 375 Virology, December 2012
