Wolbachia and The Biological Control of Dengue Virus: Difference between revisions
No edit summary |
|||
| (2 intermediate revisions by the same user not shown) | |||
| Line 3: | Line 3: | ||
<br> | <br> | ||
=Introduction = | =Introduction = | ||
<br> [[File:Global_dengue_risk.png| | <br> [[File:Global_dengue_risk.png|330px|thumb|right|Global dengue risk [[#References|[4]]] ]] [http://en.wikipedia.org/wiki/Dengue_virus Dengue virus] is a [http://en.wikipedia.org/wiki/Mosquito-borne_disease mosquito-borne disease] that causes an enormous health burden to people living in tropical and subtropical regions. No effective vaccine is yet available. Traditional methods for controlling the spread of mosquito-born disease, such as using bed nets and draining wetlands, failed to control the spread of dengue virus by the <i>Aedes aegytpi</i> mosquitoes because they bit during the days and only occupy urban areas. [[#References|[1]]] [[#References|[2]]] The [http://en.wiktionary.org/wiki/transinfection transinfection] of mosquitos with certain strains of the maternally inherited, [http://en.wikipedia.org/wiki/Endosymbiont endosymbiotic] bacteria [http://en.wikipedia.org/wiki/Wolbachia <i>Wolbachia</i>] seems to be a new approach in controlling the spread of dengue virus. Certain strains of this bacterium can invade and sustain themselves in wild mosquito populations, affect mosquito reproduction, reduce lifespan of its host, and interfere with pathogen replication. | ||
=Dengue virus= | =Dengue virus= | ||
[[File:Average_number_of_dengue_cases_reported_to_the_WHO.png| | [[File:Average_number_of_dengue_cases_reported_to_the_WHO.png|200px|thumb|right|Average number of dengue cases reported to the WHO [[#References|[4]]] ]] | ||
Dengue is the most rapidly spreading mosquito-borne viral disease in the world. According to the latest report by the World Health Organization (WHO), an estimated 50-100 million dengue infections occur annually and approximately 2.5 billion people live in dengue endemic countries. [[#References|[3]]] [[#References|[4]]] | Dengue is the most rapidly spreading mosquito-borne viral disease in the world. According to the latest report by the World Health Organization (WHO), an estimated 50-100 million dengue infections occur annually and approximately 2.5 billion people live in dengue endemic countries. [[#References|[3]]] [[#References|[4]]] | ||
==Structure == | ==Structure == | ||
[[File:Dengue_Virion.png | | [[File:Dengue_Virion.png |200px|thumb|right|Virion structure of Dengue virus [[#References|[23]]] ]] | ||
Dengue virus (DENV) is a small single-stranded positive sense RNA virus comprising four distinct serotypes (DENV-1 to -4). The virion of the dengue virus is enveloped, spherical with a diameter of 50nm. The surface proteins are arranged in an icosahedral-like symmetry. The immature virions contain a membrane protein precursor which is cleaved into two virus-encoded membrane proteins (M and E) once it matures. [[#References|[3]]] [[#References|[5]]] | Dengue virus (DENV) is a small single-stranded positive sense RNA virus comprising four distinct serotypes (DENV-1 to -4). The virion of the dengue virus is enveloped, spherical with a diameter of 50nm. The surface proteins are arranged in an icosahedral-like symmetry. The immature virions contain a membrane protein precursor which is cleaved into two virus-encoded membrane proteins (M and E) once it matures. [[#References|[3]]] [[#References|[5]]] | ||
| Line 19: | Line 19: | ||
==The host== | ==The host== | ||
Humans are the primary host of the virus. Although most infections are asymptomatic or subclinical, in severe cases patients can develop plasma leakage, bleeding, or organ impairment in liver, heart and the central nervous system. Primary infection can induce lifelong protective immunity to the infecting serotype, but only short-term immunity to the other serotypes. [[#References|[3]]] | Humans are the primary host of the virus. Although most infections are asymptomatic or subclinical, in severe cases patients can develop plasma leakage, bleeding, or organ impairment in liver, heart and the central nervous system. [[File:Transmission_of_Dengue.png |200px|thumb|right|Transmission of dengue virus via <i>Aedes aegypti</i>]] Primary infection can induce lifelong protective immunity to the infecting serotype, but only short-term immunity to the other serotypes. [[#References|[3]]] | ||
==Transmission== | ==Transmission== | ||
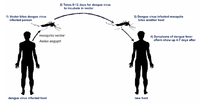
Female mosquitoes take up dengue virus during feeding on blood of [http://en.wikipedia.org/wiki/Viraemia viraemic] human. The viruses then infect the female mosquitoes’ mid-gut and replicate in various tissues before infecting the salivary glands, where the virus can then pass on to other humans during feeding. This [http://en.wikipedia.org/wiki/Incubation_period extrinsic incubation period] (EIP) usually takes about 8-12 days.[[#References|[3]]] | Female mosquitoes take up dengue virus during feeding on blood of [http://en.wikipedia.org/wiki/Viraemia viraemic] human. The viruses then infect the female mosquitoes’ mid-gut and replicate in various tissues before infecting the salivary glands, where the virus can then pass on to other humans during feeding. This [http://en.wikipedia.org/wiki/Incubation_period extrinsic incubation period] (EIP) usually takes about 8-12 days.[[#References|[3]]] | ||
=Wolbachia pipientis and its effect on dengue virus transmission = | =Wolbachia pipientis and its effect on dengue virus transmission = | ||
[http://en.wikipedia.org/wiki/Wolbachia_pipientis <i>Wolbachia pipientis</i>] is an inherited bacterial [http://en.wikipedia.org/wiki/Symbiosis symbiont] that is present in up to 76% of the estimated 2-5 million insect species on Earth. [[#References|[6]]] [[#References|[7]]] However, the bacterium is not found in mosquitoes involved in the transmission of dengue and [http://en.wikipedia.org/wiki/Malaria malaria]. [[#References|[8]]] One significant characteristic of this bacterium is its ability to induce [http://en.wikipedia.org/wiki/Cytoplasmic_incompatibility cytoplasmic incompatibility] (IC).Since Wolbachia pipientis is maternally inherited, over a few generations majority of the population will be infected with <i>Wolbachia pipientis</i> as the result of cytoplasmic incompatibility. [[#References|[9]]] [[#References|[10]]] | [[File:Cytoplasmic_incompatibility_and_wolbachia.jpeg |270px|thumb|left|Mating of infected males and uninfected females results in CI. Any other combination of un-/infected male/female crosses are compatible.[[#References|[25]]] ]] | ||
[http://en.wikipedia.org/wiki/Wolbachia_pipientis <i>Wolbachia pipientis</i>] is an inherited bacterial [http://en.wikipedia.org/wiki/Symbiosis symbiont] that is present in up to 76% of the estimated 2-5 million insect species on Earth. [[#References|[6]]] [[#References|[7]]] However, the bacterium is not found in mosquitoes involved in the transmission of dengue and [http://en.wikipedia.org/wiki/Malaria malaria]. [[#References|[8]]] One significant characteristic of this bacterium is its ability to induce [http://en.wikipedia.org/wiki/Cytoplasmic_incompatibility cytoplasmic incompatibility] (IC). Since <i>Wolbachia pipientis</i> is maternally inherited, over a few generations majority of the population will be infected with <i>Wolbachia pipientis</i> as the result of cytoplasmic incompatibility. [[#References|[9]]] [[#References|[10]]] | |||
Some Wolbachia strains lost their symbiosis ability with their hosts and can reduce the lifespan of their hosts. One such strain – wMelPop – proved to shorten the lifespan of adult [http://en.wikipedia.org/wiki/Drosophila_melanogaster <i>Drosophila melanogaster</i>] (a natural host) by up to 50%. [[#References|[11]]] Although the exact mechanisms are not known, the reduction in host’s lifespan is likely to be due to wMelPop- Wolbachia’s ability of over-replicating in the mosquito brain and causing damage in the central nervous system tissues.[[#References|[11]]] [[#References|[12]]] [[#References|[13]]] | Some <i>Wolbachia</i> strains lost their symbiosis ability with their hosts and can reduce the lifespan of their hosts. One such strain – wMelPop – proved to shorten the lifespan of adult [http://en.wikipedia.org/wiki/Drosophila_melanogaster <i>Drosophila melanogaster</i>] (a natural host) by up to 50%. [[#References|[11]]] Although the exact mechanisms are not known, the reduction in host’s lifespan is likely to be due to wMelPop- <i>Wolbachia’s</i> ability of over-replicating in the mosquito brain and causing damage in the central nervous system tissues.[[#References|[11]]] [[#References|[12]]] [[#References|[13]]] | ||
The transmission of Dengue virus by <i>Aedes aegypti </i>can be influenced by the age of the mosquito. Since the extrinsic incubation period of dengue virus in <i>Aedes aegypti</i> takes 8-12 days, only female mosquitoes that are older than the extrinsic incubation period – usually 10-14 days for dengue fever- are vectors of epidemiological importance. [[#References|[14]]] Therefore, transferring the life-shortening <i>Wolbachi</i> strain, wMelPop, to mosquito have the potential to reduce dengue transmission by eliminating the older mosquitoes that contain more viruses in their tissues. [[#References|[15]]] [[#References|[16]]] By [http://en.wikipedia.org/wiki/Microinjection microinjecting] purified wMelPop-CLA (mosquito cell-line-adapted wMelPop), which has a ~100% maternal transmission rate and CI rate, into naturally uninfected <i>A. aegypti</i> embryos researchers were able to observe promising result of a 50% reduction in adult life span of <i>A. aegypti</i>. [[#References|[12]]] | The transmission of Dengue virus by <i>Aedes aegypti </i>can be influenced by the age of the mosquito. Since the extrinsic incubation period of dengue virus in <i>Aedes aegypti</i> takes 8-12 days, only female mosquitoes that are older than the extrinsic incubation period – usually 10-14 days for dengue fever- are vectors of epidemiological importance. [[#References|[14]]] Therefore, transferring the life-shortening <i>Wolbachi</i> strain, wMelPop, to mosquito have the potential to reduce dengue transmission by eliminating the older mosquitoes that contain more viruses in their tissues. [[#References|[15]]] [[#References|[16]]] By [http://en.wikipedia.org/wiki/Microinjection microinjecting] purified wMelPop-CLA (mosquito cell-line-adapted wMelPop), which has a ~100% maternal transmission rate and CI rate, into naturally uninfected <i>A. aegypti</i> embryos researchers were able to observe promising result of a 50% reduction in adult life span of <i>A. aegypti</i>. [[#References|[12]]] | ||
Although the mechanisms are unclear, some <i>Wolbachia</i> strains such as wAlbB can also interfere with dengue virus replication in transinfected mosquitoes.[[#References|[17]]] Some hypotheses include: 1) induced expression of immune effector molecules 2) direct competition for cellular resources between dengue virus and Wolbachia in host tissues, such as salivary gland 3) use of host microRNA [[#References|[18]]] [[#References|[19]]] [[#References|[20]]] | Although the mechanisms are unclear, some <i>Wolbachia</i> strains such as wAlbB can also interfere with dengue virus replication in transinfected mosquitoes.[[#References|[17]]] Some hypotheses include: 1) induced expression of immune effector molecules 2) direct competition for cellular resources between dengue virus and Wolbachia in host tissues, such as salivary gland 3) use of host microRNA. [[#References|[18]]] [[#References|[19]]] [[#References|[20]]] | ||
=Field trial results= | =Field trial results= | ||
| Line 41: | Line 41: | ||
=References= | =References= | ||
(1) Wilder-SA, Ooi EE, Vasudevan SG, Gubler DJ. 2010. Update on dengue: epidemiology, virus evolution, antiviral drugs, and vaccine development. Curr Infect Dis Rep 12: 157-164. | (1) <b>Wilder-SA, Ooi EE, Vasudevan SG, Gubler DJ.</b> 2010. Update on dengue: epidemiology, virus evolution, antiviral drugs, and vaccine development. Curr Infect Dis Rep 12: 157-164. | ||
(2) Mullard A. 2009. Bacteria could help control dengue fever. Nature News: doi:10.1038 | (2) <b>Mullard A</b>. 2009. Bacteria could help control dengue fever. Nature News: doi:10.1038 | ||
(3) World Health Organization. 2009. Dengue: guidelines for diagnosis, treatment, prevention and control. 2009 edition. France: World Health Organization. ISBN: 978 924 1547871. | (3) <b>World Health Organization</b>. 2009. Dengue: guidelines for diagnosis, treatment, prevention and control. 2009 edition. France: World Health Organization. ISBN: 978 924 1547871. | ||
(4) World Health Organization. 2012. Global Strategy for dengue prevention and control, 2012-2020. France: World Health Orgainzation. ISBN: 9789241504034. | (4) <b>World Health Organization</b>. 2012. Global Strategy for dengue prevention and control, 2012-2020. France: World Health Orgainzation. ISBN: 9789241504034. | ||
(5) Zhang W, Chpman PR, Corve J, Johnson PR, Zhang Y, Mukhopadhyay S, Baker TS, Strauss JH, Rossmann MG, Kuhn RJ. 2003. Visualization of membrane protein domains by cryo-electron microscopy of dengue virus.Nat Struct Biol. 11: 907-912. | (5) <b>Zhang W, Chpman PR, Corve J, Johnson PR, Zhang Y, Mukhopadhyay S, Baker TS, Strauss JH, Rossmann MG, Kuhn RJ</b>. 2003. Visualization of membrane protein domains by cryo-electron microscopy of dengue virus.Nat Struct Biol. 11: 907-912. | ||
(6) Rance`s E, Ye YH, Woolfit M, McGraw EA, O’Neill SL. 2012. The relative importance of innate immune priming in Wolbachia-mediated dengue interference. PLOS Pathog. 8: e1002548. | (6) <b>Rance`s E, Ye YH, Woolfit M, McGraw EA, O’Neill SL</b>. 2012. The relative importance of innate immune priming in Wolbachia-mediated dengue interference. PLOS Pathog. 8: e1002548. | ||
(7) Hilgenboecker K, Hammerstein P, Schlattmann P, Telschow A, Werren JH. 2008. How many species are infected with Wolbachia?— A statistical analysis of current data. FEMS Microbiolo Lett 281: 215-220. | (7) <b>Hilgenboecker K, Hammerstein P, Schlattmann P, Telschow A, Werren JH</b>. 2008. How many species are infected with Wolbachia?— A statistical analysis of current data. FEMS Microbiolo Lett 281: 215-220. | ||
(8) Moreira LA, Iturbe-Ormaetex I, Jeffery JA, LU G, Pyke AT, Hedges LM, Rocha BC, Hall-Mendelin S, Day A, Riegler M, Hugo LE, Johnson KN, Kay BH, McGraw EA, van den Hurk AF, Ryan PA, O’Neill SL. 2009. A Wolbachia symbiont in Aedes aegypti limites infection with dengue, Chikungunya, and Plasmodim. Cell 7: 1268-1278. | (8) <b>Moreira LA, Iturbe-Ormaetex I, Jeffery JA, LU G, Pyke AT, Hedges LM, Rocha BC, Hall-Mendelin S, Day A, Riegler M, Hugo LE, Johnson KN, Kay BH, McGraw EA, van den Hurk AF, Ryan PA, O’Neill SL</b>. 2009. A Wolbachia symbiont in Aedes aegypti limites infection with dengue, Chikungunya, and Plasmodim. Cell 7: 1268-1278. | ||
(9) Turelli M, and Hoffmann AA. 1991. Rapid spread of an inherited incompatibility factor in California Drosophila. Nature 353: 440-442. | (9) <b>Turelli M, and Hoffmann AA</b>. 1991. Rapid spread of an inherited incompatibility factor in California Drosophila. Nature 353: 440-442. | ||
(10) Hoffmann AA, Montgomery BL, Popovici J, Iturbe-Ormaetxe I, Johnson PH, Muzzi F, Greenfield M, Durkan M, Leong YS, Dong Y, Cook H, Axford J, Callahan AG, Kenny N, Omodei C, McGraw EA, Ryan PA, Ritchie SA, Turelli M, O’Neill SL. 2011. Successful establishment of Wolbachia in Aedes popluations to suppress dengue transmission. Nature 476: 454–457. | (10) <b>Hoffmann AA, Montgomery BL, Popovici J, Iturbe-Ormaetxe I, Johnson PH, Muzzi F, Greenfield M, Durkan M, Leong YS, Dong Y, Cook H, Axford J, Callahan AG, Kenny N, Omodei C, McGraw EA, Ryan PA, Ritchie SA, Turelli M, O’Neill SL</b>. 2011. Successful establishment of Wolbachia in Aedes popluations to suppress dengue transmission. Nature 476: 454–457. | ||
(11) Min KTai and Benzer S. 1997. Wolbachia, normally a symbiont of Drosophila, can be virulent, causing degeneration and early death. Proc Natl Acad Sci USA 2: 10792 – 10796. | (11) <b>Min KTai and Benzer S</b>. 1997. Wolbachia, normally a symbiont of Drosophila, can be virulent, causing degeneration and early death. Proc Natl Acad Sci USA 2: 10792 – 10796. | ||
(12) McMeniman CJ, Lane RV, Cass BN, Fong AWC, Sidhu M, Wang YF, O’Neill SL. 2009. Stable Introduction of a Life-Shortening Wolbachia Infeciton into the Mosquito Aedes aegypti. Science 2 323: 141-144. | (12) <b>McMeniman CJ, Lane RV, Cass BN, Fong AWC, Sidhu M, Wang YF, O’Neill SL</b>. 2009. Stable Introduction of a Life-Shortening Wolbachia Infeciton into the Mosquito Aedes aegypti. Science 2 323: 141-144. | ||
(13) McGraw EA, Merritt DJ, Droller JN, O’Neill SL. 2002. Wolbachia density and virulence attenuation after transfer into a novel host. Proc. Natl. Acad. Sci. USA 99:2918-2923. | (13) <b>McGraw EA, Merritt DJ, Droller JN, O’Neill SL</b>. 2002. Wolbachia density and virulence attenuation after transfer into a novel host. Proc. Natl. Acad. Sci. USA 99:2918-2923. | ||
(14) Salazar M, Richardson JH, Sanchez-Vargas I, Olson KE, Beaty BJ. 2007. Dengue virus type 2: replication and tropisms in orally infected Aedesaegypti mosquitoes. BMC Microbiol. 7: 9. | (14) <b>Salazar M, Richardson JH, Sanchez-Vargas I, Olson KE, Beaty BJ</b>. 2007. Dengue virus type 2: replication and tropisms in orally infected Aedesaegypti mosquitoes. BMC Microbiol. 7: 9. | ||
(15) Brownlie JC, Cass BN, Riegler M, Witsenburg JJ, Iturbe-Ormaetxe I, McGraw EA, O’Neill SL. 2009. Evidence for metabolic provisioning by a common invertebrate endosymbiont, Wolbachia pipientis, durin periods of nutritional stress. PLoS Pathog 5: e1000368 | (15) <b>Brownlie JC, Cass BN, Riegler M, Witsenburg JJ, Iturbe-Ormaetxe I, McGraw EA, O’Neill SL</b>. 2009. Evidence for metabolic provisioning by a common invertebrate endosymbiont, Wolbachia pipientis, durin periods of nutritional stress. PLoS Pathog 5: e1000368 | ||
(16) Rasgon JL, Styer LM, Scott TW. 2003. Wolbachia-induced mortality as a mechanism to modulate pathogen transmission by vector arthropods. J Med Entomol 40: 125-132. | (16) <b>Rasgon JL, Styer LM, Scott TW</b>. 2003. Wolbachia-induced mortality as a mechanism to modulate pathogen transmission by vector arthropods. J Med Entomol 40: 125-132. | ||
(17) Bian G, Xu Y, Lu P, Xie Y, Xi Z. 2010. The endosymbiotic bacterium Wolbachia induces resistance to dengue virus in Aedes aegypti. PLoS Pathog 6: e1000833. | (17) <b>Bian G, Xu Y, Lu P, Xie Y, Xi Z</b>. 2010. The endosymbiotic bacterium Wolbachia induces resistance to dengue virus in Aedes aegypti. PLoS Pathog 6: e1000833. | ||
(18) Kambris Z, Cook PE, Phuc HK, Sinkins SP. 2009. Immune activation by life-shortening Wolbachia and reduced filarial competence in mosquitoes. Science 326: 134-136. | (18) <b>Kambris Z, Cook PE, Phuc HK, Sinkins SP</b>. 2009. Immune activation by life-shortening Wolbachia and reduced filarial competence in mosquitoes. Science 326: 134-136. | ||
(19) Heaton NS, Randall G. 2010. Dengue virus-induced autophagy regulates lipid metabolism. Cell Host Microb 8: 422-432. | (19) <b>Heaton NS, Randall G</b>. 2010. Dengue virus-induced autophagy regulates lipid metabolism. Cell Host Microb 8: 422-432. | ||
(20) Zhang G, Hussain M, O’Neil SL, Asgari S. 2013. Wolbachia uses a host microRNA to regulate transcripts of a methyltransferase, contributing to dengue virus inhibition in Aedesaegypti. Proc. Natl. Acad. Sci. USA 110: 10276 | (20) <b>Zhang G, Hussain M, O’Neil SL, Asgari S</b>. 2013. Wolbachia uses a host microRNA to regulate transcripts of a methyltransferase, contributing to dengue virus inhibition in Aedesaegypti. Proc. Natl. Acad. Sci. USA 110: 10276 | ||
(21) Eliminate Dengue Program. 2013. Cairns Field Trial Update – September 2013. Clayton, Victoria, Australia. Retrieved from http://www.eliminatedengue.com/library/publication/document/field_trial_update/20130919_ba_mb_trial_update_handout_combined.pdf | (21) Eliminate Dengue Program. 2013. Cairns Field Trial Update – September 2013. Clayton, Victoria, Australia. Retrieved from http://www.eliminatedengue.com/library/publication/document/field_trial_update/20130919_ba_mb_trial_update_handout_combined.pdf | ||
(22) Popovici J, Moreira LA, Poinsignon A, Iturbe-Ormaetxe I, McNaughton D, O’Neill SL. 2010. Assessing key safety concerns of a Wolbachia-based strategy to control dengue transmission by Aedes mosquitoes. Mem Inst Oswaldo Cruz 105: 957 – 964 | (22) <b>Popovici J, Moreira LA, Poinsignon A, Iturbe-Ormaetxe I, McNaughton D, O’Neill SL</b>. 2010. Assessing key safety concerns of a Wolbachia-based strategy to control dengue transmission by Aedes mosquitoes. Mem Inst Oswaldo Cruz 105: 957 – 964 | ||
(23) ViralZone:www.expasy.org/viralzone. Swiss Institute of Bioinformatics | (23) ViralZone:www.expasy.org/viralzone. Swiss Institute of Bioinformatics | ||
(24) Eliminate Dengue Program. School of Biological Sciences, Monash University. http://www.eliminatedengue.com/our-research/dengue-fever | (24) Eliminate Dengue Program. School of Biological Sciences, Monash University. http://www.eliminatedengue.com/our-research/dengue-fever | ||
Latest revision as of 05:40, 27 November 2013
By: Elisa Xiao
Introduction
Dengue virus is a mosquito-borne disease that causes an enormous health burden to people living in tropical and subtropical regions. No effective vaccine is yet available. Traditional methods for controlling the spread of mosquito-born disease, such as using bed nets and draining wetlands, failed to control the spread of dengue virus by the Aedes aegytpi mosquitoes because they bit during the days and only occupy urban areas. [1] [2] The transinfection of mosquitos with certain strains of the maternally inherited, endosymbiotic bacteria Wolbachia seems to be a new approach in controlling the spread of dengue virus. Certain strains of this bacterium can invade and sustain themselves in wild mosquito populations, affect mosquito reproduction, reduce lifespan of its host, and interfere with pathogen replication.
Dengue virus

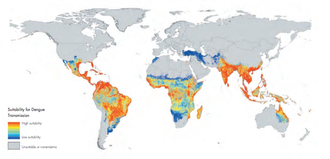
Dengue is the most rapidly spreading mosquito-borne viral disease in the world. According to the latest report by the World Health Organization (WHO), an estimated 50-100 million dengue infections occur annually and approximately 2.5 billion people live in dengue endemic countries. [3] [4]
Structure
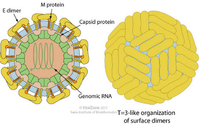
Dengue virus (DENV) is a small single-stranded positive sense RNA virus comprising four distinct serotypes (DENV-1 to -4). The virion of the dengue virus is enveloped, spherical with a diameter of 50nm. The surface proteins are arranged in an icosahedral-like symmetry. The immature virions contain a membrane protein precursor which is cleaved into two virus-encoded membrane proteins (M and E) once it matures. [3] [5]
The vectors
The various serotypes of the dengue virus are transmitted to humans through the bites of dengue-infected Aedes mosquitoes, mainly Aedes aegypti. This mosquito is commonly found in the tropical and subtropical regions and it is widely distributed around the world, mostly between latitudes 35°N and 35°S below an elevation of 1000 metres. These mosquitoes live in water-filled habitats and prefer to live in and around human habitations. [3]
The host
Humans are the primary host of the virus. Although most infections are asymptomatic or subclinical, in severe cases patients can develop plasma leakage, bleeding, or organ impairment in liver, heart and the central nervous system.
Primary infection can induce lifelong protective immunity to the infecting serotype, but only short-term immunity to the other serotypes. [3]
Transmission
Female mosquitoes take up dengue virus during feeding on blood of viraemic human. The viruses then infect the female mosquitoes’ mid-gut and replicate in various tissues before infecting the salivary glands, where the virus can then pass on to other humans during feeding. This extrinsic incubation period (EIP) usually takes about 8-12 days.[3]
Wolbachia pipientis and its effect on dengue virus transmission
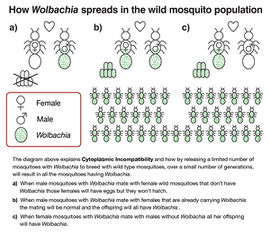
Wolbachia pipientis is an inherited bacterial symbiont that is present in up to 76% of the estimated 2-5 million insect species on Earth. [6] [7] However, the bacterium is not found in mosquitoes involved in the transmission of dengue and malaria. [8] One significant characteristic of this bacterium is its ability to induce cytoplasmic incompatibility (IC). Since Wolbachia pipientis is maternally inherited, over a few generations majority of the population will be infected with Wolbachia pipientis as the result of cytoplasmic incompatibility. [9] [10]
Some Wolbachia strains lost their symbiosis ability with their hosts and can reduce the lifespan of their hosts. One such strain – wMelPop – proved to shorten the lifespan of adult Drosophila melanogaster (a natural host) by up to 50%. [11] Although the exact mechanisms are not known, the reduction in host’s lifespan is likely to be due to wMelPop- Wolbachia’s ability of over-replicating in the mosquito brain and causing damage in the central nervous system tissues.[11] [12] [13]
The transmission of Dengue virus by Aedes aegypti can be influenced by the age of the mosquito. Since the extrinsic incubation period of dengue virus in Aedes aegypti takes 8-12 days, only female mosquitoes that are older than the extrinsic incubation period – usually 10-14 days for dengue fever- are vectors of epidemiological importance. [14] Therefore, transferring the life-shortening Wolbachi strain, wMelPop, to mosquito have the potential to reduce dengue transmission by eliminating the older mosquitoes that contain more viruses in their tissues. [15] [16] By microinjecting purified wMelPop-CLA (mosquito cell-line-adapted wMelPop), which has a ~100% maternal transmission rate and CI rate, into naturally uninfected A. aegypti embryos researchers were able to observe promising result of a 50% reduction in adult life span of A. aegypti. [12]
Although the mechanisms are unclear, some Wolbachia strains such as wAlbB can also interfere with dengue virus replication in transinfected mosquitoes.[17] Some hypotheses include: 1) induced expression of immune effector molecules 2) direct competition for cellular resources between dengue virus and Wolbachia in host tissues, such as salivary gland 3) use of host microRNA. [18] [19] [20]
Field trial results
After receiving regulatory approval from the Australian Governments a trial release of Wolbachia (wMelPop)-infected A. aegypti in July 2013 is taking place in two communities in Australia. After two months of releasing Wolbachia-infected mosquitoes, monitor results show 86% and 77% of A. aegypti mosquitoes collected in the two areas carried Wolbachia. [21]
Risk assessment
Experimental results indicate that Woblachia antigens are not injected into human through mosquito bites and no immune response is initiated in human host.[22] Lateral transfer of Woblachia to non-target species had not been observed since Woblachia are maternally transmitted; horizontal transfer was also not observed in mosquitoes’ natural spider predators. [22] However, no solid answer is yet available for the potential long-term consequences, such as the evolution of the virus in response to the presence of Woblachia.
References
(1) Wilder-SA, Ooi EE, Vasudevan SG, Gubler DJ. 2010. Update on dengue: epidemiology, virus evolution, antiviral drugs, and vaccine development. Curr Infect Dis Rep 12: 157-164.
(2) Mullard A. 2009. Bacteria could help control dengue fever. Nature News: doi:10.1038
(3) World Health Organization. 2009. Dengue: guidelines for diagnosis, treatment, prevention and control. 2009 edition. France: World Health Organization. ISBN: 978 924 1547871.
(4) World Health Organization. 2012. Global Strategy for dengue prevention and control, 2012-2020. France: World Health Orgainzation. ISBN: 9789241504034.
(5) Zhang W, Chpman PR, Corve J, Johnson PR, Zhang Y, Mukhopadhyay S, Baker TS, Strauss JH, Rossmann MG, Kuhn RJ. 2003. Visualization of membrane protein domains by cryo-electron microscopy of dengue virus.Nat Struct Biol. 11: 907-912.
(6) Rance`s E, Ye YH, Woolfit M, McGraw EA, O’Neill SL. 2012. The relative importance of innate immune priming in Wolbachia-mediated dengue interference. PLOS Pathog. 8: e1002548.
(7) Hilgenboecker K, Hammerstein P, Schlattmann P, Telschow A, Werren JH. 2008. How many species are infected with Wolbachia?— A statistical analysis of current data. FEMS Microbiolo Lett 281: 215-220.
(8) Moreira LA, Iturbe-Ormaetex I, Jeffery JA, LU G, Pyke AT, Hedges LM, Rocha BC, Hall-Mendelin S, Day A, Riegler M, Hugo LE, Johnson KN, Kay BH, McGraw EA, van den Hurk AF, Ryan PA, O’Neill SL. 2009. A Wolbachia symbiont in Aedes aegypti limites infection with dengue, Chikungunya, and Plasmodim. Cell 7: 1268-1278.
(9) Turelli M, and Hoffmann AA. 1991. Rapid spread of an inherited incompatibility factor in California Drosophila. Nature 353: 440-442.
(10) Hoffmann AA, Montgomery BL, Popovici J, Iturbe-Ormaetxe I, Johnson PH, Muzzi F, Greenfield M, Durkan M, Leong YS, Dong Y, Cook H, Axford J, Callahan AG, Kenny N, Omodei C, McGraw EA, Ryan PA, Ritchie SA, Turelli M, O’Neill SL. 2011. Successful establishment of Wolbachia in Aedes popluations to suppress dengue transmission. Nature 476: 454–457.
(11) Min KTai and Benzer S. 1997. Wolbachia, normally a symbiont of Drosophila, can be virulent, causing degeneration and early death. Proc Natl Acad Sci USA 2: 10792 – 10796.
(12) McMeniman CJ, Lane RV, Cass BN, Fong AWC, Sidhu M, Wang YF, O’Neill SL. 2009. Stable Introduction of a Life-Shortening Wolbachia Infeciton into the Mosquito Aedes aegypti. Science 2 323: 141-144.
(13) McGraw EA, Merritt DJ, Droller JN, O’Neill SL. 2002. Wolbachia density and virulence attenuation after transfer into a novel host. Proc. Natl. Acad. Sci. USA 99:2918-2923.
(14) Salazar M, Richardson JH, Sanchez-Vargas I, Olson KE, Beaty BJ. 2007. Dengue virus type 2: replication and tropisms in orally infected Aedesaegypti mosquitoes. BMC Microbiol. 7: 9.
(15) Brownlie JC, Cass BN, Riegler M, Witsenburg JJ, Iturbe-Ormaetxe I, McGraw EA, O’Neill SL. 2009. Evidence for metabolic provisioning by a common invertebrate endosymbiont, Wolbachia pipientis, durin periods of nutritional stress. PLoS Pathog 5: e1000368
(16) Rasgon JL, Styer LM, Scott TW. 2003. Wolbachia-induced mortality as a mechanism to modulate pathogen transmission by vector arthropods. J Med Entomol 40: 125-132.
(17) Bian G, Xu Y, Lu P, Xie Y, Xi Z. 2010. The endosymbiotic bacterium Wolbachia induces resistance to dengue virus in Aedes aegypti. PLoS Pathog 6: e1000833.
(18) Kambris Z, Cook PE, Phuc HK, Sinkins SP. 2009. Immune activation by life-shortening Wolbachia and reduced filarial competence in mosquitoes. Science 326: 134-136.
(19) Heaton NS, Randall G. 2010. Dengue virus-induced autophagy regulates lipid metabolism. Cell Host Microb 8: 422-432.
(20) Zhang G, Hussain M, O’Neil SL, Asgari S. 2013. Wolbachia uses a host microRNA to regulate transcripts of a methyltransferase, contributing to dengue virus inhibition in Aedesaegypti. Proc. Natl. Acad. Sci. USA 110: 10276
(21) Eliminate Dengue Program. 2013. Cairns Field Trial Update – September 2013. Clayton, Victoria, Australia. Retrieved from http://www.eliminatedengue.com/library/publication/document/field_trial_update/20130919_ba_mb_trial_update_handout_combined.pdf
(22) Popovici J, Moreira LA, Poinsignon A, Iturbe-Ormaetxe I, McNaughton D, O’Neill SL. 2010. Assessing key safety concerns of a Wolbachia-based strategy to control dengue transmission by Aedes mosquitoes. Mem Inst Oswaldo Cruz 105: 957 – 964
(23) ViralZone:www.expasy.org/viralzone. Swiss Institute of Bioinformatics
(24) Eliminate Dengue Program. School of Biological Sciences, Monash University. http://www.eliminatedengue.com/our-research/dengue-fever