Caffeine and its effect on the gut microbiome: Difference between revisions
No edit summary |
|||
| (5 intermediate revisions by the same user not shown) | |||
| Line 6: | Line 6: | ||
<b>By Riley Chlupsa</b> | <b>By Riley Chlupsa</b> | ||
The gut microbiome is defined as a grouping of microbial organisms in the gastrointestinal tract (GIT). The microbes in the human body are made up of one hundred trillion cells which significantly more than human cells in the body. The majority of these microbes are found in the gut, meaning they have a large influence on the human health and are crucial for their survival. The relationship between microbial communities and their host is entirely mutualistic. The state of the microbiome in the GIT greatly impacts human inflammation, digestion, and their immunity<ref name==aa>Bull, Matthew & Plummer, Nigel (2014). Part One: The Human Gut Microbiome in Health and Disease<i>Integrative Medicine: A Clinician’s Journal</i>, 13(6), 17-22. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4566439/</ref>. The microbiota have the ability to extract energy from resources, assist in building a protective barrier against harmful pathogens, and enhances the synthesis of amino acids and other nutrients. In return, the host provides a habitat and vital resources to help the microbes thrive. The survival of both the host and the microbiota are extremely dependent one another, but this bond is extremely sensitive until altering conditions. When the microbiota are faced with imbalances in composition, the host’s health and wellbeing will potentially be negatively affected.<ref name==aa>Bull, Matthew & Plummer, Nigel (2014). Part One: The Human Gut Microbiome in Health and Disease<i>Integrative Medicine: A Clinician’s Journal</i>, 13(6), 17-22. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4566439/</ref><ref name== | The gut microbiome is defined as a grouping of microbial organisms in the gastrointestinal tract (GIT). The microbes in the human body are made up of one hundred trillion cells which significantly more than human cells in the body. The majority of these microbes are found in the gut, meaning they have a large influence on the human health and are crucial for their survival. The relationship between microbial communities and their host is entirely mutualistic. The state of the microbiome in the GIT greatly impacts human inflammation, digestion, and their immunity<ref name==aa>Bull, Matthew & Plummer, Nigel (2014). Part One: The Human Gut Microbiome in Health and Disease<i>Integrative Medicine: A Clinician’s Journal</i>, 13(6), 17-22. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4566439/</ref>. The microbiota have the ability to extract energy from resources, assist in building a protective barrier against harmful pathogens, and enhances the synthesis of amino acids and other nutrients. In return, the host provides a habitat and vital resources to help the microbes thrive. The survival of both the host and the microbiota are extremely dependent one another, but this bond is extremely sensitive until altering conditions. When the microbiota are faced with imbalances in composition, the host’s health and wellbeing will potentially be negatively affected.<ref name==aa>Bull, Matthew & Plummer, Nigel (2014). Part One: The Human Gut Microbiome in Health and Disease<i>Integrative Medicine: A Clinician’s Journal</i>, 13(6), 17-22. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4566439/</ref><ref name==ab> Shreiner, Andrew, Kao, John, & Young, Vincent (2016). The gut microbiome in health and in disease | ||
<i>Current Opinion in Gastroenterology</i>, 31(1), 69-75. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4290017/</ref>.This microbial imbalance is called “dysbiosis”. This is specifically a disturbance to the homeostasis of the gut microbiome which changed their ability to perform metabolic functions because their composition is changed. Dysbiosis is a dangerous war within the human gut which could result in loss of important organisms, the growth of harmful organisms in the gut, and the loss of all microbial diversity leading to an inevitable end to the host’s gut microbiome. There is evidence stating that dysbiosis has been a known cause for an array of diseases such as irritable bowel disease (IBD), obesity, allergic disorders, Type 1 diabetes mellitus, autism, obesity, and colorectal cancer<ref name== | <i>Current Opinion in Gastroenterology</i>, 31(1), 69-75. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4290017/</ref>.This microbial imbalance is called “dysbiosis”. This is specifically a disturbance to the homeostasis of the gut microbiome which changed their ability to perform metabolic functions because their composition is changed. Dysbiosis is a dangerous war within the human gut which could result in loss of important organisms, the growth of harmful organisms in the gut, and the loss of all microbial diversity leading to an inevitable end to the host’s gut microbiome. There is evidence stating that dysbiosis has been a known cause for an array of diseases such as irritable bowel disease (IBD), obesity, allergic disorders, Type 1 diabetes mellitus, autism, obesity, and colorectal cancer<ref name==ac>DeGruttola, Arianna, Low, Daren, & Mizoguchi, Atsushi (2016). Current understanding of dysbiosis in disease in human and animal models<i>Oxford Academic</i>, 22(5),1137-1150. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4838534/#:~:text=This%20imbalance%20in%20the%20microbial,distribution%20%5B1%2C%204%5D</ref>. Stimulant intake potentially has largely negative consequences on the diversity and composition of the gut microbiome. The gut-brain communication is evidently altered as a result to the effects of stimulants targeting monoamine trasporters responsible in the release of adrenaline, dopamine, alter thermoregulation, and increase locomotor activity<ref name==ad>Angora-Perez, Mariana, Kuhn, Donald, Theis, Kevin, Greenberg, Jonathan, Winters, Andrew(2020). Differential effects of synthetic psychoactive cathinones and amphetamine stimulants on the gut microbiome in mice<i>Pubmed Central</i>,15(1). From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6980639/#:~:text=In%20summary%2C%20the%20gut%20microbiome,the%20gut%20microbiome%20of%20mice.</ref>. | ||
The aim of this page is to focus on a common stimulant that many individuals consume worldwide, caffeine, which is a substance found in coffee, tea, energy drinks, and many other products. Known for it’s affect of alertness, cognition, and mood, caffeine is rapidly absorbed into the GIT to exert its effects primarily through antagonism of adenosine receptors in the central nervous system<ref name== | The aim of this page is to focus on a common stimulant that many individuals consume worldwide, caffeine, which is a substance found in coffee, tea, energy drinks, and many other products. Known for it’s affect of alertness, cognition, and mood, caffeine is rapidly absorbed into the GIT to exert its effects primarily through antagonism of adenosine receptors in the central nervous system<ref name==af>Angora-Perez, Mariana, Kuhn, Donald, Theis, Kevin, Greenberg, Jonathan, Winters, Andrew(2020). Differential effects of synthetic psychoactive cathinones and amphetamine stimulants on the gut microbiome in mice<i>Pubmed Central</i>,15(1). From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6980639/#:~:text=In%20summary%2C%20the%20gut%20microbiome,the%20gut%20microbiome%20of%20mice.</ref>. According to the American Medical Association (AMA), about 85% of adults in America consume 135 milligams of caffeine daily which is equal to about 12 ounces of coffee<ref name==ag>Berg, Sarah(2024).What doctors wich patients knew about the impact of caffeine<i>American Medical Association</i>. From https://www.ama-assn.org/delivering-care/public-health/what-doctors-wish-patients-knew-about-impact-caffeine</ref>. It is very well established that caffeine is a source of energy that many humans rely on to go about their daily lives. The overuse of caffeine is greatly advertised by medical professionals and their effects on the gut microbiome, central nervous system, gut motility, and gastrointestinal function are evident. Studies have shown that these long term effects can vary depending on genetics, body weight, other medication being taken, unrelated health conditions, and the amount of regular consumption. However, the overuse of caffeine evidently affects anyone in a number of negative long term ways. Individuals who intake caffeine above the recommended amount have developed IBD, Type 1 diabetes, osteoperosis, anxiety, and many more metabolic, gastrointestinal, and mental health disorders<ref name==ah>Alcohol and Drug Foundation(2024).What is Caffeine?<i>Alcohol and Drug Foundation</i>. From https://adf.org.au/drug-facts/caffeine/#:~:text=Long%2Dterm%20effects,seizures%2C%20confusion%20or%20delirium</ref>. This may be true, but this review is intended to address caffeine’s specific impact on the microbiota in the gastrointestinal tract and the positive implications for human health. | ||
==The synthesis and degradation of caffeine== | ==The synthesis and degradation of caffeine== | ||
[[File:|thumb|350px|left| | [[File:Gutgeneex.jpeg|thumb|350px|left|Expression of Enzymes in the Bacterial Degration Pathway of Caffeine. <ref name==ai>Vega F.E., Emche S., Shao J., Simpkins A., Summers R.M., Mock M.B., Ebert D., Infante F., Aoki S., Maul J.E.(2021).Cultivation and Genome Sequencing of Bacteria Isolated From the Coffee Berry Borer (<i>Hypothenemus hampei</i>), With Emphasis on the Role of Caffeine Degradation<i>Front. Microbiol.</i>. From https://pubmed.ncbi.nlm.nih.gov/33889142/</ref>. | ||
Photo credit: [https:// | Photo credit: [https://pubmed.ncbi.nlm.nih.gov/33889142/ Front. Microbiol.]]] | ||
| Line 24: | Line 24: | ||
===Caffeine synthesis in plants=== | ===Caffeine synthesis in plants=== | ||
Caffeine is a metabolite that is derived exclusively from purine nucleotides. They will typically account for 2-5% of the dry weight of teas and 1-2% of the dry weight of coffee. The xanthine alkaloid is derived from plants<ref name== | Caffeine is a metabolite that is derived exclusively from purine nucleotides. They will typically account for 2-5% of the dry weight of teas and 1-2% of the dry weight of coffee. The xanthine alkaloid is derived from plants<ref name==aj>Ashihara H., Mizuno K., Yokota T., Crozier A(2017).Xanthine Alkaloids: Occurrence, Biosynthesis, and Function in Plants<i>Progress in the Chemistry of Organic Natural Products 105</i>, 105, 1-88. From https://pubmed.ncbi.nlm.nih.gov/28194561/</ref>. Currently, the synthesis of caffeine is derived from plants consisting of a main pathway and xanthosine pathway. The core pathway is divided into four steps. There is three methylation reactions involved and each is catalyzed by three different N-methyltransferases and a singular nucleosidase reaction calayzed from N-methylnucleosidase<ref name==ak>Ashihara H., Sano H., Yokota T., Crozier A(2008).Caffeine and Related Purine Alkaloids: Biosynthesis, Catabolism, Function and Genetic Engineering<i>Phytochemistry</i>, 69, 841-856. From https://pubmed.ncbi.nlm.nih.gov/18068204/</ref>/. Caffeine’s purine ring comes from purine nucleotides with a methyl donor that is involved in methylation is S-adenosyl-L-methionine (SAM)<ref name==al>Suzuki T(1972).The Participation of S-Adenosylmethionine in the Biosynthesis of Caffeine in the Tea Plant<i>FEBS Lett</i>, 24, 18-20. From https://core.ac.uk/download/pdf/82684049.pdf</ref>/. Now that there’s some background provided, here let’s take a closer look at each of the four steps for caffein synthesis in plants. The first route is called the de novo route which happens to be the most significant synthesis pathway of xanthosine out of the four pathways. The pathway starts with 5-phosphoribose-1-pyrophosphate (PRPP), which turns into inosine 5’-monophosphate (IMP) from a series of enzyme-catalyzed reactions until the product becomes xanthosine<ref name==am>Ashihara H., Mizuno K., Yokota T., Crozier A(2017).Xanthine Alkaloids: Occurrence, Biosynthesis, and Function in Plants<i>Progress in the Chemistry of Organic Natural Products 105</i>, 105, 1-88. From https://pubmed.ncbi.nlm.nih.gov/28194561/</ref>. The next step is the AMP route which is when xanthine nucleosides are able to be synthesized from adenine nucleosides going from AMP to IMP to XMP to xanthosine<ref name==an>Ashihara H.(1993).Purine Metabolism and the Biosynthesis of Caffeine in Maté Leaves<i>Phytochemistry</i>, 33, 1427-1430. From 10.1016/0031-9422(93)85103-X/</ref>. Another possible route of synthesis is the SAM cycle route. This is where the synthesis of xanthosine occurs through S-adenosylhomocysteine (SAH) in the SAM cycle. The pathway from SAM to xanthine nucleosides goes from SAH to adenosine to adenine to AMP to AMP to IMP to XMP and them to xanthosine<ref name==ao>Koshiishi C., Kato A., Yama S., Crozier A., Ashihara H.(2001).A New Caffeine Biosynthetic Pathway in Tea Leaves: Utilisation of Adenosine Released from the S-Adenosyl-L-Methionine Cycle<i>FEBS Lett</i>, 499, 50-54. From 10.1016/S0014-5793(01)02512-1</ref>. This route is arguably the longest pathway considering the amount of steps it takes to finally get to xanthosine. The last possible route for caffeine synthesis in plants is the GMP route. This route involves guanine nucleotides which are synthesized starting from GMP to guanosine and then to xanthosine<ref name==ap>Ashihara H., Takasawa Y., Suzuki T.(1997).Metabolic Fate of Guanosine in Higher Plants<i>Physiol. Plant</i>, 100, 909-916. From 110.1111/j.1399-3054.1997.tb00017.x.</ref>. Plants utillize the synthesis of caffeine in two major ecological ways: allelopathy, which is the inhibition of the growth of other plants and chemical defense against predators and harmful pathogens<ref name==aq>Ashihara H., Takasawa Y., Suzuki T.(1997).Metabolic Fate of Guanosine in Higher Plants<i>Physiol. Plant</i>, 100, 909-916. From 110.1111/j.1399-3054.1997.tb00017.x.</ref>. | ||
===Caffeine metabolism in bacteria=== | ===Caffeine metabolism in bacteria=== | ||
There are at least 71 bacterial strains that are capable of degrading caffeine globally. <i>Pseudomonas</i> are the most commonly studied strain that is present in the gut microbiome. There are two possible metabolic pathways that <i>Pseudomonas</i> utilize to degrade caffeine: N-terminal demethylation and C-terminal oxidation. These two pathways will coexist more often than not in bacteria, however, the N-demethylation pathway is considered to be the main route that is utilized. Diving deeper into the N-demethylation pathway, the process starts when caffeine is transported into the bacteria. Multiple N-demethylases will break down the three N-methyl bonds forming xanthine. To note, the enzymes involved in this degradation pathway are known to be inducible, referring to the ability of a gene, enzyme, or system to be activated or upregulated in response to specific stimuli, allowing organisms to adapt to the changing environmental conditions or physiologial needs. During the demethylation, caffeine will then binf to the C-terminus of N-demethylase A (NdmB). The NdmB will then catalyze the the demethylation of caffeine N-1 methylbromine, the major product, and paraxanthine, the minor product<ref name== | There are at least 71 bacterial strains that are capable of degrading caffeine globally. <i>Pseudomonas</i> are the most commonly studied strain that is present in the gut microbiome. There are two possible metabolic pathways that <i>Pseudomonas</i> utilize to degrade caffeine: N-terminal demethylation and C-terminal oxidation. These two pathways will coexist more often than not in bacteria, however, the N-demethylation pathway is considered to be the main route that is utilized. Diving deeper into the N-demethylation pathway, the process starts when caffeine is transported into the bacteria. Multiple N-demethylases will break down the three N-methyl bonds forming xanthine. To note, the enzymes involved in this degradation pathway are known to be inducible, referring to the ability of a gene, enzyme, or system to be activated or upregulated in response to specific stimuli, allowing organisms to adapt to the changing environmental conditions or physiologial needs. During the demethylation, caffeine will then binf to the C-terminus of N-demethylase A (NdmB). The NdmB will then catalyze the the demethylation of caffeine N-1 methylbromine, the major product, and paraxanthine, the minor product<ref name==ar>Vega F.E., Emche S., Shao J., Simpkins A., Summers R.M., Mock M.B., Ebert D., Infante F., Aoki S., Maul J.E.(2021).Cultivation and Genome Sequencing of Bacteria Isolated From the Coffee Berry Borer (<i>Hypothenemus hampei</i>), With Emphasis on the Role of Caffeine Degradation<i>Front. Microbiol.</i>. From https://pubmed.ncbi.nlm.nih.gov/33889142/</ref>. After caffeine is broken down into xanthine, it becomes uric acid from oxidation by xanthine oxidase. The uric acid will then go further to become hydrolyzed to allantoic acid. Then, the allantoic acid will become further hydrolyzed to urea and glyocylic acid. Then finally, urea will be decomposed into the products carbon dioxide and ammonia<ref name==as>Asano Y., Komeda T., Yamada H. (1994).Enzymes Involved in Theobromine Production from Caffeine by <i>Pseudomonas putida</i>. <i>Biosci. Biotechnol. Biochem.</i>. From https://www.tandfonline.com/doi/abs/10.1271/bbb.58.2303</ref>. The lesser pathway, C-8 oxidative degradation pathway is also present in Pseudomona coexists with the N-demethylation pathway. This pathway was discovered in 1998 in which caffeine is hydrolyzed to trimethyluric acid that is catalyzed by caffiene dehydrogenase It’s important to note qualities of genuses in bacteria that are specialized to degrade caffeine. The certain sequence characteristics can be characterized as N-Demethylase genes<ref name==at>Glück M., Lingens F. (1988).Heteroxanthinedemethylase, a New Enzyme in the Degradation of Caffeine by <i>Pseudomonas putida</i>.<i>Applied Microbiology</i>. From https://link.springer.com/article/10.1007/BF00250499</ref>. | ||
===Gene sequencing in caffeine degration=== | ===Gene sequencing in caffeine degration=== | ||
Bacterial genes that have the ability to degrade caffeine contain a certain sequence of genes. <i>Pseudomonas</i> sp. S31, S32, S37, and S60 contain the complete NdmABCDE gene within the same gene cluster. Some other types of bacteria however, only have partial N-Demethylase genes. This may be true, however, these genes are each flanked by similar genes including ABC family and other permeases, transporters, GntR family, MarR family, and LysR family regardless of the complete absence of the N-Demethylase gene<ref name== | Bacterial genes that have the ability to degrade caffeine contain a certain sequence of genes. <i>Pseudomonas</i> sp. S31, S32, S37, and S60 contain the complete NdmABCDE gene within the same gene cluster. Some other types of bacteria however, only have partial N-Demethylase genes. This may be true, however, these genes are each flanked by similar genes including ABC family and other permeases, transporters, GntR family, MarR family, and LysR family regardless of the complete absence of the N-Demethylase gene<ref name==au>Vega F.E., Emche S., Shao J., Simpkins A., Summers R.M., Mock M.B., Ebert D., Infante F., Aoki S., Maul J.E.(2021).Cultivation and Genome Sequencing of Bacteria Isolated From the Coffee Berry Borer (<i>Hypothenemus hampei</i>), With Emphasis on the Role of Caffeine Degradation<i>Front. Microbiol.</i>. From https://pubmed.ncbi.nlm.nih.gov/33889142/</ref>. This gene is about 14kb in size and contains the enzyme NdmABCDE which is used for demethylation. The open reading frame 1 (Orf1) of this gene lso happens to be involved in the degradation of formaldehyde as well<ref name==av>Retnadhas S., Gummadi S.N.(2018).Identification and Characterization of Oxidoreductase Component (NdmD) of Methylxanthine Oxygenase System in <i>Pseudomonas</i> sp. NCIM 5235<i>Applied Microbiology and Biotechnology</i>, 104(7), 3025-3036. From https://pubmed.ncbi.nlm.nih.gov/32009202/</ref>. | ||
==The effects on the gut microbiome== | ==The effects on the gut microbiome== | ||
The connection between higher caffeine intake and bacterial diversity and richness in the gut microbiome is interestingly increased. Bacterial diversity and richness are the most common indicators of a healthy gut, refelcting the presence of an array of bacterial species with diverse funtions and metabolic capabilities. Caffeine consumption has been shown to be linked to changes in gut motility, and the secretion of specific digestive enzymes which indirectly affects the overall composition of the gut microbiome<ref name== | The connection between higher caffeine intake and bacterial diversity and richness in the gut microbiome is interestingly increased. Bacterial diversity and richness are the most common indicators of a healthy gut, refelcting the presence of an array of bacterial species with diverse funtions and metabolic capabilities. Caffeine consumption has been shown to be linked to changes in gut motility, and the secretion of specific digestive enzymes which indirectly affects the overall composition of the gut microbiome<ref name==aw>Mills C.E., Tzounis X., Oruna-Concha M.-J., Mottram D.S., Gibson G.R., Spencer J.P.E.(2015).In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growth<i>The British Journal of Nutrition</i>, 113(8), 1220-1227. From https://doi.org/10.1017/S0007114514003948</ref>. There are multiple factors that support and explain how an increased caffeine intake would make this all possible. | ||
===The super vitamin B12=== | ===The super vitamin B12=== | ||
Further analysis from studies suggest that the vitamin B12 which is in coffee is a reason for this. The vitamin B12 serves as a cofactor for growth and metabolism. They assist enzymes involved in many metabolic pathways, like amino acid and nucleotide synthesis, and energy metabolism. When bacteria is able to synthesize B12, they become better equipped to perform these essential functions that result in their survivability in the gut microbiome. In addition, the presence of B12 is able to enhance both competition and cooperation of the microbiota of the gut microbiome. Bacteria that are able to synthesize B12 will gain an advantage over other bacteria in the microbiome due to the enhancing qualities of B12. In addition to increased competition, the abundance of B12 in the microbiome can also result in the cooperation of the bacteria in the microbial community because it plays the role as a shared resource that supports the growth and development of the entire microbial community<ref name== | Further analysis from studies suggest that the vitamin B12 which is in coffee is a reason for this. The vitamin B12 serves as a cofactor for growth and metabolism. They assist enzymes involved in many metabolic pathways, like amino acid and nucleotide synthesis, and energy metabolism. When bacteria is able to synthesize B12, they become better equipped to perform these essential functions that result in their survivability in the gut microbiome. In addition, the presence of B12 is able to enhance both competition and cooperation of the microbiota of the gut microbiome. Bacteria that are able to synthesize B12 will gain an advantage over other bacteria in the microbiome due to the enhancing qualities of B12. In addition to increased competition, the abundance of B12 in the microbiome can also result in the cooperation of the bacteria in the microbial community because it plays the role as a shared resource that supports the growth and development of the entire microbial community<ref name==ax>Gonzalez, Sonia, Salazar, Nuria, Gueimonde, Miguel(2020).Long-Term Coffee Consumption is Associated with Fecal Microbial Composition in Humans | ||
<i>MDPI</i>, 12(5), 1287. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7282261/</ref> | <i>MDPI</i>, 12(5), 1287. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7282261/</ref> | ||
| Line 45: | Line 45: | ||
===The reduction of <i>bacteroidetes</i>=== | ===The reduction of <i>bacteroidetes</i>=== | ||
In addition to the increased presence of B12 in the microbial community, there is a decrease in <i>bacteroidetes</i>, which is an activator that triggers inflammatory responses in the gut. There aren’t published studies yet that describe exactly how caffeine decreases <i>bacteroidetes</i>, but the decrease is apparent across many different studies revolving around obesity and gut health. They are able to degrade complex polysacchardies from dietary fiber and produce short chain fatty acids as a result<ref name== | In addition to the increased presence of B12 in the microbial community, there is a decrease in <i>bacteroidetes</i>, which is an activator that triggers inflammatory responses in the gut. There aren’t published studies yet that describe exactly how caffeine decreases <i>bacteroidetes</i>, but the decrease is apparent across many different studies revolving around obesity and gut health. They are able to degrade complex polysacchardies from dietary fiber and produce short chain fatty acids as a result<ref name==ay>Mills C.E., Tzounis X., Oruna-Concha M.-J., Mottram D.S., Gibson G.R., Spencer J.P.E.(2015).In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growth<i>The British Journal of Nutrition</i>, 113(8), 1220-1227. From https://doi.org/10.1017/S0007114514003948</ref> This may be true, however, and overabundace of <i>bacteroidetes</i> can result in many issues in the microbiota of the gut. Some studies suggest that too high of a level will result in metabolic dysregulation and weight gain. Gut barrier dysfunction is another effect of increased levels of <i>bacteroidetes</i> accompanied with the reduction of other beneficial bacteria. This will contribute to gut barrier dysfunction, which leaves the gut to become more susceptible to harmful substances entering and triggering the wrong immune responses and inflammation<ref name==az>Jaquet M., Rochat I., Moulin J., Cavin C., Bibiloni R.(2009).Impact of coffee consumption on the gut microbiota: a human volunteer study | ||
<i>International Journal of Food Microbiology</i>, 130(2), 117-121. From https://doi.org/10.1016/j.ijfoodmicro.2009.01.011</ref> So, having an increase of caffeine in your diet will reduce inflammation and other implications that will come with a high level of <i>bacteroidetes</i> in the gut. | <i>International Journal of Food Microbiology</i>, 130(2), 117-121. From https://doi.org/10.1016/j.ijfoodmicro.2009.01.011</ref> So, having an increase of caffeine in your diet will reduce inflammation and other implications that will come with a high level of <i>bacteroidetes</i> in the gut. | ||
===The increase in <i>bifidobacterium</i>=== | ===The increase in <i>bifidobacterium</i>=== | ||
<i>Bifidobacterium</i> is a genus of beneficial bacteria that is commonly found in the gut microbiome. They are considered to be a probiotic meaning they provide many health benefits to the host when they’re abundant<ref name== | <i>Bifidobacterium</i> is a genus of beneficial bacteria that is commonly found in the gut microbiome. They are considered to be a probiotic meaning they provide many health benefits to the host when they’re abundant<ref name==aaa>Mills C.E., Tzounis X., Oruna-Concha M.-J., Mottram D.S., Gibson G.R., Spencer J.P.E.(2015).In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growth<i>The British Journal of Nutrition</i>, 113(8), 1220-1227. From https://doi.org/10.1017/S0007114514003948</ref>. With an increased caffeine intake, an increase in <i>bifidobacterium</i> has also been observed which is greatly beneficial to overall health of the gut microbiome. With their probiotic properties taken into consideration, <i>bifidobacterium</i> aids in the balance and diversity of the gut microbiota. They regulate the balance by inhibiting the growth of harmful bacteria, contributing to the growth of a healthy microbial community for the host. They have the ability to produce antimicrobial compounds which enable them to better compete with pathogens. These antimicrobial compounds such as organic acids and bacteriocins, greatly inhibit the growth of pathogens. In addition, they are also known for their ability to ferment dietary fibers and some complex carbohydrates that are difficult for the body to digest. They produce short chain fatty acids to help with fermentation such as butyrate, acetate, and propionate which also all serve as an energy source for intestinal epithelial cells, promoting gut barrier function<ref name==aab>Nakayama, T., & Oishi, K.(2013).Influence of coffee (Coffea arabica) and galacto-oligosaccharide consumption on intestinal microbiota and the host responses.<i>FEMS Microbiology Letters</i>, 343(2), 161-168. From https://doi.org/10.1111/1574-6968.12142</ref>. | ||
==Caffeine and its effects on the gut-brain axis== | ==Caffeine and its effects on the gut-brain axis== | ||
The gut-brain axis refers to the communication network between the gastrointestinal tract and the central nervous system. This is an extremely complex system that controls all neural, hormonal, and immunological signaling pathways. All enable the constant communication between the gut and the brain. The gut-brain axis plays a crucial role in the regulation of an array of physiological processes. Some such as digestion, metabolism, immune function, and even mood or behhavior. The disruption of this communication could result in health conditions such as gastrointestinal disorders and even mental health disorders. Having the ability to understand the innerworking of the gut-brain axis is essential to uncovering connections between gut health and the coorelation to overall well being<ref name== | The gut-brain axis refers to the communication network between the gastrointestinal tract and the central nervous system. This is an extremely complex system that controls all neural, hormonal, and immunological signaling pathways. All enable the constant communication between the gut and the brain. The gut-brain axis plays a crucial role in the regulation of an array of physiological processes. Some such as digestion, metabolism, immune function, and even mood or behhavior. The disruption of this communication could result in health conditions such as gastrointestinal disorders and even mental health disorders. Having the ability to understand the innerworking of the gut-brain axis is essential to uncovering connections between gut health and the coorelation to overall well being<ref name==aac>Gwak, Min-Gyu, & Chang, Sun-Young(2021).Gut-Brain Connection: Microbiome, Gut Barrier, and Environmental Sensors.<i>Immune Network</i>, 21(3), 20. From https://immunenetwork.org/DOIx.php?id=10.4110/in.2021.21.e20</ref>. | ||
====The connection to the gut microbiome and the gut-brain axis==== | ====The connection to the gut microbiome and the gut-brain axis==== | ||
[[File:Gut-Brain-Axis-1.jpeg|thumb|350px|left| The Gut Brain Axis. | |||
Photo credit: [https://www.soma-analytics.com/gut-brain-axis// SOMA]]] | |||
Disruptions in microbial communities within the gut have been founded in various studies to have correlation to the development of various neurological disorders, highlighting the profound influence of the gut microbiota on brain health. Recent research has begun to uncover mechanisms that shed light on the impact of the gut microbiota and gut barrier integrity on brain function and behavior<ref name==aad>Gwak, Min-Gyu, & Chang, Sun-Young(2021).Gut-Brain Connection: Microbiome, Gut Barrier, and Environmental Sensors.<i>Immune Network</i>, 21(3), 20. From https://immunenetwork.org/DOIx.php?id=10.4110/in.2021.21.e20</ref>. Understanding the role of the gut barrier environment and the pathways of the gut-brain axis is crucial, particularly in the context of neurological and gastrointestinal diseases. Environmental sensors produced by gut bacteria play a significant role in mediating acute diseases via the gut-brain axis. By exploring these pathways and mechanisms, we can gain valuable insights into the intricate connections between the gut microbiome and brain health, potentially paving the way for novel therapeutic interventions targeting the gut-brain axis. Caffeine, the prominent stimulant found in coffee, tea, and various beverages, has gathered attention not only for its stimulating effects on alertness but also for its potential impact on gut health and the gut-brain axis. Recent research has shed light on the intricate relationship between caffeine consumption, the gut microbiome, and the bidirectional communication between the gut and the brain. One of the primary ways through which caffeine positively influences the gut-brain axis is by regulating the gut microbiome. Studies have shown that caffeine consumption can lead to alterations in the composition and diversity of the gut microbiota, as mentioned before. Specifically, caffeine has been associated with increases in the abundance of beneficial bacteria, such as <i>bifidobacterium</i>, which are known for their probiotic properties and their ability to promote gut health<ref name==aae>Mills C.E., Tzounis X., Oruna-Concha M.-J., Mottram D.S., Gibson G.R., Spencer J.P.E.(2015).In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growth<i>The British Journal of Nutrition</i>, 113(8), 1220-1227. From https://doi.org/10.1017/S0007114514003948</ref>. By fostering the growth of these beneficial microbes, caffeine may contribute to a more balanced and resilient gut microbiome. In addition, caffeine has been found to enhance gut barrier function, a critical aspect of gut health that helps prevent the leakage of harmful substances from the gut into the bloodstream. By promoting gut barrier integrity, caffeine may help reduce systemic inflammation and improve overall gut health. Furthermore, this effect caffeine has on the gut microbiome, can have indirect effects on brain function and mental well-being, as emerging evidence suggests that gut health plays a crucial role in modulating mood and cognitive function. In addition, caffeine's anti-inflammatory properties may further contribute to its positive effects on the gut-brain axis. Chronic inflammation in the gut has been implicated in various gastrointestinal disorders as well as neurological conditions, and caffeine's ability to mitigate inflammation may help alleviate symptoms associated with these disorders<ref name==aaf>Gribble, F. M., & Reimann, F.(2016).Enteroendocrine Cells: Chemosensors in the Intestinal Epithelium.<i>Annual Review of Physiology</i>, 78, 277-299. From https://doi.org/10.1146/annurev-physiol-021115-105439</ref>. While one enjoys cup of coffee or tea they can contribute to their overall health and well-being. With further research and understanding of caffeine's effects on the gut-brain axis and human health, research has found the true health benfits behind caffein consumption. | |||
===The health benefits behind caffeine consumption with regards to a healthy gut microbiome=== | ===The health benefits behind caffeine consumption with regards to a healthy gut microbiome=== | ||
In the past years, coffee has gone from being made out to be a harmful substance that affects the human body negatively in many ways. In 1991, the International Agency for Research on Cancer (IARC), the specialized cancer agency of the World Health Organization (WHO), classified coffee as “possibly carcinogenic to humans”. This assessment was made on the basis of limited evidence on the association of urinary bladder cancer and coffee consumption<ref name== | In the past years, coffee has gone from being made out to be a harmful substance that affects the human body negatively in many ways. In 1991, the International Agency for Research on Cancer (IARC), the specialized cancer agency of the World Health Organization (WHO), classified coffee as “possibly carcinogenic to humans”. This assessment was made on the basis of limited evidence on the association of urinary bladder cancer and coffee consumption<ref name==aag>Loomis, D., Guyton, K. Z., Grosse, Y., Lauby-Secretan, B., El Ghissassi, F., Bouvard, V., Benbrahim-Tallaa, L., Guha, N., Mattock, H., Straif, K., & International Agency for Research on Cancer Monograph Working Group (2016).Carcinogenicity of drinking coffee, mate, and very hot beverages.<i>The Lancet Onocology</i>, 343(2), 17(7), 877-878. From https://doi.org/10.1016/S1470-2045(16)30239-X</ref>. The trend however, was still present in the results to the study. In 2016, a re-evaluation was conduction to validate the results further based on more than 1000 observational and experimental studies, 23 scientists from 10 different countries concluded that the extensive scientific literature does show evidence of an association between coffee consumption and cancer. In addition, the IARC found that there is evidence that coffee consumption may actually help reduce occurrences of certain cancers such as colon, prostate, endometrial, melanoma, and liver<ref name==aah>Guallar, E., Blasco-Colmenares, E., Arking, D. E., & Zhao, D.(2017).Moderate Coffee Intake Can Be Part of a Healthy Diet.<i>Annals of Internal Medicine</i>,167(4), 283-284. From 283–284. https://doi.org/10.7326/M17-1503</ref>. The idea of the “coffee paradox” came into light from an observation that caffeine raises blood pressure. However, studies have proven that drinking coffee is associated with a lower risk of hypertension. As a matter of fact, daily coffee consumption is associated with a decrease in the prevalence of heart attack, despite the apparent tendency to smoke in coffee drinkers. In addition, moderate consumption of 3–4 cups of coffee a day is associated with greater longevity and lower risk of all-cause mortality<ref name==aai>Grosso, G., Stepaniak, U., Polak, M., Micek, A., Topor-Madry, R., Stefler, D., Szafraniec, K., & Pajak, A.(2016).Coffee consumption and risk of hypertension in the Polish arm of the HAPIEE cohort study.<i>European Journal of Clinical Nutrition</i>, 70(1), 109-115. From https://doi.org/10.1038/ejcn.2015.119</ref>. Coffee consumption has also evidence-based beneficial associations with metabolic diseases such as type 2 diabetes, metabolic syndrome, renal stones, and different liver conditions. Neurodegenerative diseases and mental disorders connecting to the gut-brain axis have also been observed to be decreases as a result to regular caffeine consumption such as Parkinson’s disease, Alzheimer’s disease, anxiety, and depression<ref name==aaj>Grosso, G., Godos, J., Galvano, F., & Giovannucci, E. L.(2017).Coffee, Caffeine, and Health Outcomes: An Umbrella Review.<i>Annual Review of Nutrition</i>, 70(1), 109-115. From https://doi.org/10.1146/annurev-nutr-071816-064941</ref>. Therefore, coffee consumption is proven and recommended to become a part of an individual’s healthy diet. | ||
Latest revision as of 01:49, 18 April 2024
Introduction
By Riley Chlupsa
The gut microbiome is defined as a grouping of microbial organisms in the gastrointestinal tract (GIT). The microbes in the human body are made up of one hundred trillion cells which significantly more than human cells in the body. The majority of these microbes are found in the gut, meaning they have a large influence on the human health and are crucial for their survival. The relationship between microbial communities and their host is entirely mutualistic. The state of the microbiome in the GIT greatly impacts human inflammation, digestion, and their immunity[1]. The microbiota have the ability to extract energy from resources, assist in building a protective barrier against harmful pathogens, and enhances the synthesis of amino acids and other nutrients. In return, the host provides a habitat and vital resources to help the microbes thrive. The survival of both the host and the microbiota are extremely dependent one another, but this bond is extremely sensitive until altering conditions. When the microbiota are faced with imbalances in composition, the host’s health and wellbeing will potentially be negatively affected.[1][2].This microbial imbalance is called “dysbiosis”. This is specifically a disturbance to the homeostasis of the gut microbiome which changed their ability to perform metabolic functions because their composition is changed. Dysbiosis is a dangerous war within the human gut which could result in loss of important organisms, the growth of harmful organisms in the gut, and the loss of all microbial diversity leading to an inevitable end to the host’s gut microbiome. There is evidence stating that dysbiosis has been a known cause for an array of diseases such as irritable bowel disease (IBD), obesity, allergic disorders, Type 1 diabetes mellitus, autism, obesity, and colorectal cancer[3]. Stimulant intake potentially has largely negative consequences on the diversity and composition of the gut microbiome. The gut-brain communication is evidently altered as a result to the effects of stimulants targeting monoamine trasporters responsible in the release of adrenaline, dopamine, alter thermoregulation, and increase locomotor activity[4]. The aim of this page is to focus on a common stimulant that many individuals consume worldwide, caffeine, which is a substance found in coffee, tea, energy drinks, and many other products. Known for it’s affect of alertness, cognition, and mood, caffeine is rapidly absorbed into the GIT to exert its effects primarily through antagonism of adenosine receptors in the central nervous system[5]. According to the American Medical Association (AMA), about 85% of adults in America consume 135 milligams of caffeine daily which is equal to about 12 ounces of coffee[6]. It is very well established that caffeine is a source of energy that many humans rely on to go about their daily lives. The overuse of caffeine is greatly advertised by medical professionals and their effects on the gut microbiome, central nervous system, gut motility, and gastrointestinal function are evident. Studies have shown that these long term effects can vary depending on genetics, body weight, other medication being taken, unrelated health conditions, and the amount of regular consumption. However, the overuse of caffeine evidently affects anyone in a number of negative long term ways. Individuals who intake caffeine above the recommended amount have developed IBD, Type 1 diabetes, osteoperosis, anxiety, and many more metabolic, gastrointestinal, and mental health disorders[7]. This may be true, but this review is intended to address caffeine’s specific impact on the microbiota in the gastrointestinal tract and the positive implications for human health.
The synthesis and degradation of caffeine

Before the impact of caffeine on the microbiota in the gastrointestinal tract can be further explored, it is important to understand how caffeine is degraded and synthesized by the microbiota. This is important because one must know what happens once caffeine is synthesized and how this can lead to health complications in the event of overusage. The structures and sequences that will be discussed here are important to understand the bigger picture of this review going forward.
.
Caffeine synthesis in plants
Caffeine is a metabolite that is derived exclusively from purine nucleotides. They will typically account for 2-5% of the dry weight of teas and 1-2% of the dry weight of coffee. The xanthine alkaloid is derived from plants[9]. Currently, the synthesis of caffeine is derived from plants consisting of a main pathway and xanthosine pathway. The core pathway is divided into four steps. There is three methylation reactions involved and each is catalyzed by three different N-methyltransferases and a singular nucleosidase reaction calayzed from N-methylnucleosidase[10]/. Caffeine’s purine ring comes from purine nucleotides with a methyl donor that is involved in methylation is S-adenosyl-L-methionine (SAM)[11]/. Now that there’s some background provided, here let’s take a closer look at each of the four steps for caffein synthesis in plants. The first route is called the de novo route which happens to be the most significant synthesis pathway of xanthosine out of the four pathways. The pathway starts with 5-phosphoribose-1-pyrophosphate (PRPP), which turns into inosine 5’-monophosphate (IMP) from a series of enzyme-catalyzed reactions until the product becomes xanthosine[12]. The next step is the AMP route which is when xanthine nucleosides are able to be synthesized from adenine nucleosides going from AMP to IMP to XMP to xanthosine[13]. Another possible route of synthesis is the SAM cycle route. This is where the synthesis of xanthosine occurs through S-adenosylhomocysteine (SAH) in the SAM cycle. The pathway from SAM to xanthine nucleosides goes from SAH to adenosine to adenine to AMP to AMP to IMP to XMP and them to xanthosine[14]. This route is arguably the longest pathway considering the amount of steps it takes to finally get to xanthosine. The last possible route for caffeine synthesis in plants is the GMP route. This route involves guanine nucleotides which are synthesized starting from GMP to guanosine and then to xanthosine[15]. Plants utillize the synthesis of caffeine in two major ecological ways: allelopathy, which is the inhibition of the growth of other plants and chemical defense against predators and harmful pathogens[16].
Caffeine metabolism in bacteria
There are at least 71 bacterial strains that are capable of degrading caffeine globally. Pseudomonas are the most commonly studied strain that is present in the gut microbiome. There are two possible metabolic pathways that Pseudomonas utilize to degrade caffeine: N-terminal demethylation and C-terminal oxidation. These two pathways will coexist more often than not in bacteria, however, the N-demethylation pathway is considered to be the main route that is utilized. Diving deeper into the N-demethylation pathway, the process starts when caffeine is transported into the bacteria. Multiple N-demethylases will break down the three N-methyl bonds forming xanthine. To note, the enzymes involved in this degradation pathway are known to be inducible, referring to the ability of a gene, enzyme, or system to be activated or upregulated in response to specific stimuli, allowing organisms to adapt to the changing environmental conditions or physiologial needs. During the demethylation, caffeine will then binf to the C-terminus of N-demethylase A (NdmB). The NdmB will then catalyze the the demethylation of caffeine N-1 methylbromine, the major product, and paraxanthine, the minor product[17]. After caffeine is broken down into xanthine, it becomes uric acid from oxidation by xanthine oxidase. The uric acid will then go further to become hydrolyzed to allantoic acid. Then, the allantoic acid will become further hydrolyzed to urea and glyocylic acid. Then finally, urea will be decomposed into the products carbon dioxide and ammonia[18]. The lesser pathway, C-8 oxidative degradation pathway is also present in Pseudomona coexists with the N-demethylation pathway. This pathway was discovered in 1998 in which caffeine is hydrolyzed to trimethyluric acid that is catalyzed by caffiene dehydrogenase It’s important to note qualities of genuses in bacteria that are specialized to degrade caffeine. The certain sequence characteristics can be characterized as N-Demethylase genes[19].
Gene sequencing in caffeine degration
Bacterial genes that have the ability to degrade caffeine contain a certain sequence of genes. Pseudomonas sp. S31, S32, S37, and S60 contain the complete NdmABCDE gene within the same gene cluster. Some other types of bacteria however, only have partial N-Demethylase genes. This may be true, however, these genes are each flanked by similar genes including ABC family and other permeases, transporters, GntR family, MarR family, and LysR family regardless of the complete absence of the N-Demethylase gene[20]. This gene is about 14kb in size and contains the enzyme NdmABCDE which is used for demethylation. The open reading frame 1 (Orf1) of this gene lso happens to be involved in the degradation of formaldehyde as well[21].
The effects on the gut microbiome
The connection between higher caffeine intake and bacterial diversity and richness in the gut microbiome is interestingly increased. Bacterial diversity and richness are the most common indicators of a healthy gut, refelcting the presence of an array of bacterial species with diverse funtions and metabolic capabilities. Caffeine consumption has been shown to be linked to changes in gut motility, and the secretion of specific digestive enzymes which indirectly affects the overall composition of the gut microbiome[22]. There are multiple factors that support and explain how an increased caffeine intake would make this all possible.
The super vitamin B12
Further analysis from studies suggest that the vitamin B12 which is in coffee is a reason for this. The vitamin B12 serves as a cofactor for growth and metabolism. They assist enzymes involved in many metabolic pathways, like amino acid and nucleotide synthesis, and energy metabolism. When bacteria is able to synthesize B12, they become better equipped to perform these essential functions that result in their survivability in the gut microbiome. In addition, the presence of B12 is able to enhance both competition and cooperation of the microbiota of the gut microbiome. Bacteria that are able to synthesize B12 will gain an advantage over other bacteria in the microbiome due to the enhancing qualities of B12. In addition to increased competition, the abundance of B12 in the microbiome can also result in the cooperation of the bacteria in the microbial community because it plays the role as a shared resource that supports the growth and development of the entire microbial community[23]
The reduction of bacteroidetes
In addition to the increased presence of B12 in the microbial community, there is a decrease in bacteroidetes, which is an activator that triggers inflammatory responses in the gut. There aren’t published studies yet that describe exactly how caffeine decreases bacteroidetes, but the decrease is apparent across many different studies revolving around obesity and gut health. They are able to degrade complex polysacchardies from dietary fiber and produce short chain fatty acids as a result[24] This may be true, however, and overabundace of bacteroidetes can result in many issues in the microbiota of the gut. Some studies suggest that too high of a level will result in metabolic dysregulation and weight gain. Gut barrier dysfunction is another effect of increased levels of bacteroidetes accompanied with the reduction of other beneficial bacteria. This will contribute to gut barrier dysfunction, which leaves the gut to become more susceptible to harmful substances entering and triggering the wrong immune responses and inflammation[25] So, having an increase of caffeine in your diet will reduce inflammation and other implications that will come with a high level of bacteroidetes in the gut.
The increase in bifidobacterium
Bifidobacterium is a genus of beneficial bacteria that is commonly found in the gut microbiome. They are considered to be a probiotic meaning they provide many health benefits to the host when they’re abundant[26]. With an increased caffeine intake, an increase in bifidobacterium has also been observed which is greatly beneficial to overall health of the gut microbiome. With their probiotic properties taken into consideration, bifidobacterium aids in the balance and diversity of the gut microbiota. They regulate the balance by inhibiting the growth of harmful bacteria, contributing to the growth of a healthy microbial community for the host. They have the ability to produce antimicrobial compounds which enable them to better compete with pathogens. These antimicrobial compounds such as organic acids and bacteriocins, greatly inhibit the growth of pathogens. In addition, they are also known for their ability to ferment dietary fibers and some complex carbohydrates that are difficult for the body to digest. They produce short chain fatty acids to help with fermentation such as butyrate, acetate, and propionate which also all serve as an energy source for intestinal epithelial cells, promoting gut barrier function[27].
Caffeine and its effects on the gut-brain axis
The gut-brain axis refers to the communication network between the gastrointestinal tract and the central nervous system. This is an extremely complex system that controls all neural, hormonal, and immunological signaling pathways. All enable the constant communication between the gut and the brain. The gut-brain axis plays a crucial role in the regulation of an array of physiological processes. Some such as digestion, metabolism, immune function, and even mood or behhavior. The disruption of this communication could result in health conditions such as gastrointestinal disorders and even mental health disorders. Having the ability to understand the innerworking of the gut-brain axis is essential to uncovering connections between gut health and the coorelation to overall well being[28].
The connection to the gut microbiome and the gut-brain axis
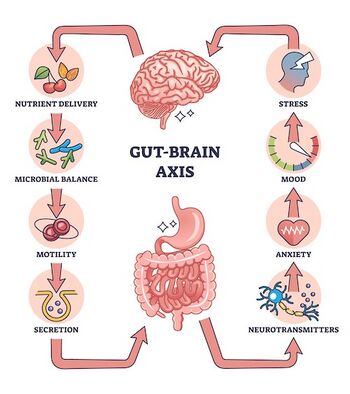
Disruptions in microbial communities within the gut have been founded in various studies to have correlation to the development of various neurological disorders, highlighting the profound influence of the gut microbiota on brain health. Recent research has begun to uncover mechanisms that shed light on the impact of the gut microbiota and gut barrier integrity on brain function and behavior[29]. Understanding the role of the gut barrier environment and the pathways of the gut-brain axis is crucial, particularly in the context of neurological and gastrointestinal diseases. Environmental sensors produced by gut bacteria play a significant role in mediating acute diseases via the gut-brain axis. By exploring these pathways and mechanisms, we can gain valuable insights into the intricate connections between the gut microbiome and brain health, potentially paving the way for novel therapeutic interventions targeting the gut-brain axis. Caffeine, the prominent stimulant found in coffee, tea, and various beverages, has gathered attention not only for its stimulating effects on alertness but also for its potential impact on gut health and the gut-brain axis. Recent research has shed light on the intricate relationship between caffeine consumption, the gut microbiome, and the bidirectional communication between the gut and the brain. One of the primary ways through which caffeine positively influences the gut-brain axis is by regulating the gut microbiome. Studies have shown that caffeine consumption can lead to alterations in the composition and diversity of the gut microbiota, as mentioned before. Specifically, caffeine has been associated with increases in the abundance of beneficial bacteria, such as bifidobacterium, which are known for their probiotic properties and their ability to promote gut health[30]. By fostering the growth of these beneficial microbes, caffeine may contribute to a more balanced and resilient gut microbiome. In addition, caffeine has been found to enhance gut barrier function, a critical aspect of gut health that helps prevent the leakage of harmful substances from the gut into the bloodstream. By promoting gut barrier integrity, caffeine may help reduce systemic inflammation and improve overall gut health. Furthermore, this effect caffeine has on the gut microbiome, can have indirect effects on brain function and mental well-being, as emerging evidence suggests that gut health plays a crucial role in modulating mood and cognitive function. In addition, caffeine's anti-inflammatory properties may further contribute to its positive effects on the gut-brain axis. Chronic inflammation in the gut has been implicated in various gastrointestinal disorders as well as neurological conditions, and caffeine's ability to mitigate inflammation may help alleviate symptoms associated with these disorders[31]. While one enjoys cup of coffee or tea they can contribute to their overall health and well-being. With further research and understanding of caffeine's effects on the gut-brain axis and human health, research has found the true health benfits behind caffein consumption.
The health benefits behind caffeine consumption with regards to a healthy gut microbiome
In the past years, coffee has gone from being made out to be a harmful substance that affects the human body negatively in many ways. In 1991, the International Agency for Research on Cancer (IARC), the specialized cancer agency of the World Health Organization (WHO), classified coffee as “possibly carcinogenic to humans”. This assessment was made on the basis of limited evidence on the association of urinary bladder cancer and coffee consumption[32]. The trend however, was still present in the results to the study. In 2016, a re-evaluation was conduction to validate the results further based on more than 1000 observational and experimental studies, 23 scientists from 10 different countries concluded that the extensive scientific literature does show evidence of an association between coffee consumption and cancer. In addition, the IARC found that there is evidence that coffee consumption may actually help reduce occurrences of certain cancers such as colon, prostate, endometrial, melanoma, and liver[33]. The idea of the “coffee paradox” came into light from an observation that caffeine raises blood pressure. However, studies have proven that drinking coffee is associated with a lower risk of hypertension. As a matter of fact, daily coffee consumption is associated with a decrease in the prevalence of heart attack, despite the apparent tendency to smoke in coffee drinkers. In addition, moderate consumption of 3–4 cups of coffee a day is associated with greater longevity and lower risk of all-cause mortality[34]. Coffee consumption has also evidence-based beneficial associations with metabolic diseases such as type 2 diabetes, metabolic syndrome, renal stones, and different liver conditions. Neurodegenerative diseases and mental disorders connecting to the gut-brain axis have also been observed to be decreases as a result to regular caffeine consumption such as Parkinson’s disease, Alzheimer’s disease, anxiety, and depression[35]. Therefore, coffee consumption is proven and recommended to become a part of an individual’s healthy diet.
Conclusions
Exploring the impact of caffeine on the gut microbiome sheds light on its wide array of positive effects on human health. Caffeine plays a significant role in shaping the composition and diversity of the gut microbiota. Studies have revealed that caffeine consumption can lead to favorable alterations in the gut microbiome, including an increase in beneficial bacteria such as bifidobacterium. These beneficial microbes contribute to gut health by promoting a balanced microbial community and enhancing gut barrier function. Furthermore, caffeine's positive influence on the gut microbiome extends to the gut-brain axis, a complex communication network between the gastrointestinal tract and the central nervous system. By regulating the gut microbiota, caffeine has the ability to indirectly modulate brain function and mental well-being. Additionally, caffeine's anti-inflammatory properties contribute to its positive effects on the gut-brain axis, potentially alleviating symptoms associated with gastrointestinal and neurological disorders. Despite historical concerns regarding the health implications of caffeine consumption, emerging evidence suggests numerous health benefits associated with moderate caffeine intake. The "coffee paradox," which highlights the discrepancy between caffeine's blood pressure-elevating effects and its association with a lower risk of hypertension, underscores the complexity of caffeine's impact on human health. Furthermore, regular coffee consumption has been linked to a reduced risk of various chronic diseases, including metabolic disorders, neurodegenerative diseases, and mental health disorders. In light of these findings, incorporating caffeine into a balanced diet may offer numerous health benefits, particularly when consumed in moderation. As research continues to unravel the intricate relationship between caffeine, the gut microbiome, and overall human health, a better understanding of caffeine's mechanisms of action may pave the way for novel therapeutic interventions targeting gut-brain axis dysfunctions and chronic disease prevention.
References
- ↑ 1.0 1.1 Bull, Matthew & Plummer, Nigel (2014). Part One: The Human Gut Microbiome in Health and DiseaseIntegrative Medicine: A Clinician’s Journal, 13(6), 17-22. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4566439/
- ↑ Shreiner, Andrew, Kao, John, & Young, Vincent (2016). The gut microbiome in health and in disease Current Opinion in Gastroenterology, 31(1), 69-75. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4290017/
- ↑ DeGruttola, Arianna, Low, Daren, & Mizoguchi, Atsushi (2016). Current understanding of dysbiosis in disease in human and animal modelsOxford Academic, 22(5),1137-1150. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4838534/#:~:text=This%20imbalance%20in%20the%20microbial,distribution%20%5B1%2C%204%5D
- ↑ Angora-Perez, Mariana, Kuhn, Donald, Theis, Kevin, Greenberg, Jonathan, Winters, Andrew(2020). Differential effects of synthetic psychoactive cathinones and amphetamine stimulants on the gut microbiome in micePubmed Central,15(1). From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6980639/#:~:text=In%20summary%2C%20the%20gut%20microbiome,the%20gut%20microbiome%20of%20mice.
- ↑ Angora-Perez, Mariana, Kuhn, Donald, Theis, Kevin, Greenberg, Jonathan, Winters, Andrew(2020). Differential effects of synthetic psychoactive cathinones and amphetamine stimulants on the gut microbiome in micePubmed Central,15(1). From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6980639/#:~:text=In%20summary%2C%20the%20gut%20microbiome,the%20gut%20microbiome%20of%20mice.
- ↑ Berg, Sarah(2024).What doctors wich patients knew about the impact of caffeineAmerican Medical Association. From https://www.ama-assn.org/delivering-care/public-health/what-doctors-wish-patients-knew-about-impact-caffeine
- ↑ Alcohol and Drug Foundation(2024).What is Caffeine?Alcohol and Drug Foundation. From https://adf.org.au/drug-facts/caffeine/#:~:text=Long%2Dterm%20effects,seizures%2C%20confusion%20or%20delirium
- ↑ Vega F.E., Emche S., Shao J., Simpkins A., Summers R.M., Mock M.B., Ebert D., Infante F., Aoki S., Maul J.E.(2021).Cultivation and Genome Sequencing of Bacteria Isolated From the Coffee Berry Borer (Hypothenemus hampei), With Emphasis on the Role of Caffeine DegradationFront. Microbiol.. From https://pubmed.ncbi.nlm.nih.gov/33889142/
- ↑ Ashihara H., Mizuno K., Yokota T., Crozier A(2017).Xanthine Alkaloids: Occurrence, Biosynthesis, and Function in PlantsProgress in the Chemistry of Organic Natural Products 105, 105, 1-88. From https://pubmed.ncbi.nlm.nih.gov/28194561/
- ↑ Ashihara H., Sano H., Yokota T., Crozier A(2008).Caffeine and Related Purine Alkaloids: Biosynthesis, Catabolism, Function and Genetic EngineeringPhytochemistry, 69, 841-856. From https://pubmed.ncbi.nlm.nih.gov/18068204/
- ↑ Suzuki T(1972).The Participation of S-Adenosylmethionine in the Biosynthesis of Caffeine in the Tea PlantFEBS Lett, 24, 18-20. From https://core.ac.uk/download/pdf/82684049.pdf
- ↑ Ashihara H., Mizuno K., Yokota T., Crozier A(2017).Xanthine Alkaloids: Occurrence, Biosynthesis, and Function in PlantsProgress in the Chemistry of Organic Natural Products 105, 105, 1-88. From https://pubmed.ncbi.nlm.nih.gov/28194561/
- ↑ Ashihara H.(1993).Purine Metabolism and the Biosynthesis of Caffeine in Maté LeavesPhytochemistry, 33, 1427-1430. From 10.1016/0031-9422(93)85103-X/
- ↑ Koshiishi C., Kato A., Yama S., Crozier A., Ashihara H.(2001).A New Caffeine Biosynthetic Pathway in Tea Leaves: Utilisation of Adenosine Released from the S-Adenosyl-L-Methionine CycleFEBS Lett, 499, 50-54. From 10.1016/S0014-5793(01)02512-1
- ↑ Ashihara H., Takasawa Y., Suzuki T.(1997).Metabolic Fate of Guanosine in Higher PlantsPhysiol. Plant, 100, 909-916. From 110.1111/j.1399-3054.1997.tb00017.x.
- ↑ Ashihara H., Takasawa Y., Suzuki T.(1997).Metabolic Fate of Guanosine in Higher PlantsPhysiol. Plant, 100, 909-916. From 110.1111/j.1399-3054.1997.tb00017.x.
- ↑ Vega F.E., Emche S., Shao J., Simpkins A., Summers R.M., Mock M.B., Ebert D., Infante F., Aoki S., Maul J.E.(2021).Cultivation and Genome Sequencing of Bacteria Isolated From the Coffee Berry Borer (Hypothenemus hampei), With Emphasis on the Role of Caffeine DegradationFront. Microbiol.. From https://pubmed.ncbi.nlm.nih.gov/33889142/
- ↑ Asano Y., Komeda T., Yamada H. (1994).Enzymes Involved in Theobromine Production from Caffeine by Pseudomonas putida. Biosci. Biotechnol. Biochem.. From https://www.tandfonline.com/doi/abs/10.1271/bbb.58.2303
- ↑ Glück M., Lingens F. (1988).Heteroxanthinedemethylase, a New Enzyme in the Degradation of Caffeine by Pseudomonas putida.Applied Microbiology. From https://link.springer.com/article/10.1007/BF00250499
- ↑ Vega F.E., Emche S., Shao J., Simpkins A., Summers R.M., Mock M.B., Ebert D., Infante F., Aoki S., Maul J.E.(2021).Cultivation and Genome Sequencing of Bacteria Isolated From the Coffee Berry Borer (Hypothenemus hampei), With Emphasis on the Role of Caffeine DegradationFront. Microbiol.. From https://pubmed.ncbi.nlm.nih.gov/33889142/
- ↑ Retnadhas S., Gummadi S.N.(2018).Identification and Characterization of Oxidoreductase Component (NdmD) of Methylxanthine Oxygenase System in Pseudomonas sp. NCIM 5235Applied Microbiology and Biotechnology, 104(7), 3025-3036. From https://pubmed.ncbi.nlm.nih.gov/32009202/
- ↑ Mills C.E., Tzounis X., Oruna-Concha M.-J., Mottram D.S., Gibson G.R., Spencer J.P.E.(2015).In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growthThe British Journal of Nutrition, 113(8), 1220-1227. From https://doi.org/10.1017/S0007114514003948
- ↑ Gonzalez, Sonia, Salazar, Nuria, Gueimonde, Miguel(2020).Long-Term Coffee Consumption is Associated with Fecal Microbial Composition in Humans MDPI, 12(5), 1287. From https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7282261/
- ↑ Mills C.E., Tzounis X., Oruna-Concha M.-J., Mottram D.S., Gibson G.R., Spencer J.P.E.(2015).In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growthThe British Journal of Nutrition, 113(8), 1220-1227. From https://doi.org/10.1017/S0007114514003948
- ↑ Jaquet M., Rochat I., Moulin J., Cavin C., Bibiloni R.(2009).Impact of coffee consumption on the gut microbiota: a human volunteer study International Journal of Food Microbiology, 130(2), 117-121. From https://doi.org/10.1016/j.ijfoodmicro.2009.01.011
- ↑ Mills C.E., Tzounis X., Oruna-Concha M.-J., Mottram D.S., Gibson G.R., Spencer J.P.E.(2015).In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growthThe British Journal of Nutrition, 113(8), 1220-1227. From https://doi.org/10.1017/S0007114514003948
- ↑ Nakayama, T., & Oishi, K.(2013).Influence of coffee (Coffea arabica) and galacto-oligosaccharide consumption on intestinal microbiota and the host responses.FEMS Microbiology Letters, 343(2), 161-168. From https://doi.org/10.1111/1574-6968.12142
- ↑ Gwak, Min-Gyu, & Chang, Sun-Young(2021).Gut-Brain Connection: Microbiome, Gut Barrier, and Environmental Sensors.Immune Network, 21(3), 20. From https://immunenetwork.org/DOIx.php?id=10.4110/in.2021.21.e20
- ↑ Gwak, Min-Gyu, & Chang, Sun-Young(2021).Gut-Brain Connection: Microbiome, Gut Barrier, and Environmental Sensors.Immune Network, 21(3), 20. From https://immunenetwork.org/DOIx.php?id=10.4110/in.2021.21.e20
- ↑ Mills C.E., Tzounis X., Oruna-Concha M.-J., Mottram D.S., Gibson G.R., Spencer J.P.E.(2015).In vitro colonic metabolism of coffee and chlorogenic acid results in selective changes in human faecal microbiota growthThe British Journal of Nutrition, 113(8), 1220-1227. From https://doi.org/10.1017/S0007114514003948
- ↑ Gribble, F. M., & Reimann, F.(2016).Enteroendocrine Cells: Chemosensors in the Intestinal Epithelium.Annual Review of Physiology, 78, 277-299. From https://doi.org/10.1146/annurev-physiol-021115-105439
- ↑ Loomis, D., Guyton, K. Z., Grosse, Y., Lauby-Secretan, B., El Ghissassi, F., Bouvard, V., Benbrahim-Tallaa, L., Guha, N., Mattock, H., Straif, K., & International Agency for Research on Cancer Monograph Working Group (2016).Carcinogenicity of drinking coffee, mate, and very hot beverages.The Lancet Onocology, 343(2), 17(7), 877-878. From https://doi.org/10.1016/S1470-2045(16)30239-X
- ↑ Guallar, E., Blasco-Colmenares, E., Arking, D. E., & Zhao, D.(2017).Moderate Coffee Intake Can Be Part of a Healthy Diet.Annals of Internal Medicine,167(4), 283-284. From 283–284. https://doi.org/10.7326/M17-1503
- ↑ Grosso, G., Stepaniak, U., Polak, M., Micek, A., Topor-Madry, R., Stefler, D., Szafraniec, K., & Pajak, A.(2016).Coffee consumption and risk of hypertension in the Polish arm of the HAPIEE cohort study.European Journal of Clinical Nutrition, 70(1), 109-115. From https://doi.org/10.1038/ejcn.2015.119
- ↑ Grosso, G., Godos, J., Galvano, F., & Giovannucci, E. L.(2017).Coffee, Caffeine, and Health Outcomes: An Umbrella Review.Annual Review of Nutrition, 70(1), 109-115. From https://doi.org/10.1146/annurev-nutr-071816-064941
Authored for BIOL 238 Microbiology, taught by Joan Slonczewski,at Kenyon College,2024
